RDX
| |||
 RDX crystal
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,3,5-Trinitro-1,3,5-triazinane | |||
| Other names
1,3,5-Trinitroperhydro-1,3,5-triazine
RDX cyclonite, hexogen 1,3,5-Trinitro-1,3,5-triazacyclohexane 1,3,5-Trinitrohexahydro-s-triazine Cyclotrimethylenetrinitramine Hexahydro-1,3,5-trinitro-s-triazine Trimethylenetrinitramine hexolite[1] | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.092 | ||
PubChem CID
|
|||
| UNII | |||
| UN number | 0072, 0391, 0483 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H6N6O6 | |||
| Molar mass | 222.117 g·mol−1 | ||
| Appearance | Colorless or yellowish crystals | ||
| Density | 1.806 g/cm3[2] | ||
| Melting point | 205.5 °C (401.9 °F; 478.6 K) | ||
| Boiling point | 234 °C (453 °F; 507 K) | ||
| insoluble [3] | |||
| Explosive data | |||
| Shock sensitivity | Low | ||
| Friction sensitivity | Low | ||
| Detonation velocity | 8750 m/s | ||
| RE factor | 1.60 | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Explosive, detonates on contact with mercury fulminate,[3] highly toxic | ||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H201, H301, H370, H373 | |||
| P210, P250, P280, P370, P372, P373, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Explosive [3] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
100 mg/kg | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none [3] | ||
REL (Recommended)
|
TWA 1.5 mg/m3 ST 3 mg/m3 [skin][3] | ||
IDLH (Immediate danger)
|
N.D.[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
RDX (abbreviation of "Research Department eXplosive" or Royal Demolition eXplosive) or hexogen,[4] among other names, is an organic compound with the formula (CH2N2O2)3. It is white, odorless, and tasteless, widely used as an explosive.[5] Chemically, it is classified as a nitroamine alongside HMX, which is a more energetic explosive than TNT. It was used widely in World War II and remains common in military applications.
RDX is often used in mixtures with other explosives and plasticizers or phlegmatizers (desensitizers); it is the explosive agent in C-4 plastic explosive and a key ingredient in Semtex. It is stable in storage and is considered one of the most energetic and brisant of the military high explosives,[2] with a relative effectiveness factor of 1.60.
Name
[edit]RDX is also less commonly known as cyclonite, hexogen (particularly in Russian, French and German-influenced languages), T4, and, chemically, as cyclotrimethylene trinitramine.[6] In the 1930s, the Royal Arsenal, Woolwich, started investigating cyclonite to use against German U-boats that were being built with thicker hulls. The goal was to develop an explosive more energetic than TNT. For security reasons, Britain termed cyclonite "Research Department Explosive" (R.D.X.).[7] The term RDX appeared in the United States in 1946.[8] The first public reference in the United Kingdom to the name RDX, or R.D.X., to use the official title, appeared in 1948; its authors were the managing chemist, ROF Bridgwater, the chemical research and development department, Woolwich, and the director of Royal Ordnance Factories, Explosives.[9]
Usage
[edit]This section needs additional citations for verification. (January 2021) |

RDX was widely used during World War II, often in explosive mixtures with TNT such as Torpex, Composition B, Cyclotols, and H6. RDX was used in one of the first plastic explosives. The bouncing bomb depth charges used in the "Dambusters Raid" each contained 6,600 pounds (3,000 kg) of Torpex;[10] The Tallboy and Grand Slam bombs designed by Barnes Wallis also used Torpex.
RDX is believed to have been used in many bomb plots, including terrorist plots.
RDX is the base for a number of common military explosives:
- Composition A: Granular explosive consisting of RDX and plasticizing wax, such as composition A-3 (91% RDX coated with 9% wax)[11] and composition A-5 (98.5 to 99.1% RDX coated with 0.95 to 1.54% stearic acid).[12]
- Composition B: Castable mixtures of 59.5% RDX and 39.4% TNT with 1% wax as desensitizer.[13]
- Composition C: The original composition C was used in World War II, but there have been subsequent variations including C-2, C-3, and C-4. C-4 consists of RDX (91%); a plasticizer, dioctyl sebacate (5.3%); and a binder, which is usually polyisobutylene (2.1%); and oil (1.6%).[14]
- Composition CH-6: 97.5% RDX, 1.5% calcium stearate, 0.5% polyisobutylene, and 0.5% graphite[15]
- DBX (Depth Bomb Explosive): Castable mixture consisting of 21% RDX, 21% ammonium nitrate, 40% TNT, and 18% powdered aluminium, developed during World War II, it was to be used in underwater munitions as a substitute for Torpex employing only half the amount of then-scarce RDX,[2][16] as the supply of RDX became more adequate, however, the mixture was shelved
- Cyclotol: Castable mixture of RDX (50–80%) with TNT (20–50%) designated by the amount of RDX/TNT, such as Cyclotol 70/30
- HBX: Castable mixtures of RDX, TNT, powdered aluminium, and D-2 wax with calcium chloride
- H-6: Castable mixture of RDX, TNT, powdered aluminum, and paraffin wax (used as a phlegmatizing agent)
- PBX: RDX is also used as a major component of many polymer-bonded explosives (PBX); RDX-based PBXs typically consist of RDX and at least thirteen different polymer/co-polymer binders.[17] Examples of RDX-based PBX formulations include, but are not limited to: PBX-9007, PBX-9010, PBX-9205, PBX-9407, PBX-9604, PBXN-106, PBXN-3, PBXN-6, PBXN-10, PBXN-201, PBX-0280, PBX Type I, PBXC-116, PBXAF-108, etc.[citation needed]
- Semtex (trade name): Plastic demolition explosive containing RDX and PETN as major energetic components [18]
- Torpex: 42% RDX, 40% TNT, and 18% powdered aluminium; the mixture was designed during World War II and used mainly in underwater ordnance [19]
