Cellular respiration
This article needs additional citations for verification. (September 2014) |

Cellular respiration is a set of metabolic reactions and processes that take place in the cells of organisms to convert chemical energy from oxygen molecules[1] or nutrients into adenosine triphosphate (ATP), and then release waste products.[2] The reactions involved in respiration are catabolic reactions, which break large molecules into smaller ones, releasing energy because weak high-energy bonds, in particular in molecular oxygen,[3] are replaced by stronger bonds in the products. Respiration is one of the key ways a cell releases chemical energy to fuel cellular activity. The overall reaction occurs in a series of biochemical steps, some of which are redox reactions. Although cellular respiration is technically a combustion reaction, it clearly does not resemble one when it occurs in a living cell because of the slow, controlled release of energy from the series of reactions.
Nutrients that are commonly used by animal and plant cells in respiration include sugar, amino acids and fatty acids, and the most common oxidizing agent providing most of the chemical energy is molecular oxygen (O2).[1] The chemical energy stored in ATP (the bond of its third phosphate group to the rest of the molecule can be broken allowing more stable products to form, thereby releasing energy for use by the cell) can then be used to drive processes requiring energy, including biosynthesis, locomotion or transport of molecules across cell membranes.
Aerobic respiration
Aerobic respiration requires oxygen (O2) in order to create ATP. Although carbohydrates, fats, and proteins are consumed as reactants, aerobic respiration is the preferred method of pyruvate breakdown in glycolysis, and requires pyruvate to the mitochondria in order to be fully oxidized by the citric acid cycle. The products of this process are carbon dioxide and water, and the energy transferred is used to break bonds in ADP to add a third phosphate group to form ATP (adenosine triphosphate), by substrate-level phosphorylation, NADH and FADH2
| Simplified reaction: | C6H12O6 (s) + 6 O2 (g) → 6 CO2 (g) + 6 H2O (l) + heat |
| ΔG = −2880 kJ per mol of C6H12O6 |
The negative ΔG indicates that the reaction can occur spontaneously.
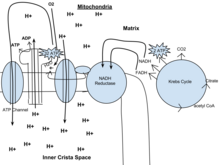
The potential of NADH and FADH2 is converted to more ATP through an electron transport chain with oxygen and protons (hydrogen) as the "terminal electron acceptors".[1] Most of the ATP produced by aerobic cellular respiration is made by oxidative phosphorylation. The energy of O2 [1] released is used to create a chemiosmotic potential by pumping protons across a membrane. This potential is then used to drive ATP synthase and produce ATP from ADP and a phosphate group. Biology textbooks often state that 38 ATP molecules can be made per oxidized glucose molecule during cellular respiration (2 from glycolysis, 2 from the Krebs cycle, and about 34 from the electron transport system).[4] However, this maximum yield is never quite reached because of losses due to leaky membranes as well as the cost of moving pyruvate and ADP into the mitochondrial matrix, and current estimates range around 29 to 30 ATP per glucose.[4]
Aerobic metabolism is up to 15 times more efficient than anaerobic metabolism (which yields 2 molecules ATP per 1 molecule glucose) because the double bond in O2 is of higher energy than other double bonds or pairs of single bonds in other common molecules in the biosphere.[3] However, some anaerobic organisms, such as methanogens are able to continue with anaerobic respiration, yielding more ATP by using other inorganic molecules (not oxygen) of high energy as final electron acceptors in the electron transport chain. They share the initial pathway of glycolysis but aerobic metabolism continues with the Krebs cycle and oxidative phosphorylation. The post-glycolytic reactions take place in the mitochondria in eukaryotic cells, and in the cytoplasm in prokaryotic cells.
