Sodium naphthalene
| |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium naphthalenide | |
| Systematic IUPAC name
Sodium naphthalen-1-ide | |
| Other names
sodium naphthalenide, sodium naphthalide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.020.420 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
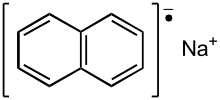
| Na+[C10H8]− | |
| Molar mass | 151.164 g·mol−1 |
| Appearance | Deep green crystals |
| Related compounds | |
Other anions
|
Lithium naphthalene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium naphthalene is an organic salt with the chemical formula Na+[C10H8]−. In the research laboratory, it is used as a reductant in the synthesis of organic, organometallic, and inorganic chemistry. It is usually generated in situ. When isolated, it invariably crystallizes as a solvate with ligands bound to Na+.[1]
Preparation and properties[edit]

The alkali metal naphthalene salts are prepared by stirring the metal with naphthalene in an ethereal solvent, usually as tetrahydrofuran or dimethoxyethane. The resulting salt is dark green.[2][3][4] The anion is a radical, giving a strong EPR signal near g = 2.0. Its deep green color arises from absorptions centered at 463 and 735 nm.
Several solvates of sodium naphthalenide have been characterized by X-ray crystallography. The effects are subtle, the outer pair of CH−CH bonds contract by 3 pm and the other nine C−C bonds elongate by 2–3 pm. The net effect is that reduction weakens the bonding.[5][6]
Reactions[edit]
Redox[edit]
With a reduction potential near −2.5 V vs NHE, the naphthalene radical anion is a strong reducing agent.[1]
Protonation[edit]
The anion is strongly basic, and a typical degradation pathway involves reaction with water and related protic sources such as alcohols. These reactions afford dihydronaphthalene:
As a ligand[edit]
Alkali metal salts of the naphthalene radical anion are used to prepare complexes of naphthalene.[7]
Related reagents[edit]
References[edit]
- ^ a b Connelly, Neil G.; Geiger, William E. (1996). "Chemical Redox Agents for Organometallic Chemistry". Chemical Reviews. 96 (2): 877–910. doi:10.1021/cr940053x. PMID 11848774.
- ^ Corey, E. J.; Gross, Andrew W. (1987). "tert-Butyl-tert-octylamine". Org. Syntheses. 65: 166. doi:10.15227/orgsyn.065.0166.
- ^ Cotton, F. Albert; Wilkinson, Geoffrey (1988), Advanced Inorganic Chemistry (5th ed.), New York: Wiley-Interscience, p. 139, ISBN 0-471-84997-9
- ^ Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 111. ISBN 978-0-08-022057-4.
- ^ Bock, Hans; Arad, Claudia; Näther, Christian; Havlas, Zdenek (1995). "The Structures of Solvent-Separated Naphthalene and Anthracene Radical Anions". J. Chem. Soc., Chem. Commun. (23): 2393–2394. doi:10.1039/C39950002393.
- ^ Castillo, Maximiliano; Metta-Magaña, Alejandro J.; Fortier, Skye (2016). "Isolation of Gravimetrically Quantifiable Alkali Metal Arenides Using 18-Crown-6". New Journal of Chemistry. 40 (3): 1923–1926. doi:10.1039/C5NJ02841H.
- ^ Ellis, John E. (2019). "The Chatt Reaction: Conventional Routes to homoleptic Arenemetalates of d-Block Elements". Dalton Transactions. 48 (26): 9538–9563. doi:10.1039/C8DT05029E. PMID 30724934. S2CID 73436073.