Outside military applications, RDX is also used in controlled demolition to raze structures.[20] The demolition of the Jamestown Bridge in the U.S. state of Rhode Island was one instance where RDX shaped charges were used to remove the span.[21]
Synthesis
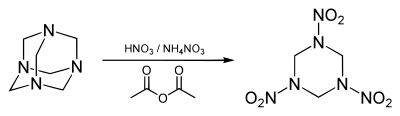
[edit]RDX is classified by chemists as a hexahydro-1,3,5-triazine derivative. In laboratory settings (industrial routes are described below separately) it is obtained by treating hexamine with white fuming nitric acid.[22]
This nitrolysis reaction also produces methylene dinitrate, ammonium nitrate, and water as by-products. The overall reaction is:[22]
- C6H12N4 + 10 HNO3 → C3H6N6O6 + 3 CH2(ONO2)2 + NH4NO3 + 3 H2O
The conventional cheap nitration agent, called "mixed acid", cannot be used for RDX synthesis because concentrated sulfuric acid conventionally used to stimulate the nitronium ion formation decomposes hexamine into formadehyde and ammonia.
Modern syntheses employ hexahydro triacyl triazine as it avoids formation of HMX.[23]
History
[edit]RDX was used by both sides in World War II. The US produced about 15,000 long tons (15,000 t) per month during WWII and Germany about 7,100 tonnes (7,000 long tons) per month.[24] RDX had the major advantages of possessing greater explosive force than TNT and required no additional raw materials for its manufacture. Thus, it was also extensively used in World War I[24]
Germany
[edit]RDX was reported in 1898 by Georg Friedrich Henning, who obtained a German patent (patent No. 104280) for its manufacture by nitrolysis of hexamine (hexamethylenetetramine) with concentrated nitric acid.[25][26] In this patent, the medical properties of RDX were mentioned; however, three further German patents obtained by Henning in 1916 proposed its use in smokeless propellants.[25] The German military started investigating its use in 1920, referring to it as hexogen.[27] Research and development findings were not published further until Edmund von Herz,[28] described as an Austrian and later a German citizen, obtained a British patent in 1921[29] and a United States patent in 1922.[30] Both patent claims were initiated in Austria; and described the manufacture of RDX by nitrating hexamethylenetetramine.[29][30] The British patent claims included the manufacture of RDX by nitration, its use with or without other explosives, its use as a bursting charge and as an initiator.[29] The US patent claim was for the use of a hollow explosive device containing RDX and a detonator cap containing RDX.[30] In the 1930s, Germany developed improved production methods.[27]
During World War II, Germany used the code names W Salt, SH Salt, K-method, the E-method, and the KA-method. These names represented the identities of the developers of the various chemical routes to RDX. The W-method was developed by Wolfram in 1934 and gave RDX the code name "W-Salz". It used sulfamic acid, formaldehyde, and nitric acid.[31] SH-Salz (SH salt) was from Schnurr, who developed a batch-process in 1937–38 based on nitrolysis of hexamine.[32] The K-method, from Knöffler, involved addition of ammonium nitrate to the hexamine/nitric acid process.[33] The E-method, developed by Ebele, proved to be identical to the Ross and Schiessler process described below.[34] The KA-method, also developed by Knöffler, turned out to be identical to the Bachmann process described below.[35]
The explosive shells fired by the MK 108 cannon and the warhead of the R4M rocket, both used in Luftwaffe fighter aircraft as offensive armament, both used hexogen as their explosive base.[36]
UK
[edit]In the United Kingdom (UK), RDX was manufactured from 1933 by the research department in a pilot plant at the Royal Arsenal in Woolwich, London, a larger pilot plant being built at the RGPF Waltham Abbey just outside London in 1939.[37][38] In 1939 a twin-unit industrial-scale plant was designed to be installed at a new 700-acre (280 ha) site, ROF Bridgwater, away from London and production of RDX started at Bridgwater on one unit in August 1941.[37][39] The ROF Bridgwater plant brought in ammonia and methanol as raw materials: the methanol was converted to formaldehyde and some of the ammonia converted to nitric acid, which was concentrated for RDX production.[9] The rest of the ammonia was reacted with formaldehyde to produce hexamine. The hexamine plant was supplied by Imperial Chemical Industries. It incorporated some features based on data obtained from the United States (US).[9] RDX was produced by continually adding hexamine and concentrated nitric acid to a cooled mixture of hexamine and nitric acid in the nitrator.[9] The RDX was purified and processed for its intended use; recovery and reuse of some methanol and nitric acid also was carried out.[9] The hexamine-nitration and RDX purification plants were duplicated (i.e. twin-unit) to provide some insurance against loss of production due to fire, explosion, or air attack.[37]
The United Kingdom and British Empire were fighting without allies against Nazi Germany until the middle of 1941 and had to be self-sufficient. At that time (1941), the UK had the capacity to produce 70 long tons (71 t) (160,000 lb) of RDX per week; both Canada, an allied country and self-governing dominion within the British Empire, and the US were looked upon to supply ammunition and explosives, including RDX.[40] By 1942 the Royal Air Force's annual requirement was forecast to be 52,000 long tons (53,000 t) of RDX, much of which came from North America (Canada and the US).[39]
Canada
