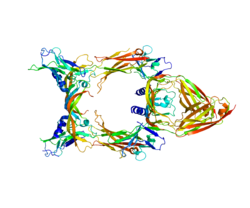
Vascular endothelial growth factor C
Vascular endothelial growth factor C (VEGF-C) is a protein that is a member of the platelet-derived growth factor / vascular endothelial growth factor (PDGF/VEGF) family. It is encoded in humans by the VEGFC gene, which is located on chromosome 4q34.[5]
Functions
[edit]The main function of VEGF-C is to promote the growth of lymphatic vessels (lymphangiogenesis). It acts on lymphatic endothelial cells (LECs) primarily via its receptor VEGFR-3 promoting survival, growth and migration. It was discovered in 1996 as a ligand for the orphan receptor VEGFR-3.[6] Soon thereafter, it was shown to be a specific growth factor for lymphatic vessels in a variety of models.[7][8] However, in addition to its effect on lymphatic vessels, it can also promote the growth of blood vessels and regulate their permeability. The effect on blood vessels can be mediated via its primary receptor VEGFR-3[9] or its secondary receptor VEGFR-2. Apart from vascular targets, VEGF-C is also important for neural development[10] and blood pressure regulation.[11]
Biosynthesis
[edit]VEGF-C is a dimeric, secreted protein, which undergoes a complex proteolytic maturation resulting in multiple processed forms. After translation, VEGF-C consists of three domains: the central VEGF homology domain (VHD), the N-terminal domain (propeptide) and a C-terminal domain (propeptide).[12] It is referred to as "uncleaved VEGF-C" and has a size of approximately 58 kDa. The first cleavage (which happens already before secretion) occurs between the VHD and the C-terminal domain and is mediated by proprotein convertases.[13] However, the resulting protein is still held together by disulfide bonds and remains inactive (although it can bind already VEGFR-3).[14] This form is referred to as "intermediate form" or pro-VEGF-C and it consists of two polypeptide chains of 29 and 31 kDa. In order to activate VEGF-C, a second cleavage has to occur between the N-terminal propeptide and the VHD. This cleavage can be performed either by ADAMTS3,[14] plasmin,[15] KLK3/PSA or cathepsin D.[16] With progressing maturation, the affinity of VEGF-C for both VEGFR-2 and VEGFR-3 increases and only the fully processed, mature forms of VEGF-C have a significant affinity for VEGFR-2.[12]
Relationship to VEGF-D
[edit]The closest structural and functional relative of VEGF-C is VEGF-D.[17] However, at least in mice, VEGF-C is absolutely essential for the development of the lymphatic system,[18] whereas VEGF-D appears to be unnecessary.[19] Whether this holds true for humans is unknown, because there are major differences between human and mouse VEGF-D.[20]
Disease relevance
[edit]In a minority of lymphedema patients, the condition is caused by mutations in the VEGFC gene[21] and VEGF-C is a potential treatment for lymphedema,[22][23] even though the underlying molecular cause appears more often in the VEGF-Receptor-3 instead of VEGF-C itself.[24] Because in Milroy's disease (Hereditary lymphedema type I), only one allele is mutated, not all VEGFR-3 molecules are non-functional and it is thought, that high amounts of VEGF-C can compensate for the mutated, nonfunctional receptors by increasing the signaling levels of the remaining functional receptors.[25] Therefore, VEGF-C is developed as a lymphedema drug under the name of Lymfactin.[26] Also indirectly VEGF-C can be responsible for hereditary lymphedema: The rare Hennekam syndrome can result from the inability of the mutated CCBE1 to assist the ADAMTS3 protease in activating VEGF-C.[14] While lack of VEGF-C results in lymphedema, VEGF-C production is implicated in tumor lymphangiogenesis and metastasis. Expression of VEGF-C by tumors induces peri-tumoral and intratumoral lymphangiogenesis what potently promotes metastatic dissemination of tumor cells.[27][28] VEGF-C primarily stimulates lymphangiogenesis by activating VEGFR-3, yet under certain conditions it can also act directly on blood vessels to promote tumor angiogenesis.[9][29]
Evolution
[edit]The PDGF family is so closely related to the VEGF family that the two are sometimes grouped together as the PDGF/VEGF family. In invertebrates, molecules from this families are not easily distinguished from each other and are collectively referred to as PVFs (PDGF/VEGF-like growth factors.[30] The comparison of human VEGFs with these PVFs allows conclusions on the structure of the ancestral molecules, which appear more closely related to today's lymphangiogenic VEGF-C than to the other members of the VEGF family and despite their large evolutionary distance are still able to interact with human VEGF receptors. The PVFs in Drosophila melanogaster have functions for the migration of hemocytes[31] and the PVFs in the jellyfish Podocoryne carnea for the development of the tentacles and the gastrovascular apparatus.[32] However, the function of the PVF-1 of the nematode Caenorhabditis elegans is unknown[30]
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000150630 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000031520 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Paavonen K, Horelli-Kuitunen N, Chilov D, Kukk E, Pennanen S, Kallioniemi OP, et al. (March 1996). "Novel human vascular endothelial growth factor genes VEGF-B and VEGF-C localize to chromosomes 11q13 and 4q34, respectively". Circulation. 93 (6): 1079–1082. doi:10.1161/01.CIR.93.6.1079. PMID 8653826.
