Peroxydicarbonate
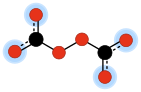
In chemistry, peroxydicarbonate (sometimes peroxodicarbonate) is a divalent anion with the chemical formula C
2O2−
6. It is one of the oxocarbon anions, which consist solely of carbon and oxygen. Its molecular structure can be viewed as two carbonate anions joined so as to form a peroxide bridge –O–O–.
The anion is formed, together with peroxocarbonate CO2−
4, at the negative electrode during electrolysis of molten lithium carbonate.[1] The anion can also be obtained by electrolysis of a saturated solution of rubidium carbonate in water.[2]
In addition, the peroxodicarbonate anion can be obtained by electrosynthesis on boron doped diamond (BDD) during water oxidation.[3][4] The formal oxidation of two carbonate ions takes place at the anode. Due to the high oxidation potential of the peroxodicarbonate anion, a high anodic overpotential is necessary. This is even more important if hydroxyl radicals are involved in the formation process. Recent publications show that a concentration of 282 mmol/L of peroxodicarbonate can be reached in an undivided cell with sodium carbonate as starting material at current densities of 720 mA/cm2.[5] The described process is suitable for the pilot scale production of sodium peroxodicarbonate.
Potassium peroxydicarbonate K2C2O6 was obtained by Constam and von Hansen in 1895;[6] its crystal structure was determined only in 2002.[7] It too can be obtained by electrolysis of a saturated potassium carbonate solution at −20 °C. It is a light blue crystalline solid that decomposes at 141 °C, releasing oxygen and carbon dioxide, and decomposes slowly at lower temperatures.[7]
Rubidium peroxodicarbonate is a light blue crystalline solid that decomposes at 424 K (151 °C). Its structure was published in 2003.[2] In both salts, each of the two carbonate units is planar. In the rubidium salt the whole molecule is planar, whereas in the potassium salt the two units lie on different and nearly perpendicular planes, both of which contain the O–O bond.[2]
See also
[edit]References
[edit]- ^ Chen, Li-Jiang; Lin, Chang-Jian; Zuo, Juan; Song, Ling-Chun; Huang, Chao-Ming (2004). "First Spectroscopic Observation of Peroxocarbonate/ Peroxodicarbonate in Molten Carbonate". The Journal of Physical Chemistry B. 108 (23): 7553–7556. doi:10.1021/jp035749l.
- ^ a b c Dinnebier, Robert E.; Vensky, Sascha; Jansen, Martin (2003). "Crystal and Molecular Structure of Rubidium Peroxodicarbonate Rb2[C2O6]". Chemistry: A European Journal. 9 (18): 4391–4395. doi:10.1002/chem.200304914. PMID 14502625.
- ^ Mavrikis, Sotirios; Göltz, Maximilian; Rosiwal, Stefan; Wang, Ling; Ponce de León, Carlos (2022-02-18). "Carbonate‐Induced Electrosynthesis of Hydrogen Peroxide via Two‐Electron Water Oxidation". ChemSusChem. 15 (4). doi:10.1002/cssc.202102137. ISSN 1864-5631. PMID 34935302. S2CID 245384604.
- ^ Seitz, Ann‐Katrin; Kohlpaintner, Philipp J.; Lingen, Tim; Dyga, Marco; Sprang, Fiona; Zirbes, Michael; Waldvogel, Siegfried R.; Gooßen, Lukas J. (2022-04-25). "Concentrated Aqueous Peroxodicarbonate: Efficient Electrosyn‐ thesis and Use as Oxidizer in Epoxidations, S ‐, and N ‐Oxidations". Angewandte Chemie International Edition. 61 (25). doi:10.1002/anie.202117563. ISSN 1433-7851. PMC 9324847. PMID 35384198.
- ^ Chardon, Chloé P.; Matthée, Thorsten; Neuber, Rieke; Fryda, Matthias; Comninellis, Christos (2017-01-23). "Efficient Electrochemical Production of Peroxodicarbonate Applying DIACHEM®Diamond Electrodes". ChemistrySelect. 2 (3): 1037–1040. doi:10.1002/slct.201601583. ISSN 2365-6549.
- ^ Constam, E. J.; von Hansen, A. (1896). "Elektrolytische Darstellung einer neuen Klasse oxidierender Substanzen". Zeitschrift für Elektrochemie. 3 (7): 137–144. doi:10.1002/bbpc.189600032. S2CID 179087570.
- ^ a b Dinnebier, Robert E.; Vensky, Sascha; Stephens, Peter W.; Jansen, Martin (2002). "Crystal Structure of K2[C2O6]". Angewandte Chemie International Edition. 41 (11): 1922–1924. doi:10.1002/1521-3773(20020603)41:11<1922::AID-ANIE1922>3.0.CO;2-T.