[edit]A different method of production to the Woolwich process was found and used in Canada, possibly at the McGill University department of chemistry. This was based on reacting paraformaldehyde and ammonium nitrate in acetic anhydride.[41] A UK patent application was made by Robert Walter Schiessler (Pennsylvania State University) and James Hamilton Ross (McGill, Canada) in May 1942; the UK patent was issued in December 1947.[42] Gilman states that the same method of production had been independently discovered by Ebele in Germany prior to Schiessler and Ross, but that this was not known by the Allies.[25][41] Urbański provides details of five methods of production, and he refers to this method as the (German) E-method.[34]
UK, US, and Canadian production and development
[edit]At the beginning of the 1940s, the major US explosive manufacturers, E. I. du Pont de Nemours & Company and Hercules, had several decades of experience of manufacturing trinitrotoluene (TNT) and had no wish to experiment with new explosives. US Army Ordnance held the same viewpoint and wanted to continue using TNT.[43] RDX had been tested by Picatinny Arsenal in 1929, and it was regarded as too expensive and too sensitive.[40] The Navy proposed to continue using ammonium picrate.[43] In contrast, the National Defense Research Committee (NDRC), who had visited The Royal Arsenal, Woolwich, thought new explosives were necessary.[43] James B. Conant, chairman of Division B, wished to involve academic research into this area. Conant therefore set up an experimental explosives research laboratory at the Bureau of Mines, Bruceton, Pennsylvania, using Office of Scientific Research and Development (OSRD) funding.[40]
Woolwich method
[edit]In 1941, the UK's Tizard Mission visited the US Army and Navy departments and part of the information handed over included details of the "Woolwich" method of manufacture of RDX and its stabilisation by mixing it with beeswax.[40] The UK was asking that the US and Canada, combined, supply 220 short tons (200 t) (440,000 lb) of RDX per day.[40] A decision was taken by William H. P. Blandy, chief of the Bureau of Ordnance, to adopt RDX for use in mines and torpedoes.[40] Given the immediate need for RDX, the US Army Ordnance, at Blandy's request, built a plant that copied the equipment and process used at Woolwich. The result was the Wabash River Ordnance Works run by E. I. du Pont de Nemours & Company.[44] At that time, this works had the largest nitric acid plant in the world.[40] The Woolwich process was expensive: it needed 11 pounds (5.0 kg) of strong nitric acid for every pound of RDX.[45]
By early 1941, the NDRC was researching new processes.[45] The Woolwich or direct nitration process has at least two serious disadvantages: (1) it used large amounts of nitric acid and (2) at least one-half of the formaldehyde is lost. One mole of hexamethylenetetramine could produce at most one mole of RDX.[46] At least three laboratories with no previous explosive experience were instructed to develop better production methods for RDX; they were based at Cornell, Michigan, and Pennsylvania State universities.[40][a] Werner Emmanuel Bachmann, from Michigan, successfully developed the "combination process" by combining the Ross and Schiessler process used in Canada (aka the German E-method) with direct nitration.[35][40] The combination process required large quantities of acetic anhydride instead of nitric acid in the old British "Woolwich process". Ideally, the combination process could produce two moles of RDX from each mole of hexamethylenetetramine.[46]
The vast production of RDX could not continue to rely on the use of natural beeswax to desensitize the RDX. A substitute stabilizer based on petroleum was developed at the Bruceton Explosives Research Laboratory.[40]
Bachmann process
[edit]The National Defence Research Committee (NDRC) instructed three companies to develop pilot plants. They were the Western Cartridge Company, E. I. du Pont de Nemours & Company, and Tennessee Eastman Company, part of Eastman Kodak.[40] At the Eastman Chemical Company (TEC), a leading manufacturer of acetic anhydride, Werner Emmanuel Bachmann developed a continuous-flow process for RDX utilizing an ammonium nitrate/nitric acid mixture as a nitrating agent in a medium of acetic acid and acetic anhydride. RDX was crucial to the war effort and the current batch-production process was too slow. In February 1942, TEC began producing small amounts of RDX at its Wexler Bend pilot plant, which led to the US government authorizing TEC to design and build Holston Ordnance Works (H.O.W.) in June 1942. By April 1943, RDX was being manufactured there.[47] At the end of 1944, the Holston plant and the Wabash River Ordnance Works, which used the Woolwich process, were producing 25,000 short tons (23,000 t) (50 million pounds) of Composition B per month.[48]
The Bachmann process yields both RDX and HMX, with the major product determined by the specific reaction conditions.[49]
Military compositions
[edit]The United Kingdom's intention in World War II was to use "desensitised" RDX. In the original Woolwich process, RDX was phlegmatized with beeswax, but later paraffin wax was used, based on the work carried out at Bruceton. In the event the UK was unable to obtain sufficient RDX to meet its needs, some of the shortfall was met by substituting amatol, a mixture of ammonium nitrate and TNT.[39]
Karl Dönitz was reputed to have claimed that "an aircraft can no more kill a U-boat than a crow can kill a mole".[50] Nonetheless, by May 1942 Wellington bombers began to deploy depth charges containing Torpex, a mixture of RDX, TNT, and aluminium, which had up to 50 percent more destructive power than TNT-filled depth charges.[50] Considerable quantities of the RDX–TNT mixture were produced at the Holston Ordnance Works, with Tennessee Eastman developing an automated mixing and cooling process based around the use of stainless steel conveyor belts.[51]
Terrorism
