Oxaloacetic acid

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Oxobutanedioic acid | |
| Other names
Oxaloacetic acid
Oxalacetic acid 2-Oxosuccinic acid Ketosuccinic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.005.755 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H4O5 | |
| Molar mass | 132.07 g/mol |
| Density | 1.6 g/cm3 |
| Melting point | 161 °C (322 °F; 434 K) |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
-943.21 kJ/mol |
Std enthalpy of
combustion (ΔcH⦵298) |
-1205.58 kJ/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
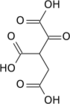
Oxaloacetic acid (also known as oxalacetic acid or OAA) is a crystalline organic compound with the chemical formula HO2CC(O)CH2CO2H. Oxaloacetic acid, in the form of its conjugate base oxaloacetate, is a metabolic intermediate in many processes that occur in animals. It takes part in gluconeogenesis, the urea cycle, the glyoxylate cycle, amino acid synthesis, fatty acid synthesis and the citric acid cycle.[1]
Properties
[edit]Oxaloacetic acid undergoes successive deprotonations to give the dianion:
- HO2CC(O)CH2CO2H ⇌ −O2CC(O)CH2CO2H + H+, pKa = 2.22
- −O2CC(O)CH2CO2H ⇌ −O2CC(O)CH2CO2− + H+, pKa = 3.89
At high pH, the enolizable proton is ionized:
- −O2CC(O)CH2CO2− ⇌ −O2CC(O−)CHCO2− + H+, pKa = 13.03
The enol forms of oxaloacetic acid are particularly stable. Keto-enol tautomerization is catalyzed by the enzyme oxaloacetate tautomerase. trans-Enol-oxaloacetate also appears when tartrate is the substrate for fumarase.[2]

Biosynthesis
[edit]Oxaloacetate forms in several ways in nature. A principal route is upon oxidation of L-malate, catalyzed by malate dehydrogenase, in the citric acid cycle. Malate is also oxidized by succinate dehydrogenase in a slow reaction with the initial product being enol-oxaloacetate.[3]
It also arises from the condensation of pyruvate with carbonic acid, driven by the hydrolysis of ATP:
- CH3C(O)CO2− + HCO3− + ATP → −O2CCH2C(O)CO2− + ADP + Pi
Occurring in the mesophyll of plants, this process proceeds via phosphoenolpyruvate, catalysed by phosphoenolpyruvate carboxylase.
Oxaloacetate can also arise from trans- or de- amination of aspartic acid.
Biochemical functions
[edit]Oxaloacetate is an intermediate of the citric acid cycle, where it reacts with acetyl-CoA to form citrate, catalyzed by citrate synthase. It is also involved in gluconeogenesis, the urea cycle, the glyoxylate cycle, amino acid synthesis, and fatty acid synthesis. Oxaloacetate is also a potent inhibitor of complex II.
Gluconeogenesis
[edit]Gluconeogenesis[1] is a metabolic pathway consisting of a series of eleven enzyme-catalyzed reactions, resulting in the generation of glucose from non-carbohydrates substrates. The beginning of this process takes place in the mitochondrial matrix, where pyruvate molecules are found. A pyruvate molecule is carboxylated by a pyruvate carboxylase enzyme, activated by a molecule each of ATP and water. This reaction results in the formation of oxaloacetate. NADH reduces oxaloacetate to malate. This transformation is needed to transport the molecule out of the mitochondria. Once in the cytosol, malate is oxidized to oxaloacetate again using NAD+. Then oxaloacetate remains in the cytosol, where the rest of reactions will take place. Oxaloacetate is later decarboxylated and phosphorylated by phosphoenolpyruvate carboxykinase and becomes 2-phosphoenolpyruvate using guanosine triphosphate (GTP) as phosphate source. Glucose is obtained after further downstream processing.
Urea cycle
[edit]The urea cycle is a metabolic pathway that results in the formation of urea using one ammonium molecule from degraded amino acids, another ammonium group from aspartate and one bicarbonate molecule.[1] This route commonly occurs in hepatocytes. The reactions related to the urea cycle produce NADH, and NADH can be produced in two different ways. One of these uses oxaloacetate. In the cytosol there are fumarate molecules. Fumarate can be transformed into malate by the actions of the enzyme fumarase. Malate is acted on by malate dehydrogenase to become oxaloacetate, producing a molecule of NADH. After that, oxaloacetate will be recycled to aspartate, as transaminases prefer these keto acids over the others. This recycling maintains the flow of nitrogen into the cell.

