Nabitan
This is an old revision of this page, as edited by Jytdog (talk | contribs) at 13:49, 12 August 2015 (remove content added by sock of blocked user Nuklear). The present address (URL) is a permanent link to this revision, which may differ significantly from the current revision.
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| Chemical and physical data | |
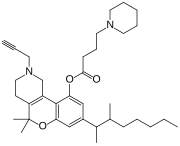
| Formula | C35H52N2O3 |
| Molar mass | 548.799 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Nabitan (Nabutam, Benzopyranoperidine, SP-106, Abbott 40656) is a synthetic cannabinoid analog of dronabinol (Marinol).[1] It exhibits antiemetic and analgesic effects, most likely by binding to and activating the CB1 and CB2 cannabinoid receptors, and reduced intraocular pressure in animal tests, making it potentially useful in the treatment of glaucoma.[2]
Nabitan has the advantage of being water-soluble, unlike most cannabinoid derivatives, and was researched for potential use as an analgesic or sedative,[3] although it was never developed for clinical use and is not currently used in medicine, as dronabinol or nabilone were felt to be more useful. However it is sometimes used in research into the potential therapeutic applications of cannabinoids.
References
- ^ Razdan RK. The Total Synthesis of Cannabinoids. Wiley-Interscience 1980
- ^ Razdan RK, Howes JF. "Drugs related to tetrahydrocannabinol." Medicinal Research Reviews 1983; 3(2):119-146. PMID 6134882
- ^ Archer RA. "The cannabinoids: therapeutic potentials." Annual Reports in Medicinal Chemistry 1974; 9: 253-259. PMID 12307093
| Psychedelics (5-HT2A agonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dissociatives (NMDAR antagonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Deliriants (mAChR antagonists) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Receptor (ligands) |
| ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Transporter (modulators) |
| ||||||||||||||||||||||||||||||
| Enzyme (modulators) |
| ||||||||||||||||||||||||||||||
| Others |
| ||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||
This cannabinoid related article is a stub. You can help Wikipedia by expanding it. |
- Chem-molar-mass both hardcoded and calculated
- Infobox-drug molecular-weight unexpected-character
- Pages using infobox drug with unknown parameters
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Drugs with no legal status
- All stub articles