[edit]A Semtex bomb was used in the Pan Am Flight 103 (known also as the Lockerbie) bombing in 1988.[52] A belt laden with 700 g (1.5 lb) of RDX explosives tucked under the dress of the assassin was used in the assassination of former Indian prime minister Rajiv Gandhi in 1991.[53] The 1993 Bombay bombings used RDX placed into several vehicles as bombs. RDX was the main component used for the 2006 Mumbai train bombings and the Jaipur bombings in 2008.[54][55] It also is believed to be the explosive used in the 2010 Moscow Metro bombings.[56]
Traces of RDX were found on pieces of wreckage from 1999 Russian apartment bombings[57][58] and 2004 Russian aircraft bombings.[59] FSB reports on the bombs used in the 1999 apartment bombings indicated that while RDX was not a part of the main charge, each bomb contained plastic explosive used as a booster charge.[60][61]
Ahmed Ressam, the al-Qaeda Millennium Bomber, used a small quantity of RDX as one of the components in the bomb that he prepared to detonate in Los Angeles International Airport on New Year's Eve 1999–2000; the bomb could have produced a blast forty times greater than that of a devastating car bomb.[62][63]
In July 2012, the Kenyan government arrested two Iranian nationals and charged them with illegal possession of 15 kilograms (33 pounds) of RDX. According to the Kenyan Police, the Iranians planned to use the RDX for "attacks on Israeli, US, UK and Saudi Arabian targets".[64]
RDX was used in the assassination of Lebanese Prime Minister Rafic Hariri on February 14, 2005.[65]
In the 2019 Pulwama attack in India, 250 kg of high-grade RDX was used by Jaish-e-Mohammed. The attack resulted in the deaths of 44 Central Reserve Police Force (CRPF) personnel as well as the attacker.[66]
Two letter bombs sent to journalists in Ecuador were disguised as USB flash drives which contained RDX that would detonate when plugged in.[67]
Stability
[edit]RDX has a high nitrogen content and a high oxygen to carbon ratio, (O:C ratio), both of which indicate its explosive potential for formation of N2 and CO2.
RDX undergoes a deflagration to detonation transition (DDT) in confinement and certain circumstances.[68]
The velocity of detonation of RDX at a density of 1.80 g/cm3 is 8750 m/s.[69]
It starts to decompose at approximately 170 °C and melts at 204 °C. At room temperature, it is very stable. It burns rather than explodes. It detonates only with a detonator, being unaffected even by small arms fire. This property makes it a useful military explosive. It is less sensitive than pentaerythritol tetranitrate (PETN). Under normal conditions, RDX has a Figure of Insensitivity of exactly 80 (RDX defines the reference point).[70]
RDX sublimes in vacuum, which restricts or prevents its use in some applications.[71]
RDX, when exploded in air, has about 1.5 times the explosive energy of TNT per unit weight and about 2.0 times per unit volume.[51][72]
RDX is insoluble in water, with solubility 0.05975 g/L at temperature of 25 °C.[73]
Toxicity
[edit]The substance's toxicity has been studied for many years.[74] RDX has caused convulsions (seizures) in military field personnel ingesting it, and in munition workers inhaling its dust during manufacture. At least one fatality was attributed to RDX toxicity in a European munitions manufacturing plant.[75]
During the Vietnam War, at least 40 American soldiers were hospitalized with composition C-4 (which is 91% RDX) intoxication from December 1968 to December 1969. C-4 was frequently used by soldiers as a fuel to heat food, and the food was generally mixed by the same knife that was used to cut C-4 into small pieces prior to burning. Soldiers were exposed to C-4 either due to inhaling the fumes, or due to ingestion, made possible by many small particles adhering to the knife having been deposited into the cooked food. The symptom complex involved nausea, vomiting, generalized seizures, and prolonged postictal confusion and amnesia; which indicated toxic encephalopathy.[76]
Oral toxicity of RDX depends on its physical form; in rats, the LD50 was found to be 100 mg/kg for finely powdered RDX, and 300 mg/kg for coarse, granular RDX.[75] A case has been reported of a human child hospitalized in status epilepticus following the ingestion of 84.82 mg/kg dose of RDX (or 1.23 g for the patient's body weight of 14.5 kg) in the "plastic explosive" form.[77]
The substance has low to moderate toxicity with a possible human carcinogen classification.[78][79][80] Further research is ongoing, however, and this classification may be revised by the United States Environmental Protection Agency (EPA).[81][82] Remediating RDX-contaminated water supplies has proven to be successful.[83] It is known to be a kidney toxin in humans and highly toxic to earthworms and plants, thus army testing ranges where RDX was used heavily may need to undergo environmental remediation.[84] Concerns have been raised by research published in late 2017 indicating that the issue has not been addressed correctly by U.S. officials.[85]
Civilian use
[edit]RDX has been used as a rodenticide because of its toxicity.[86]
Biodegradation
[edit]RDX is degraded by the organisms in sewage sludge as well as the fungus Phanaerocheate chrysosporium.[87] Both wild and transgenic plants can phytoremediate explosives from soil and water.[88][89]
Alternatives
[edit]FOX-7 is considered to be approximately a 1-to-1 replacement for RDX in almost all applications.[90][91]
See also
[edit]Notes
[edit]- ^ These were not the only laboratories to work on RDX, Gilman's 1953 account of the Ross–Schiessler method was based on unpublished work from laboratories at the Universities of Michigan, Pennsylvania, Cornell, Harvard, Vanderbilt, McGill (Canada), Bristol (UK), Sheffield (UK), Pennsylvania State College, and the UK's research department.
