Curium(III) hydroxide
| |
| Names | |
|---|---|
| IUPAC name
Curium hydroxide
| |
| Systematic IUPAC name
Curium(3+) oxidanide | |
| Identifiers | |
3D model (JSmol)
|
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CmH3O3 | |
| Molar mass | 298 g·mol−1 |
| Appearance | colorless or pale yellow solid |
| insoluble | |
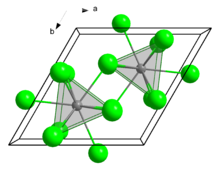
| Structure | |
| hexagonal, UCl3 structure[1] | |
| P63/m, No. 176[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Curium hydroxide Cm(OH)3 is a radioactive compound first discovered in measurable quantities in 1947. It is composed of a single curium atom and three hydroxy groups. It was the first curium compound ever isolated.[3][4]
Curium hydroxide is an anhydrous colorless[2] or light-yellow[5] amorphous gelatinous solid that is insoluble in water.[1]
Due to self-irradiation, the crystal structure of 244Cm(OH)3 decomposes[clarification needed] within one day (244Cm has a half-life of 18.11 years); for 241Am(OH)3 the same process takes 4 to 6 months (241Am has a half-life of 432.2 years).[2]
See also[edit]
References[edit]
- ^ a b Macintyre, Jane E. (1992). Dictionary of Inorganic Compounds. CRC Press. p. 3046. ISBN 978-0-412-30120-9.
- ^ a b c d e Krivovichev, Sergey; Burns, Peter; Tananaev, Ivan (2006). Structural Chemistry of Inorganic Actinide Compounds. Elsevier. p. 68. ISBN 978-0-08-046791-7.
- ^ Seaborg, Glenn T. (1963). Man-Made Transuranium Elements. Prentice-Hall.
- ^ "WebElements Periodic Table: Curium". webelements.com. Retrieved January 20, 2019.
- ^ Koch, Günter (1972). Transurane Teil C: Die Verbindungen. Gmelins Handbuch (in German). Springer-Verlag. p. 35. ISBN 978-3-662-11547-3.