Glycolysis
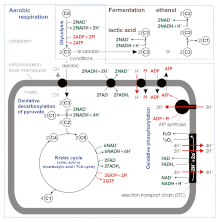
Glycolysis is a metabolic pathway that takes place in the cytosol of cells in all living organisms. Glycolysis can be literally translated as "sugar splitting"[5], and occurs with or without the presence of oxygen. In aerobic conditions, the process converts one molecule of glucose into two molecules of pyruvate (pyruvic acid), generating energy in the form of two net molecules of ATP. Four molecules of ATP per glucose are actually produced, however, two are consumed as part of the preparatory phase. The initial phosphorylation of glucose is required to increase the reactivity (decrease its stability) in order for the molecule to be cleaved into two pyruvate molecules by the enzyme aldolase. During the pay-off phase of glycolysis, four phosphate groups are transferred to ADP by substrate-level phosphorylation to make four ATP, and two NADH are produced when the pyruvate is oxidized. The overall reaction can be expressed this way:
- Glucose + 2 NAD+ + 2 Pi + 2 ADP → 2 pyruvate + 2 H+ + 2 NADH + 2 ATP + 2 H+ + 2 H2O + energy
Starting with glucose, 1 ATP is used to donate a phosphate to glucose to produce glucose 6-phosphate. Glycogen can be converted into glucose 6-phosphate as well with the help of glycogen phosphorylase. During energy metabolism, glucose 6-phosphate becomes fructose 6-phosphate. An additional ATP is used to phosphorylate fructose 6-phosphate into fructose 1,6-bisphosphate by the help of phosphofructokinase. Fructose 1,6-biphosphate then splits into two phosphorylated molecules with three carbon chains which later degrades into pyruvate.
Oxidative decarboxylation of pyruvate
Pyruvate is oxidized to acetyl-CoA and CO2 by the pyruvate dehydrogenase complex (PDC). The PDC contains multiple copies of three enzymes and is located in the mitochondria of eukaryotic cells and in the cytosol of prokaryotes. In the conversion of pyruvate to acetyl-CoA, one molecule of NADH and one molecule of CO2 is formed.
Citric acid cycle
This is also called the Krebs cycle or the tricarboxylic acid cycle. When oxygen is present, acetyl-CoA is produced from the pyruvate molecules created from glycolysis. Once acetyl-CoA is formed, aerobic or anaerobic respiration can occur.[6] When oxygen is present, the mitochondria will undergo aerobic respiration which leads to the Krebs cycle. However, if oxygen is not present, fermentation of the pyruvate molecule will occur. In the presence of oxygen, when acetyl-CoA is produced, the molecule then enters the citric acid cycle (Krebs cycle) inside the mitochondrial matrix, and is oxidized to CO2 while at the same time reducing NAD to NADH. NADH can be used by the electron transport chain to create further ATP as part of oxidative phosphorylation. To fully oxidize the equivalent of one glucose molecule, two acetyl-CoA must be metabolized by the Krebs cycle. Two low-energy waste products, H2O and CO2, are created during this cycle.
The citric acid cycle is an 8-step process involving 18 different enzymes and co-enzymes.[6] During the cycle, acetyl-CoA (2 carbons) + oxaloacetate (4 carbons) yields citrate (6 carbons), which is rearranged to a more reactive form called isocitrate (6 carbons). Isocitrate is modified to become α-ketoglutarate (5 carbons), succinyl-CoA, succinate, fumarate, malate, and, finally, oxaloacetate.
The net gain from one cycle is 3 NADH and 1 FADH2 as hydrogen- (proton plus electron)-carrying compounds and 1 high-energy GTP, which may subsequently be used to produce ATP. Thus, the total yield from 1 glucose molecule (2 pyruvate molecules) is 6 NADH, 2 FADH2, and 2 ATP.
Oxidative phosphorylation
In eukaryotes, oxidative phosphorylation occurs in the mitochondrial cristae. It comprises the electron transport chain that establishes a proton gradient (chemiosmotic potential) across the boundary of the inner membrane by oxidizing the NADH produced from the Krebs cycle. ATP is synthesized by the ATP synthase enzyme when the chemiosmotic gradient is used to drive the phosphorylation of ADP. The electron transfer is driven by the chemical energy of exogenous oxygen[1] and, with the addition of two protons, water is formed.