- ^ Joukov V, Pajusola K, Kaipainen A, Chilov D, Lahtinen I, Kukk E, et al. (January 1996). "A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases". The EMBO Journal. 15 (2): 290–298. doi:10.1002/j.1460-2075.1996.tb00359.x. PMC 449944. PMID 8617204.
- ^ Oh SJ, Jeltsch MM, Birkenhäger R, McCarthy JE, Weich HA, Christ B, et al. (August 1997). "VEGF and VEGF-C: specific induction of angiogenesis and lymphangiogenesis in the differentiated avian chorioallantoic membrane". Developmental Biology. 188 (1): 96–109. doi:10.1006/dbio.1997.8639. PMID 9245515.
- ^ Jeltsch M, Kaipainen A, Joukov V, Meng X, Lakso M, Rauvala H, et al. (May 1997). "Hyperplasia of lymphatic vessels in VEGF-C transgenic mice". Science. 276 (5317): 1423–1425. doi:10.1126/science.276.5317.1423. PMID 9162011. S2CID 21835142.
- ^ a b Tammela T, Zarkada G, Wallgard E, Murtomäki A, Suchting S, Wirzenius M, et al. (July 2008). "Blocking VEGFR-3 suppresses angiogenic sprouting and vascular network formation". Nature. 454 (7204): 656–660. Bibcode:2008Natur.454..656T. doi:10.1038/nature07083. PMID 18594512. S2CID 2251527.
- ^ Le Bras B, Barallobre MJ, Homman-Ludiye J, Ny A, Wyns S, Tammela T, et al. (March 2006). "VEGF-C is a trophic factor for neural progenitors in the vertebrate embryonic brain". Nature Neuroscience. 9 (3): 340–348. doi:10.1038/nn1646. PMID 16462734. S2CID 24197350.
- ^ Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, et al. (May 2009). "Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism". Nature Medicine. 15 (5): 545–552. doi:10.1038/nm.1960. PMID 19412173. S2CID 10526891.
- ^ a b Joukov V, Sorsa T, Kumar V, Jeltsch M, Claesson-Welsh L, Cao Y, et al. (July 1997). "Proteolytic processing regulates receptor specificity and activity of VEGF-C". The EMBO Journal. 16 (13): 3898–3911. doi:10.1093/emboj/16.13.3898. PMC 1170014. PMID 9233800.
- ^ Siegfried G, Basak A, Cromlish JA, Benjannet S, Marcinkiewicz J, Chrétien M, et al. (June 2003). "The secretory proprotein convertases furin, PC5, and PC7 activate VEGF-C to induce tumorigenesis". The Journal of Clinical Investigation. 111 (11): 1723–1732. doi:10.1172/JCI17220. PMC 156106. PMID 12782675.
- ^ a b c Jeltsch M, Jha SK, Tvorogov D, Anisimov A, Leppänen VM, Holopainen T, et al. (May 2014). "CCBE1 enhances lymphangiogenesis via A disintegrin and metalloprotease with thrombospondin motifs-3-mediated vascular endothelial growth factor-C activation". Circulation. 129 (19): 1962–1971. doi:10.1161/CIRCULATIONAHA.113.002779. PMID 24552833.
- ^ McColl BK, Baldwin ME, Roufail S, Freeman C, Moritz RL, Simpson RJ, et al. (September 2003). "Plasmin activates the lymphangiogenic growth factors VEGF-C and VEGF-D". The Journal of Experimental Medicine. 198 (6): 863–868. doi:10.1084/jem.20030361. PMC 2194198. PMID 12963694.