References
[edit]- ^ "Hexolite, CAS Number: 82030-42-0". Archived from the original on October 27, 2021. Retrieved April 8, 2021.
- ^ a b c Department of the Army Technical Manual TM 9-1300-214: Military Explosives. Headquarters, Department of the Army (United States). 1989.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0169". National Institute for Occupational Safety and Health (NIOSH).
- ^ "RDX explosive". britannica.com. Retrieved September 27, 2021.
- ^ Field, Simon Quellen (July 1, 2017). Boom!: The Chemistry and History of Explosives. Chicago Review Press. pp. 89–94. ISBN 978-1613738054.
- ^ Davis, Tenney L. (1943), The Chemistry of Powder and Explosives, vol. II, New York: John Wiley & Sons Inc., p. 396
- ^ MacDonald and Mack Partnership (1984, p. 18)
- ^ Baxter III 1968, pp. 27, 42, 255–259
- ^ a b c d e Simmons, W.H.; Forster, A.; Bowden, R. C. (August 1948), "The Manufacture of R.D.X. in Great Britain: Part II – Raw Materials and Ancillary Processes", The Industrial Chemist, 24: 530–545; Simmons, W.H.; Forster, A.; Bowden, R. C. (September 1948), "The Manufacture of R.D.X. in Great Britain: Part III – Production of the Explosive", The Industrial Chemist, 24: 593–601
- ^ Sweetman, John (2002) The Dambusters Raid. London: Cassell Military Paperbacks. p. 144.
- ^ Pichtel, John (2012). "Distribution and Fate of Military Explosives and Propellants in Soil: A Review". Applied and Environmental Soil Science. 2012 (Article ID 617236). Hindawi: 3. doi:10.1155/2012/617236.
- ^ Ritchie, Robert (March 1984). Tech. Report ARLCD-TR-84004, Improving Quality and Performance of Leads Loaded with Composition A-5 (PDF). Dover, NJ: Large Caliber Weapons Systems Laboratory, US Army ARDC. p. 7. Archived (PDF) from the original on February 15, 2017. Retrieved November 9, 2018.
- ^ DOD (March 13, 1974). "MIL-C-401E, Composition B, Rev. C". EverySpec. p. 3. Retrieved November 9, 2018.
- ^ Reardon, Michelle R.; Bender, Edward C. (2005). "Differentiation of Composition C4 Based on the Analysis of the Process Oil". Journal of Forensic Sciences. 50 (3). Ammendale, MD: Bureau of Alcohol, Tobacco, Firearms, and Explosives, Forensic Science Laboratory: 1–7. doi:10.1520/JFS2004307. ISSN 0022-1198.
- ^ Hampton, L. D. (June 15, 1960), The Development of RDX Composition CH-6 (PDF), White Oak, MD: U. S. Naval Ordnance Laboratory, NavOrd Report 680, archived from the original (PDF) on January 19, 2012
- ^ US Explosive Ordnance; Ordnance Pamphlet OP 1664. Vol. 1. Washington, D.C.: Navy Department, Bureau of Ordnance. May 28, 1947. pp. 3–4. OP 1664 states 21% "aluminum nitrate", but the immediately following text refers to ammonium nitrate.
- ^ Akhavan, Jacqueline (2011). The Chemistry of Explosives (3rd ed.). Cambridge: Royal Society of Chemistry. p. 14. ISBN 978-1-84973-330-4. Retrieved November 15, 2018.
- ^ "Semtex". PubChem Open Chemistry Database. Nat. Center for Biotechnology Information, US Library of Medicine. Retrieved November 15, 2018.
- ^ Pekelney, Richard. "U.S. Explosive Ordnance (1947)". San Francisco Maritime National Park. Retrieved April 24, 2017.
- ^ Beebe, S. M.; Pherson, R. H. (2011). Cases in Intelligence Analysis: Structured Analytic Techniques in Action. SAGE Publications. p. 182. ISBN 978-1-4833-0517-2. Retrieved April 24, 2017.
- ^ "Demolition of the Lake Champlain Bridge" (PDF). New York State Department of Transportation. December 12, 2009. p. 13. Retrieved May 1, 2018.
- ^ a b Luo, K.-M.; Lin, S.-H.; Chang, J.-G.; Huang, T.-H. (2002), "Evaluations of kinetic parameters and critical runaway conditions in the reaction system of hexamine-nitric acid to produce RDX in a non-isothermal batch reactor", Journal of Loss Prevention in the Process Industries, 15 (2): 119–127, Bibcode:2002JLPPI..15..119L, doi:10.1016/S0950-4230(01)00027-4.
