Dissolved inorganic carbon: Difference between revisions
Appearance
Content deleted Content added
Epipelagic (talk | contribs) remove material ported to total inorganic carbon |
Epipelagic (talk | contribs) add replacement text including copy from total carbon |
||
| Line 3: | Line 3: | ||
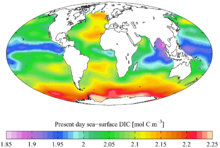
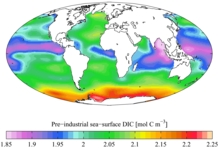
[[File:CC-BY icon.svg|50px]] Material was copied from this source, which is available under a [https://creativecommons.org/licenses/by/4.0/ Creative Commons Attribution 4.0 International License].</ref> }} Spatial distributions of DIC and nDIC. (a) DIC (normalized to year 2005); (b) salinity-normalized DIC (nDIC, DIC normalized to reference year of 2005 and salinity of 35) in the surface global ocean. The latitudinal trends are clear, particularly for nDIC.]] |
[[File:CC-BY icon.svg|50px]] Material was copied from this source, which is available under a [https://creativecommons.org/licenses/by/4.0/ Creative Commons Attribution 4.0 International License].</ref> }} Spatial distributions of DIC and nDIC. (a) DIC (normalized to year 2005); (b) salinity-normalized DIC (nDIC, DIC normalized to reference year of 2005 and salinity of 35) in the surface global ocean. The latitudinal trends are clear, particularly for nDIC.]] |
||
'''Dissolved inorganic carbon''' ('''DIC''') is the sum of the [[inorganic]] [[carbon]] species in a [[solution]]. |
'''Dissolved inorganic carbon''' ('''DIC''') is the sum of the [[inorganic]] [[carbon]] species in a [[solution]]. Carbon compounds can be distinguished as either organic or inorganic, and as dissolved or particulate, depending on their composition. Organic carbon forms the backbone of key component of organic compounds such as – [[protein]]s, [[lipid]]s, [[carbohydrate]]s, and [[nucleic acid]]s. |
||
Inorganic carbon is found primarily in simple compounds such as carbon dioxide, carbonic acid, bicarbonate, and carbonate (CO<sub>2</sub>, H<sub>2</sub>CO<sub>3</sub>, HCO<sub>3</sub><sup>−</sup>, CO<sub>3</sub><sup>2−</sup> respectively). Dissolved inorganic carbon (DIC) includes three major aqueous species, CO<sub>2</sub>, HCO<sub>3</sub>- ,CO<sub>3</sub><sup>2−</sup>, and to a lesser extent their complexes in solution with metal ions.<ref>Mackenzie FT and Lerman A (2006) [https://books.google.com/books?id=TiGgJip0l64C&pg=PA11&dq=%22Dissolved+Inorganic+Carbon%22&hl=en&sa=X&ved=2ahUKEwi78oGYu-rqAhXEZSsKHdCEDHgQ6AEwCHoECCYQAg#v=onepage&q=%22Dissolved%20Inorganic%20Carbon%22&f=false ''Carbon in the Geobiosphere: Earth's Outer Shell''], Springer Science & Business Media. {{ISBN|9781402042386}}.</ref> |
|||
[[File:Annual DOC and DIC fluxes in the Tanguro Ranch watershed.jpg|thumb|upright=1.8|left| {{center|'''Annual DOC and DIC fluxes in the Tanguro Ranch<ref>[https://www.arcgis.com/apps/MapJournal/index.html?appid=a91eb090e012491b99086e111278c127 Environmental Research at Tanguro Ranch, Brazil] ''Esri''. Retrieved 26 July 2020.</ref> watershed'''}} Average annual flux of DOC and DIC in rainfall, throughfall, stemflow, overland flow, and stream flow.<ref>Neu, V., Ward, N.D., Krusche, A.V. and Nill, C. (2016) "Dissolved organic and inorganic carbon flow paths in an Amazonian transitional forest". ''Frontiers in Marine Science'', '''3''': 114. {{doi|10.3389/fmars.2016.00114}}. [[File:CC-BY icon.svg|50px]] Material was copied from this source, which is available under a [https://creativecommons.org/licenses/by/4.0/ Creative Commons Attribution 4.0 International License].</ref>]] |
[[File:Annual DOC and DIC fluxes in the Tanguro Ranch watershed.jpg|thumb|upright=1.8|left| {{center|'''Annual DOC and DIC fluxes in the Tanguro Ranch<ref>[https://www.arcgis.com/apps/MapJournal/index.html?appid=a91eb090e012491b99086e111278c127 Environmental Research at Tanguro Ranch, Brazil] ''Esri''. Retrieved 26 July 2020.</ref> watershed'''}} Average annual flux of DOC and DIC in rainfall, throughfall, stemflow, overland flow, and stream flow.<ref>Neu, V., Ward, N.D., Krusche, A.V. and Nill, C. (2016) "Dissolved organic and inorganic carbon flow paths in an Amazonian transitional forest". ''Frontiers in Marine Science'', '''3''': 114. {{doi|10.3389/fmars.2016.00114}}. [[File:CC-BY icon.svg|50px]] Material was copied from this source, which is available under a [https://creativecommons.org/licenses/by/4.0/ Creative Commons Attribution 4.0 International License].</ref>]] |
||
Revision as of 09:19, 26 July 2020

Spatial distribution of ocean surface DIC [1]
Spatial distributions of DIC and nDIC. (a) DIC (normalized to year 2005); (b) salinity-normalized DIC (nDIC, DIC normalized to reference year of 2005 and salinity of 35) in the surface global ocean. The latitudinal trends are clear, particularly for nDIC.Dissolved inorganic carbon (DIC) is the sum of the inorganic carbon species in a solution. Carbon compounds can be distinguished as either organic or inorganic, and as dissolved or particulate, depending on their composition. Organic carbon forms the backbone of key component of organic compounds such as – proteins, lipids, carbohydrates, and nucleic acids.
Inorganic carbon is found primarily in simple compounds such as carbon dioxide, carbonic acid, bicarbonate, and carbonate (CO2, H2CO3, HCO3−, CO32− respectively). Dissolved inorganic carbon (DIC) includes three major aqueous species, CO2, HCO3- ,CO32−, and to a lesser extent their complexes in solution with metal ions.[2]

Annual DOC and DIC fluxes in the Tanguro Ranch[3] watershed
Average annual flux of DOC and DIC in rainfall, throughfall, stemflow, overland flow, and stream flow.[4]Pre-industrial (1700s)
See also
- Alkalinity (total alkalinity; AT)
- Bjerrum plot
- Dissolved organic carbon
- Fugacity (carbon dioxide fugacity; fCO2)
- Ocean acidification
- pH
- Total organic carbon
References
- ^ Wu, Y., Hain, M.P., Humphreys, M.P., Hartman, S. and Tyrrell, T. (2019) "What drives the latitudinal gradient in open-ocean surface dissolved inorganic carbon concentration?" Biogeosciences, 16(13): 2661–2681. doi:10.5194/bg-16-2661-2019.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Mackenzie FT and Lerman A (2006) Carbon in the Geobiosphere: Earth's Outer Shell, Springer Science & Business Media. ISBN 9781402042386.
- ^ Environmental Research at Tanguro Ranch, Brazil Esri. Retrieved 26 July 2020.
- ^ Neu, V., Ward, N.D., Krusche, A.V. and Nill, C. (2016) "Dissolved organic and inorganic carbon flow paths in an Amazonian transitional forest". Frontiers in Marine Science, 3: 114. doi:10.3389/fmars.2016.00114.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.