Efficiency of ATP production
The table below describes the reactions involved when one glucose molecule is fully oxidized into carbon dioxide. It is assumed that all the reduced coenzymes are oxidized by the electron transport chain and used for oxidative phosphorylation.
| Step | coenzyme yield | ATP yield | Source of ATP |
|---|---|---|---|
| Glycolysis preparatory phase | −2 | Phosphorylation of glucose and fructose 6-phosphate uses two ATP from the cytoplasm. | |
| Glycolysis pay-off phase | 4 | Substrate-level phosphorylation | |
| 2 NADH | 3 or 5 | Oxidative phosphorylation : Each NADH produces net 1.5 ATP (instead of usual 2.5) due to NADH transport over the mitochondrial membrane | |
| Oxidative decarboxylation of pyruvate | 2 NADH | 5 | Oxidative phosphorylation |
| Krebs cycle | 2 | Substrate-level phosphorylation | |
| 6 NADH | 15 | Oxidative phosphorylation | |
| 2 FADH2 | 3 | Oxidative phosphorylation | |
| Total yield | 30 or 32 ATP | From the complete oxidation of one glucose molecule to carbon dioxide and oxidation of all the reduced coenzymes. | |
Although there is a theoretical yield of 38 ATP molecules per glucose during cellular respiration, such conditions are generally not realized because of losses such as the cost of moving pyruvate (from glycolysis), phosphate, and ADP (substrates for ATP synthesis) into the mitochondria. All are actively transported using carriers that utilize the stored energy in the proton electrochemical gradient.
- Pyruvate is taken up by a specific, low Km transporter to bring it into the mitochondrial matrix for oxidation by the pyruvate dehydrogenase complex.
- The phosphate carrier (PiC) mediates the electroneutral exchange (antiport) of phosphate (H2PO4−; Pi) for OH− or symport of phosphate and protons (H+) across the inner membrane, and the driving force for moving phosphate ions into the mitochondria is the proton motive force.
- The ATP-ADP translocase (also called adenine nucleotide translocase, ANT) is an antiporter and exchanges ADP and ATP across the inner membrane. The driving force is due to the ATP (−4) having a more negative charge than the ADP (−3), and thus it dissipates some of the electrical component of the proton electrochemical gradient.
The outcome of these transport processes using the proton electrochemical gradient is that more than 3 H+ are needed to make 1 ATP. Obviously this reduces the theoretical efficiency of the whole process and the likely maximum is closer to 28–30 ATP molecules.[4] In practice the efficiency may be even lower because the inner membrane of the mitochondria is slightly leaky to protons.[7] Other factors may also dissipate the proton gradient creating an apparently leaky mitochondria. An uncoupling protein known as thermogenin is expressed in some cell types and is a channel that can transport protons. When this protein is active in the inner membrane it short circuits the coupling between the electron transport chain and ATP synthesis. The potential energy from the proton gradient is not used to make ATP but generates heat. This is particularly important in brown fat thermogenesis of newborn and hibernating mammals.
According to some of newer sources the ATP yield during aerobic respiration is not 36–38, but only about 30–32 ATP molecules / 1 molecule of glucose [8], because:
- ATP : NADH+H+ and ATP : FADH2 ratios during the oxidative phosphorylation appear to be not 3 and 2, but 2.5 and 1.5 respectively. Unlike in the substrate-level phosphorylation, the stoichiometry here is difficult to establish.
- ATP synthase produces 1 ATP / 3 H+. However the exchange of matrix ATP for cytosolic ADP and Pi (antiport with OH− or symport with H+) mediated by ATP–ADP translocase and phosphate carrier consumes 1 H+ / 1 ATP as a result of regeneration of the transmembrane potential changed during this transfer, so the net ratio is 1 ATP : 4 H+.
- The mitochondrial electron transport chain proton pump transfers across the inner membrane 10 H+ / 1 NADH+H+ (4 + 2 + 4) or 6 H+ / 1 FADH2 (2 + 4).
- So the final stoichiometry is
- 1 NADH+H+ : 10 H+ : 10/4 ATP = 1 NADH+H+ : 2.5 ATP
- 1 FADH2 : 6 H+ : 6/4 ATP = 1 FADH2 : 1.5 ATP
- ATP : NADH+H+ coming from glycolysis ratio during the oxidative phosphorylation is
- 1.5, as for FADH2, if hydrogen atoms (2H++2e−) are transferred from cytosolic NADH+H+ to mitochondrial FAD by the glycerol phosphate shuttle located in the inner mitochondrial membrane.