- ^ Gilbert, E. E.; Leccacorvi, J. R.; Warman, M. (June 1, 1976). "23. The Preparation of RDX from 1,3,5-Triacylhexahydro-s-triazines". In Albright, Lyle F.; Hanson, Carl (eds.). Industrial and Laboratory Nitrations. ACS Symposium Series. Vol. 22. pp. 327–340. doi:10.1021/bk-1976-0022.ch023. ISBN 978-0-8412-0306-8.
- ^ a b Urbański (1967, p. 78)
- ^ a b c Urbański (1967, pp. 77–119)
- ^ DE 104280, Henning, Georg Friedrich, issued June 14, 1899
- ^ a b Hexogen Archived July 26, 2011, at the Wayback Machine. economypoint.org, citing Gartz, Jochen (2007), Vom griechischen Feuer zum Dynamit: eine Kulturgeschichte der Explosivstoffe [From Greek fire to dynamite: A cultural history of explosives] (in German), Hamburg: E. S. Mittler & Sohn, ISBN 978-3-8132-0867-2
- ^ Urbański (1967, p. 125) credits "G. C. V. Herz" for the patent, but the patentee is Edmund von Herz.
- ^ a b c GB 145791, von Herz, Edmund, "Improvements relating to Explosives", issued March 17, 1921
- ^ a b c US 1402693, von Herz, Edmund, "Explosive", issued January 3, 1922
- ^ Urbański (1967, pp. 107–109)
- ^ Urbański (1967, pp. 104–105)
- ^ Urbański (1967, pp. 105–107)
- ^ a b Urbański (1967, pp. 109–110)
- ^ a b Urbański (1967, pp. 111–113)
- ^ Press, Merriam (2017). World War 2 In Review No. 23: Boeing B-17 Flying Fortress. Lulu Press. p. 17. ISBN 9781387322572.[permanent dead link]
- ^ a b c Cocroft, Wayne D. (2000), Dangerous Energy: The archaeology of gunpowder and military explosives manufacture, Swindon: English Heritage, pp. 210–211, ISBN 1-85074-718-0
- ^ Akhavan, Jacqueline (2004), The Chemistry of Explosives, Cambridge, UK: Royal Society of Chemistry, ISBN 0-85404-640-2
- ^ a b c Hornby, William (1958), Factories and Plant, History of the Second World War: United Kingdom Civil Series, London: His Majesty's Stationery Office; Longmans, Green and Co., pp. 112–114
- ^ a b c d e f g h i j k Baxter III (1968, pp. 253–239)
- ^ a b Gilman, Henry (1953), "The Chemistry of Explosives", Organic Chemistry an Advanced Treatise, vol. III, Wiley; Chapman & Hall, p. 985
- ^ GB 595354, Schiessler, Robert Walter & Ross, James Hamilton, "Method of Preparing 1.3.5. Trinitro Hexahydro S-Triazine", issued December 3, 1947
- ^ a b c Baxter III (1968, pp. 253–254)
- ^ MacDonald and Mack Partnership (1984, p. 19)
- ^ a b MacDonald and Mack Partnership (1984, p. 13) These pages need to be checked. Page 13 may actually be page 18.
- ^ a b Elderfield (1960, p. 6)
- ^ Bachmann, W. E.; Sheehan, John C. (1949), "A New Method of Preparing the High Explosive RDX", Journal of the American Chemical Society, 71 (5): 1842–1845, doi:10.1021/ja01173a092
- ^ MacDonald and Mack Partnership (1984, p. 32)
- ^ Yinon, Jehuda (June 30, 1990). Toxicity and Metabolism of Explosives. CRC Press. p. 166. ISBN 978-1-4398-0529-9.
- ^ a b Baxter III (1968, p. 42)
- ^ a b Baxter III (1968, pp. 257 & 259)
- ^ Bolz, F. (Jr.); Dudonis, K.J.; Schulz, D.P. (2012). The Counterterrorism Handbook: Tactics, Procedures, and Techniques (4th ed.). Boca Raton, FL: CRC Press. pp. 340–341. ISBN 978-1439846704.
- ^ Ramesh Vinayak (February 1, 1999). "The Nation: Terrorism: The RDX Files". India-today.com. Archived from the original on October 9, 2010. Retrieved March 7, 2010.
- ^ Singh, Anil (October 2, 2006). "Mumbai". The Times of India. Archived from the original on October 18, 2012.
- ^ "Jaipur blasts: RDX used, HuJI suspected". The Times of India. May 14, 2008. Archived from the original on August 11, 2011. Retrieved May 13, 2011.
- ^ "Moscow Metro bombing masterminds 'will be destroyed'". BBC News. March 29, 2010. Retrieved April 2, 2010.
- ^ "Debate on Cause of Moscow Blast Heats Up". New York Times. September 10, 1999. Retrieved November 14, 2011.
- ^ "U.S. Senator Ben Cardin Releases Report Detailing Two Decades of Putin's Attacks on Democracy, Calling for Policy Changes to Counter Kremlin Threat Ahead of 2018, 2020 Elections | U.S. Senator Ben Cardin of Maryland". cardin.senate.gov. Archived from the original on February 14, 2018. Retrieved January 17, 2018., pages 165-171.
