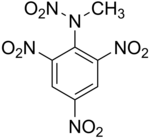
Tetryl
| |
| Names | |
|---|---|
| IUPAC name
N-methyl-N,2,4,6-tetranitroaniline
| |
| Other names
nitramine, tetralite, tetril
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.006.848 |
PubChem CID
|
|
| UN number | 0208 |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C7H5N5O8 | |
| Molar mass | 287.15 g/mol |
| Appearance | Odorless yellow crystalline solid |
| Density | 1.73 g/cm3 |
| Melting point | 129.5 °C (265.1 °F; 402.6 K) |
| Boiling point | Decomposes at 187 °C |
| Explosive data | |
| Shock sensitivity | Sensitive |
| Friction sensitivity | Sensitive |
| RE factor | 1.25 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetryl (C7H5N5O8) is a sensitive explosive compound used to make detonators and explosive booster charges.
Tetryl is an odorless yellow crystalline solid that is not found naturally in the environment. Under certain conditions, tetryl can exist as dust in air. It is slightly soluble in water and in other liquids. It is essentially TNT with an added nitrogen atom and nitro group.
Tetryl was used mainly during World Wars I and II and later conflicts. Tetryl is usually used on its own, though can sometimes be found in compositions such as tetrytol. Tetryl is no longer manufactured or used in the United States, but can still be found in legacy munitions such as the M14 anti-personnel landmine.
Production
Tetryl is produced by action of a mix of concentrated nitric acid and sulfuric acid on dimethylaniline.
See also
References
- Cooper, Paul W., Explosives Engineering, New York: Wiley-VCH, 1996. ISBN 0-471-18636-8