- 2.5 in case of malate-aspartate shuttle transferring hydrogen atoms from cytosolic NADH+H+ to mitochondrial NAD+
So finally we have, per molecule of glucose
- Substrate-level phosphorylation: 2 ATP from glycolysis + 2 ATP (directly GTP) from Krebs cycle
- Oxidative phosphorylation
- 2 NADH+H+ from glycolysis: 2 × 1.5 ATP (if glycerol phosphate shuttle transfers hydrogen atoms) or 2 × 2.5 ATP (malate-aspartate shuttle)
- 2 NADH+H+ from the oxidative decarboxylation of pyruvate and 6 from Krebs cycle: 8 × 2.5 ATP
- 2 FADH2 from the Krebs cycle: 2 × 1.5 ATP
Altogether this gives 4 + 3 (or 5) + 20 + 3 = 30 (or 32) ATP per molecule of glucose
These figures may still require further tweaking as new structural details become available. The above value of 3 H+/ATP for the synthase assumes that the synthase translocates 9 protons, and produces 3 ATP, per rotation. The number of protons depends on the number of c subunits in the Fo c-ring, and it is now known that this is 10 in yeast Fo[9] and 8 for vertebrates.[10] Including one H+ for the transport reactions, this means that synthesis of one ATP requires 1+10/3=4.33 protons in yeast and 1+8/3 = 3.67 in vertebrates. This would imply that in human mitochondria the 10 protons from oxidizing NADH would produce 2.72 ATP (instead of 2.5) and the 6 protons from oxidizing succinate or ubiquinol would produce 1.64 ATP (instead of 1.5). This is consistent with experimental results within the margin of error described in a recent review.[11]
The total ATP yield in ethanol or lactic acid fermentation is only 2 molecules coming from glycolysis, because pyruvate is not transferred to the mitochondrion and finally oxidized to the carbon dioxide (CO2), but reduced to ethanol or lactic acid in the cytoplasm.[8]
Fermentation
Without oxygen, pyruvate (pyruvic acid) is not metabolized by cellular respiration but undergoes a process of fermentation. The pyruvate is not transported into the mitochondrion, but remains in the cytoplasm, where it is converted to waste products that may be removed from the cell. This serves the purpose of oxidizing the electron carriers so that they can perform glycolysis again and removing the excess pyruvate. Fermentation oxidizes NADH to NAD+ so it can be re-used in glycolysis. In the absence of oxygen, fermentation prevents the buildup of NADH in the cytoplasm and provides NAD+ for glycolysis. This waste product varies depending on the organism. In skeletal muscles, the waste product is lactic acid. This type of fermentation is called lactic acid fermentation. In strenuous exercise, when energy demands exceed energy supply, the respiratory chain cannot process all of the hydrogen atoms joined by NADH. During anaerobic glycolysis, NAD+ regenerates when pairs of hydrogen combine with pyruvate to form lactate. Lactate formation is catalyzed by lactate dehydrogenase in a reversible reaction. Lactate can also be used as an indirect precursor for liver glycogen. During recovery, when oxygen becomes available, NAD+ attaches to hydrogen from lactate to form ATP. In yeast, the waste products are ethanol and carbon dioxide. This type of fermentation is known as alcoholic or ethanol fermentation. The ATP generated in this process is made by substrate-level phosphorylation, which does not require oxygen.
Fermentation is less efficient at using the energy from glucose: only 2 ATP are produced per glucose, compared to the 38 ATP per glucose nominally produced by aerobic respiration. This is because most of the energy of aerobic respiration derives from O2 with its relatively weak, high-energy double bond.[3][1] Glycolytic ATP, however, is created more quickly. For prokaryotes to continue a rapid growth rate when they are shifted from an aerobic environment to an anaerobic environment, they must increase the rate of the glycolytic reactions. For multicellular organisms, during short bursts of strenuous activity, muscle cells use fermentation to supplement the ATP production from the slower aerobic respiration, so fermentation may be used by a cell even before the oxygen levels are depleted, as is the case in sports that do not require athletes to pace themselves, such as sprinting.
Anaerobic respiration
Cellular respiration is the process by which biological fuels are oxidised in the presence of a high-energy inorganic electron acceptor (such as oxygen[1]) to produce large amounts of energy, to drive the bulk production of ATP.