- ^ Jha SK, Rauniyar K, Chronowska E, Mattonet K, Maina EW, Koistinen H, et al. (May 2019). "KLK3/PSA and cathepsin D activate VEGF-C and VEGF-D". eLife. 8: 44478. doi:10.7554/eLife.44478. PMC 6588350. PMID 31099754.
- ^ Achen MG, Jeltsch M, Kukk E, Mäkinen T, Vitali A, Wilks AF, et al. (January 1998). "Vascular endothelial growth factor D (VEGF-D) is a ligand for the tyrosine kinases VEGF receptor 2 (Flk1) and VEGF receptor 3 (Flt4)". Proceedings of the National Academy of Sciences of the United States of America. 95 (2): 548–553. Bibcode:1998PNAS...95..548A. doi:10.1073/pnas.95.2.548. PMC 18457. PMID 9435229.
- ^ Karkkainen MJ, Haiko P, Sainio K, Partanen J, Taipale J, Petrova TV, et al. (January 2004). "Vascular endothelial growth factor C is required for sprouting of the first lymphatic vessels from embryonic veins". Nature Immunology. 5 (1): 74–80. doi:10.1038/ni1013. PMID 14634646. S2CID 22078757.
- ^ Baldwin ME, Halford MM, Roufail S, Williams RA, Hibbs ML, Grail D, et al. (March 2005). "Vascular endothelial growth factor D is dispensable for development of the lymphatic system". Molecular and Cellular Biology. 25 (6): 2441–2449. doi:10.1128/MCB.25.6.2441-2449.2005. PMC 1061605. PMID 15743836.
- ^ Baldwin ME, Catimel B, Nice EC, Roufail S, Hall NE, Stenvers KL, et al. (June 2001). "The specificity of receptor binding by vascular endothelial growth factor-d is different in mouse and man". The Journal of Biological Chemistry. 276 (22): 19166–19171. doi:10.1074/jbc.M100097200. PMID 11279005. S2CID 41677159.
- ^ Balboa-Beltran E, Fernández-Seara MJ, Pérez-Muñuzuri A, Lago R, García-Magán C, Couce ML, et al. (July 2014). "A novel stop mutation in the vascular endothelial growth factor-C gene (VEGFC) results in Milroy-like disease". Journal of Medical Genetics. 51 (7): 475–478. doi:10.1136/jmedgenet-2013-102020. PMID 24744435. S2CID 6613861.
- ^ Enholm B, Karpanen T, Jeltsch M, Kubo H, Stenback F, Prevo R, et al. (March 2001). "Adenoviral expression of vascular endothelial growth factor-C induces lymphangiogenesis in the skin". Circulation Research. 88 (6): 623–629. doi:10.1161/01.RES.88.6.623. PMID 11282897. S2CID 28806663.
- ^ Honkonen KM, Visuri MT, Tervala TV, Halonen PJ, Koivisto M, Lähteenvuo MT, et al. (May 2013). "Lymph node transfer and perinodal lymphatic growth factor treatment for lymphedema". Annals of Surgery. 257 (5): 961–7. doi:10.1097/SLA.0b013e31826ed043. PMID 23013803. S2CID 2042145.
- ^ Brouillard P, Boon L, Vikkula M (March 2014). "Genetics of lymphatic anomalies". The Journal of Clinical Investigation. 124 (3): 898–904. doi:10.1172/JCI71614. PMC 3938256. PMID 24590274.
- ^ Karkkainen MJ, Saaristo A, Jussila L, Karila KA, Lawrence EC, Pajusola K, et al. (October 2001). "A model for gene therapy of human hereditary lymphedema". Proceedings of the National Academy of Sciences of the United States of America. 98 (22): 12677–12682. Bibcode:2001PNAS...9812677K. doi:10.1073/pnas.221449198. PMC 60113. PMID 11592985.
- ^ Herantis Pharma (2014-07-21). "Lymfactin® for lymphedema".
{{cite web}}: Missing or empty|url=(help) - ^ Skobe M, Hawighorst T, Jackson DG, Prevo R, Janes L, Velasco P, et al. (February 2001). "Induction of tumor lymphangiogenesis by VEGF-C promotes breast cancer metastasis". Nature Medicine. 7 (2): 192–198. doi:10.1038/84643. PMID 11175850. S2CID 26090359.
