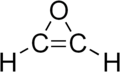Oxirene
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Oxirene[1] | |||
| Systematic IUPAC name
Oxacyclopropene | |||
| Other names
Epoxyethene
Ethyne oxide Acetylene oxide | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| MeSH | C012469 | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H2O | |||
| Molar mass | 42.04 g/mol | ||
| Related compounds | |||
Related molecules with 3-membered rings
|
Ethylene oxide cyclopropane cyclopropene aziridine thiirane thiirene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Oxirene is a heterocyclic chemical compound which contains an unsaturated three-membered ring containing two carbon atoms and one oxygen atom. The molecule was synthesized in low temperature ices and detected upon sublimation by isomer selective photoionization reflectron time-of-flight mass spectrometry.[2]
Quantum chemical computational techniques found the configuration to be extremely strained and proposed an antiaromatic 4π electron system, as such oxirene is expected to be very high energy.[3][4]
Experimental indications exist that substituted oxirenes (as intermediates or transition states) may be involved in carbonylcarbene rearrangements observed in the Wolff rearrangement.[5] Computational evidence also point to the intermediacy of oxirenes in the ozonolysis of alkynes.[6]
References
- ^ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 146. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ Wang, Jia; Marks, Joshua; Turner, Andrew; Mebel, Alexander; Eckhardt, Andre; Kaiser, Ralf (March 2023). "Gas-phase detection of oxirene". Science Advances. 9 (10): eadg1134. Bibcode:2023SciA....9G1134W. doi:10.1126/sciadv.adg1134. PMC 10005165. PMID 36897943.
- ^ Mawhinney, Robert C; Goddard, John D (2003). "Assessment of density functional theory for the prediction of the nature of the oxirene stationary point". Journal of Molecular Structure: THEOCHEM. 629 (1–3): 263–270. doi:10.1016/S0166-1280(03)00198-2.
- ^ Lewars, Errol G. (2011). Computational Chemistry - Introduction to the Theory and Applications of Molecular and Quantum Mechanics (2nd ed.). Springer. doi:10.1007/978-90-481-3862-3. ISBN 978-90-481-3862-3.
- ^ Kirmse, Wolfgang (2002). "100 Years of the Wolff Rearrangement". European Journal of Organic Chemistry. 2002 (14): 2193–2256. doi:10.1002/1099-0690(200207)2002:14<2193::AID-EJOC2193>3.0.CO;2-D.
- ^ Cremer, Dieter; Crehuet, Ramon; Anglada, Josep (June 2001). "The Ozonolysis of AcetyleneA Quantum Chemical Investigation". Journal of the American Chemical Society. 123 (25): 6127–6141. doi:10.1021/ja010166f. ISSN 0002-7863. PMID 11414847.