Anaerobic respiration is used by some microorganisms in which neither oxygen (aerobic respiration) nor pyruvate derivatives (fermentation) is the high-energy final electron acceptor. Rather, an inorganic acceptor such as sulfate or nitrate is used. Such organisms are typically found in unusual places such as underwater caves or near hydrothermal vents at the bottom of the ocean.
In July 2019, a scientific study of Kidd Mine in Canada discovered sulfur-breathing organisms which live 7900 feet below the surface, and which breathe sulfur in order to survive. These organisms are also remarkable due to consuming minerals such as pyrite as their food source. [12][13][14]
See also
- Maintenance respiration: maintenance as a functional component of cellular respiration
- Microphysiometry
- Pasteur point
- Respirometry: research tool to explore cellular respiration
- Tetrazolium chloride: cellular respiration indicator
- Complex 1: NADH:ubiquinone oxidoreductes
References
- ^ a b c d e f g Schmidt-Rohr, K. (2020). "Oxygen Is the High-Energy Molecule Powering Complex Multicellular Life: Fundamental Corrections to Traditional Bioenergetics” ACS Omega 5: 2221-2233. http://dx.doi.org/10.1021/acsomega.9b03352
- ^ Bailey, Regina. "Cellular Respiration". Archived from the original on 2012-05-05.
- ^ a b c Schmidt-Rohr, K. (2015). "Why Combustions Are Always Exothermic, Yielding About 418 kJ per Mole of O2", J. Chem. Educ. 92: 2094-2099. http://dx.doi.org/10.1021/acs.jchemed.5b00333
- ^ a b c Rich, P. R. (2003). "The molecular machinery of Keilin's respiratory chain". Biochemical Society Transactions. 31 (Pt 6): 1095–1105. doi:10.1042/BST0311095. PMID 14641005.
- ^ Reece1 Urry2 Cain3 Wasserman4 Minorsky5 Jackson6, Jane1 Lisa2 Michael3 Steven4 Peter5 Robert6 (2010). Campbell Biology Ninth Edition. Pearson Education, Inc. p. 168.
{{cite book}}: CS1 maint: numeric names: authors list (link) - ^ a b "Cellular Respiration" (PDF). Archived (PDF) from the original on 2017-05-10.
- ^ Porter, R.; Brand, M. (1 September 1995). "Mitochondrial proton conductance and H+/O ratio are independent of electron transport rate in isolated hepatocytes". The Biochemical Journal (Free full text). 310 (Pt 2): 379–382. doi:10.1042/bj3100379. ISSN 0264-6021. PMC 1135905. PMID 7654171.
- ^ a b c Stryer, Lubert (1995). Biochemistry (fourth ed.). New York – Basingstoke: W. H. Freeman and Company. ISBN 978-0716720096.
- ^ "Molecular architecture of the rotary motor in ATP synthase". Science. 286 (5445): 1700–5. 1999. doi:10.1126/science.286.5445.1700. PMID 10576729.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ "Bioenergetic Cost of Making an Adenosine Triphosphate Molecule in Animal Mitochondria". Proc. Natl. Acad. Sci. USA. 107 (39): 16823–16827. 2010. doi:10.1073/pnas.1011099107. PMC 2947889. PMID 20847295.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ P.Hinkle (2005). "P/O ratios of mitochondrial oxidative phosphorylation". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1706 (1–2): 1–11. doi:10.1016/j.bbabio.2004.09.004. PMID 15620362.
- ^ Lollar, Garnet S.; Warr, Oliver; Telling, Jon; Osburn, Magdalena R.; Sherwood Lollar, Barbara (2019). "'Follow the Water': Hydrogeochemical Constraints on Microbial Investigations 2.4 km Below Surface at the Kidd Creek Deep Fluid and Deep Life Observatory". Geomicrobiology Journal. 36: 859–872. doi:10.1080/01490451.2019.1641770.
- ^ World’s Oldest Groundwater Supports Life Through Water-Rock Chemistry Archived 2019-09-10 at the Wayback Machine, July 29, 2019, deepcarbon.net.
- ^ Strange life-forms found deep in a mine point to vast 'underground Galapagos' Archived 2019-09-09 at the Wayback Machine, By Corey S. Powell, Sept. 7, 2019, nbcnews.com.