- ^ "Explosive Suggests Terrorists Downed Plane, Russia Says". New York Times. August 28, 2004. Retrieved November 14, 2011.
- ^ Миронов, Иван (September 9, 2002). "Кто и как взрывал Москву". Rossiyskaya Gazeta (in Russian). FSB.
- ^ "О результатах расследования ряда актов терроризма" (in Russian). Federal Security Service. March 14, 2002.
- ^ U.S. Court of Appeals for the Ninth Circuit (February 2, 2010). "U.S. v. Ressam" (PDF). Archived from the original (PDF) on October 4, 2012. Retrieved February 27, 2010.
- ^ "Complaint; U.S. v. Ressam" (PDF). NEFA Foundation. December 1999. Archived from the original (PDF) on March 1, 2012. Retrieved February 26, 2010.
- ^ "Iranian agents in Kenya planned attacks on Israel, US, UK, Saudi Arabian targets". Washington Post. July 2, 2012. Archived from the original on July 3, 2012. Retrieved July 2, 2012.
- ^ Ronen Bergman (February 10, 2015). "The Hezbollah Connection". New York Times. Archived from the original on January 1, 2022. Retrieved February 16, 2015.
- ^ "2019 Pulwama attack: RDX used". The Economic Times. February 15, 2019. Retrieved February 15, 2019.[permanent dead link]
- ^ "At least 5 news stations receive letter bombs in Ecuador, one explodes: "Clear message to silence journalists" - CBS News". www.cbsnews.com. March 21, 2023. Retrieved June 21, 2023.
- ^ Price, D.; Bernecker, R. (1977). "DDT Behavior of Waxed Mixtures of RDX, HMX, and Tetryl" (PDF). Naval Surface Weapons Center. Archived (PDF) from the original on December 2, 2016.
- ^ Klapötke, Thomas M. (May 29, 2012), Chemistry of High-Energy Materials, De Gruyter, doi:10.1515/9783110273595, ISBN 978-3-11-027359-5, retrieved October 1, 2023
- ^ Akhavan, Jacqueline (March 7, 2022). The Chemistry of Explosives. The Royal Society of Chemistry. doi:10.1039/9781839168802. ISBN 978-1-83916-446-0.
- ^ Ewing, Robert G.; Waltman, Melanie J.; Atkinson, David A.; Grate, Jay W.; Hotchkiss, Peter J. (January 1, 2013). "The vapor pressures of explosives". Trends in Analytical Chemistry. 42: 35–48. doi:10.1016/j.trac.2012.09.010.
- ^ Elderfield (1960, p. 8)
- ^ Yalkowsky, S. H.; He, Y.; Jain, P. (2010). Handbook of aqueous solubility data (PDF) (2nd ed.). Boca Raton, FL: CRC Press. p. 61. ISBN 9781439802458. Archived (PDF) from the original on October 16, 2012.
- ^ Annotated Reference Outline for the Toxicological Review of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). U.S. Environmental Protection Agency (November 23, 2010)
- ^ a b Schneider, N. R.; Bradley, S. L.; Andersen, M. E. (March 1977). "Toxicology of cyclotrimethylenetrinitramine: Distribution and metabolism in the rat and the miniature swine". Toxicology and Applied Pharmacology. 39 (3): 531–41. Bibcode:1977ToxAP..39..531S. doi:10.1016/0041-008X(77)90144-2. PMID 854927.
- ^ Ketel, W. B.; Hughes, J. R. (August 1, 1972). "Toxic encephalopathy with seizures secondary to ingestion of composition C-4: A clinical and electroencephalographic study". Neurology. 22 (8): 871–6. doi:10.1212/WNL.22.8.870. PMID 4673417. S2CID 38403787.
- ^ Woody, R.C.; Kearns, G.L.; Brewster, M.A.; Turley, C.P.; Sharp, G.B.; Lake, R.S. (1986). "The Neurotoxicity of Cyclotrimethylenetrinitramine (RDX) in a Child: A Clinical and Pharmacokinetic Evaluation". Journal of Toxicology: Clinical Toxicology. 24 (4): 305–319. doi:10.3109/15563658608992595. PMID 3746987.
- ^ Faust, Rosmarie A. (December 1994) Toxicity summary for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX). Oak Ridge National Laboratory
- ^ Smith, Jordan N.; Liu, Jun; Espino, Marina A.; Cobb, George P. (2007). "Age dependent acute oral toxicity of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) and two anaerobic N-nitroso metabolites in deer mice (Peromyscus maniculatus)". Chemosphere. 67 (11): 2267–73. Bibcode:2007Chmsp..67.2267S. doi:10.1016/j.chemosphere.2006.12.005. PMID 17275885.
- ^ Pan, Xiaoping; San Francisco, Michael J.; Lee, Crystal; Ochoa, Kelly M.; Xu, Xiaozheng; Liu, Jun; Zhang, Baohong; Cox, Stephen B.; Cobb, George P. (2007). "Examination of the mutagenicity of RDX and its N-nitroso metabolites using the Salmonella reverse mutation assay". Mutation Research/Genetic Toxicology and Environmental Mutagenesis. 629 (1): 64–9. Bibcode:2007MRGTE.629...64P. doi:10.1016/j.mrgentox.2007.01.006. PMID 17360228.