Glyoxylate cycle
[edit]The glyoxylate cycle is a variant of the citric acid cycle.[4] It is an anabolic pathway occurring in plants and bacteria utilizing the enzymes isocitrate lyase and malate synthase. Some intermediate steps of the cycle are slightly different from the citric acid cycle; nevertheless oxaloacetate has the same function in both processes.[1] This means that oxaloacetate in this cycle also acts as the primary reactant and final product. In fact the oxaloacetate is a net product of the glyoxylate cycle because its loop of the cycle incorporates two molecules of acetyl-CoA.
Fatty acid synthesis
[edit]In previous stages acetyl-CoA is transferred from the mitochondria to the cytoplasm where fatty acid synthase resides. The acetyl-CoA is transported as a citrate, which has been previously formed in the mitochondrial matrix from acetyl-CoA and oxaloacetate. This reaction usually initiates the citric acid cycle, but when there is no need of energy it is transported to the cytoplasm where it is broken down to cytoplasmic acetyl-CoA and oxaloacetate.
Another part of the cycle requires NADPH for the synthesis of fatty acids.[5] Part of this reducing power is generated when the cytosolic oxaloacetate is returned to the mitochondria as long as the internal mitochondrial layer is non-permeable for oxaloacetate. Firstly the oxaloacetate is reduced to malate using NADH. Then the malate is decarboxylated to pyruvate. Now this pyruvate can easily enter the mitochondria, where it is carboxylated again to oxaloacetate by pyruvate carboxylase. In this way, the transfer of acetyl-CoA that is from the mitochondria into the cytoplasm produces a molecule of NADH. The overall reaction, which is spontaneous, may be summarized as:
- HCO3– + ATP + acetyl-CoA → ADP + Pi + malonyl-CoA
Amino acid synthesis
[edit]Six essential amino acids and three nonessential are synthesized from oxaloacetate and pyruvate.[6] Aspartate and alanine are formed from oxaloacetate and pyruvate, respectively, by transamination from glutamate. Asparagine is synthesized by amidation of aspartate, with glutamine donating the NH4. These are nonessential amino acids, and their simple biosynthetic pathways occur in all organisms. Methionine, threonine, lysine, isoleucine, valine, and leucine are essential amino acids in humans and most vertebrates. Their biosynthetic pathways in bacteria are complex and interconnected.

Oxalate biosynthesis
[edit]Oxaloacetate produces oxalate by hydrolysis.[7]
- oxaloacetate + H2O ⇌ oxalate + acetate
This process is catalyzed by the enzyme oxaloacetase. This enzyme is seen in plants, but is not known in the animal kingdom.[8]
Interactive pathway map
[edit]| Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
Glycolysis and Gluconeogenesis edit
|
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
TCACycle_WP78 edit
|
See also
[edit]References
[edit]- ^ a b c d Nelson, David L.; Cox, Michael M. (2005). Principles of Biochemistry (4th ed.). New York: W. H. Freeman. ISBN 0-7167-4339-6.
- ^ van Vugt-Lussenburg, BMA; van der Weel, L; Hagen, WR; Hagedoorn, P-L (26 February 2021), "Biochemical Similarities and Differences between the Catalytic [4Fe-4S] Cluster Containing Fumarases FumA and FumB from Escherichia coli", PLOS ONE, 8 (2) (published 6 February 2013): e55549, doi:10.1371/journal.pone.0055549, PMC 3565967, PMID 23405168
- ^ M.V. Panchenko; A.D. Vinogradov (1991). "Direct demonstration of enol-oxaloacetate as an immediate product of malate oxidation by the mammalian succinate dehydrogenase". FEBS Letters. 286 (1–2): 76–78. Bibcode:1991FEBSL.286...76P. doi:10.1016/0014-5793(91)80944-X. PMID 1864383.
- ^ "Welcome to The Chemistry Place". www.pearsonhighered.com. Retrieved 5 April 2018.
- ^ "fatty acids synthesis". www.rpi.edu.
- ^ "Animo acids synthesized from oxaloacetate and pyruvate". faculty.ksu.edu.sa. Archived from the original (PPTX) on 21 October 2013. Retrieved 21 October 2013.
- ^ Gadd, Geoffrey M. "Fungal production of citric and oxalic acid: importance in metal speciation, physiology and biogeochemical processes" Advances in Microbial Physiology (1999), 41, 47-92.
- ^ Xu, Hua-Wei. "Oxalate accumulation and regulations is independent of glycolate oxidase in rice leaves" Journal of Experimental Botany, Vol 57, No. 9 pp. 1899-1908, 2006