Cystathionine
Appearance

| |
| |
| Names | |
|---|---|
| IUPAC name
2-amino-4-(2-amino-2-carboxy-ethyl) thio-butanoic acid
| |
| Other names
L-Cystathionine; S-[(2R)-2-Amino-2-carboxyethyl]-L-homocysteine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.269 |
| KEGG | |
| MeSH | Cystathionine |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C7H14N2O4S | |
| Molar mass | 222.26 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
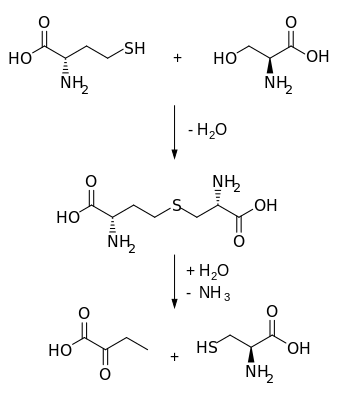
Cystathionine is an intermediate in the synthesis of cysteine.
An excess in the urine is called cystathioninuria.
Biosynthetically, cystathionineis generated from homocysteine and serine by cystathionine beta synthase (upper reaction in the diagram below). It is then cleaved into cysteine and α-ketobutyrate by cystathionine gamma-lyase (lower reaction).