- ^ Muhly, Robert L. (December 2001) Update on the Reevaluation of the Carcinogenic Potential of RDX. U.S. Army Center for Health Promotion and Preventive Medicine (CHPPM) "white paper"
- ^ "Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) (CASRN 121-82-4)". epa.gov. Retrieved January 1, 2014.
- ^ Newell, Charles (August 2008). Treatment of RDX & HMX Plumes Using Mulch Biowalls. GSI Environmental, Inc.
- ^ Klapötke, Thomas M. (2012). Chemistry of high-energy materials (Second ed.). Berlin [u.a.]: De Gruyter. ISBN 978-311027358-8.
- ^ Lustgarten, Abrahm, Canadian Research Adds to Worry Over an Environmental Threat the Pentagon Has Downplayed for Decades, a study released late last year gives environmental experts a way to quantify how much RDX, a chemical used in military explosives, is spreading into surrounding communities, Propublica, January 9, 2018
- ^ Bodeau, Donald T. (2000). "Chapter 9. Military Energetic Materials: Explosives and propellants". Disease and the Environment. Government Printing Office. CiteSeerX 10.1.1.222.8866.
- ^ Hawari, J.; Beaudet, S.; Halasz, A.; Thiboutot, S.; Ampleman, G. (2000). "Microbial degradation of explosives: biotransformation versus mineralization". Applied Microbiology and Biotechnology. 54 (5): 605–618. doi:10.1007/s002530000445. PMID 11131384. S2CID 22362850.
- ^ Panz, K.; Miksch, K. (December 2012). "Phytoremediation of explosives (TNT, RDX, HMX) by wild-type and transgenic plants". Journal of Environmental Management. 113: 85–92. Bibcode:2012JEnvM.113...85P. doi:10.1016/j.jenvman.2012.08.016. PMID 22996005.
- ^ Low, Darryl; Tan, Kui; Anderson, Todd; Cobb, George P.; Liu, Jun; Jackson, W. Andrew (2008). "Treatment of RDX using down-flow constructed wetland mesocosms". Ecological Engineering. 32 (1): 72–80. Bibcode:2008EcEng..32...72L. doi:10.1016/j.ecoleng.2007.09.005.
- ^ "FOX-7 for Insensitive Boosters Merran A. Daniel, Phil J. Davies and Ian J. Lochert" (PDF). Archived (PDF) from the original on March 3, 2017.
- ^ "Fox-7 EURENCO Indeed, DADNE (FOX-7) has been shown to increase the burning rate in propellants more than RDX does, which is very interesting in high performance propellants.". Archived from the original on August 4, 2017. Retrieved August 3, 2017.
Bibliography
[edit]- Baxter III, James Phinney (1968) [1946], Scientists Against Time (MIT Paperback ed.), Cambridge, MA: MIT Press, ISBN 978-0-262-52012-6, OCLC 476611116
- Elderfield, Robert C. (1960), Werner Emanual Bachmann: 1901–1951 (PDF), Washington DC: National Academy of Sciences, archived (PDF) from the original on June 17, 2011
- MacDonald and Mack Partnership (August 1984), Final Properties Report: Newport Army Ammunition Plant (PDF), National Park Service, AD-A175 818, archived from the original (PDF) on April 29, 2011
- Urbański, Tadeusz (1967), Laverton, Silvia (ed.), Chemistry and Technology of Explosives, vol. III, translated by Jureck, Marian (First English ed.), Warszawa: PWN – Polish Scientific Publishers and Pergamon Press, OCLC 499857211. See also ISBN 978-0-08-010401-0.
- Urbański translation openlibrary.org, Macmillan, NY, 1964, ISBN 0-08-026206-6.
Further reading
[edit]- Agrawal, Jai Prakhash; Hodgson, Robert Dale (2007), Organic Chemistry of Explosives, Wiley, ISBN 978-0-470-02967-1
- US 2680671, Bachmann, Werner E., "Method of Treating Cyclonite Mixtures", published July 16, 1943, issued June 8, 1954
- US 2798870, Bachmann, Werner E., "Method for Preparing Explosives", published July 16, 1943, issued July 9, 1957
- Baxter, Colin F. (2018), The Secret History of RDX: The Super-Explosive That Helped Win World War II., Lexington: University of Kentucky Press, ISBN 978-0-8131-7528-7
- Cooper, Paul W. (1996), Explosives Engineering, New York: Wiley-VCH, ISBN 0-471-18636-8
- Hale, George C. (1925), "The Nitration of Hexamethylenetetramine", Journal of the American Chemical Society, 47 (11): 2754–2763, doi:10.1021/ja01688a017
- Meyer, Rudolf (1987), Explosives (3rd ed.), VCH Publishers, ISBN 0-89573-600-4
External links
[edit]- ADI Limited (Australia). Archive.org leads to Thales group products page that shows some military specifications.
- NLM Hazardous Substances Databank (US) – Cyclonite (RDX)
- CDC – NIOSH Pocket Guide to Chemical Hazards
- nla.gov.au, Army News (Darwin, NT), October 2, 1943, p 3. "Britain's New Explosive: Experts Killed in Terrific Blast", uses "Research Department formula X"
- nla.gov.au, The Courier-Mail (Brisbane, Qld.), September 27, 1943, p 1.