Chloro(triphenylphosphine)gold(I)

| |

| |
| Identifiers | |
|---|---|
| ECHA InfoCard | 100.034.636 |
CompTox Dashboard (EPA)
|
|
| Properties | |
| C18H15AuClP | |
| Molar mass | 494.71 g·mol−1 |
| Appearance | Colorless solid |
| Melting point | 236–237 °C (457–459 °F; 509–510 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chloro(triphenylphosphine)gold(I) is the coordination complex with the formula (Ph3P)AuCl. This colorless solid is the complex of triphenylphosphine and gold(I) chloride.
Preparation and structure
The complex is prepared by reducing chloroauric acid with triphenylphosphine in 95% ethanol:[1]
- HAuCl4 + H2O + 2 PPh3 → (Ph3P)AuCl + Ph3PO + 3 HCl
The complex adopts a linear coordination geometry, which is typical of most gold(I) compounds. [2] It crystallizes in the orthorhombic space group P212121 with a = 12.300(4) Å, b = 13.084(4) Å, c = 10.170(3) Å with Z = 4 formula units per unit cell. [3]
Reactivity
It is a common reagent in gold chemistry. Many related gold complexes (often incorrectly called salts) can be prepared including the trifluoromethanesulfonate and the nitrate.[4]
Triphenylphosphine gold triflate (Ph3PAuOTf) catalyzes certain rearrangement reactions in organic synthesis.
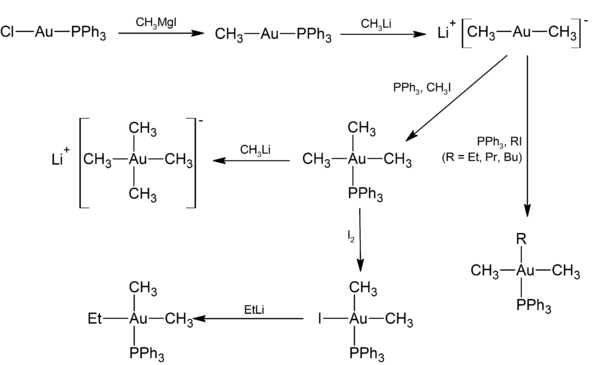
References
- ^ Pierre Braunstein, Hans Lehner, Dominique Matt "A Platinum-Gold Cluster: Chloro-1κCl-Bis(Triethylphosphine-1κP)Bis(Triphenyl- Phosphine)-2κP, 3κP-Triangulo- Digold-Platinum(1 +) Trifluoromethanesulfonate" Inorganic Syntheses, 1990, Volume 27, Pages 218–221. doi:10.1002/9780470132586.ch42
- ^ Baenziger, N. C.; Bennett, W. E.; Soborofe, D. M. (1976). "Chloro(triphenylphosphine)gold(I)". Acta Crystallographica Section B. 32 (3): 962. doi:10.1107/S0567740876004330.
- ^ Borissova, Alexandra O.; Korlyukov, Alexander A.; Antipin, Mikhail Yu.; Lyssenko, Konstantin A. (2008). "Estimation of Dissociation Energy in Donor−Acceptor Complex AuCl·PPh3via Topological Analysis of the Experimental Electron Density Distribution Function". The Journal of Physical Chemistry A. 112 (46): 11519–22. doi:10.1021/jp807258d. PMID 18959385.
- ^ A. M. Mueting, B. D. Alexander, P. D. Boyle, A. L. Casalnuovo, L. N. Ito, B. J. Johnson, L. H. Pignolet "Mixed-Metal-Gold Phosphine Cluster Compounds" Inorganic Syntheses, 1992, Volume 29, Pages 279–298, 2007. doi:10.1002/9780470132609.ch63