- ^ Mandriota SJ, Jussila L, Jeltsch M, Compagni A, Baetens D, Prevo R, et al. (February 2001). "Vascular endothelial growth factor-C-mediated lymphangiogenesis promotes tumour metastasis". The EMBO Journal. 20 (4): 672–682. doi:10.1093/emboj/20.4.672. PMC 145430. PMID 11179212.
- ^ Tvorogov D, Anisimov A, Zheng W, Leppänen VM, Tammela T, Laurinavicius S, et al. (December 2010). "Effective suppression of vascular network formation by combination of antibodies blocking VEGFR ligand binding and receptor dimerization". Cancer Cell. 18 (6): 630–640. doi:10.1016/j.ccr.2010.11.001. PMID 21130043.
- ^ a b Tarsitano M, De Falco S, Colonna V, McGhee JD, Persico MG (February 2006). "The C. elegans pvf-1 gene encodes a PDGF/VEGF-like factor able to bind mammalian VEGF receptors and to induce angiogenesis". FASEB Journal. 20 (2): 227–233. doi:10.1096/fj.05-4147com. PMID 16449794. S2CID 31963203.
- ^ Heino TI, Kärpänen T, Wahlström G, Pulkkinen M, Eriksson U, Alitalo K, Roos C (November 2001). "The Drosophila VEGF receptor homolog is expressed in hemocytes". Mechanisms of Development. 109 (1): 69–77. doi:10.1016/S0925-4773(01)00510-X. PMID 11677054. S2CID 14074572.
- ^ Seipel K, Eberhardt M, Müller P, Pescia E, Yanze N, Schmid V (October 2004). "Homologs of vascular endothelial growth factor and receptor, VEGF and VEGFR, in the jellyfish Podocoryne carnea". Developmental Dynamics. 231 (2): 303–312. doi:10.1002/dvdy.20139. PMID 15366007. S2CID 42930371.
Further reading
[edit]- Rauniyar K, Jha SK, Jeltsch M (Feb 2018). "Biology of Vascular Endothelial Growth Factor C in the Morphogenesis of Lymphatic Vessels". Frontiers in Bioengineering and Biotechnology. 6: 7. doi:10.3389/fbioe.2018.00007. PMC 5816233. PMID 29484295.
- Krebs R, Jeltsch M (June 2013). "The lymphangiogenic growth factors VEGF-C and VEGF-D. Part 1: Basic principles and embryonic development" (PDF). Lymphologie in Forschung und Praxis. 17 (1): 30–37.
- Krebs R, Jeltsch M (Dec 2013). "Die lymphangiogenic growth factors VEGF-C and VEGF-D. Part 2: The role of VEGF-C and VEGF-D in diseases of the lymphatic system" (PDF). Lymphologie in Forschung und Praxis. 17 (2): 96–104.
- Orpana A, Salven P (February 2002). "Angiogenic and lymphangiogenic molecules in hematological malignancies". Leukemia & Lymphoma. 43 (2): 219–224. doi:10.1080/10428190290005964. PMID 11999550. S2CID 21908151.
- Orpana A, Salven P (February 2002). "Angiogenic and lymphangiogenic molecules in hematological malignancies". Leukemia & Lymphoma. 43 (2): 219–224. doi:10.1080/10428190290005964. PMID 11999550. S2CID 21908151.
- Joukov V, Pajusola K, Kaipainen A, Chilov D, Lahtinen I, Kukk E, et al. (January 1996). "A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases". The EMBO Journal. 15 (2): 290–298. doi:10.1002/j.1460-2075.1996.tb00359.x. PMC 449944. PMID 8617204.
- Joukov V, Pajusola K, Kaipainen A, Chilov D, Lahtinen I, Kukk E, et al. (April 1996). "A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases". The EMBO Journal. 15 (7): 1751. doi:10.1002/j.1460-2075.1996.tb00521.x. PMC 450088. PMID 8612600.
- Paavonen K, Horelli-Kuitunen N, Chilov D, Kukk E, Pennanen S, Kallioniemi OP, et al. (March 1996). "Novel human vascular endothelial growth factor genes VEGF-B and VEGF-C localize to chromosomes 11q13 and 4q34, respectively". Circulation. 93 (6): 1079–1082. doi:10.1161/01.cir.93.6.1079. PMID 8653826.
- Lee J, Gray A, Yuan J, Luoh SM, Avraham H, Wood WI (March 1996). "Vascular endothelial growth factor-related protein: a ligand and specific activator of the tyrosine kinase receptor Flt4". Proceedings of the National Academy of Sciences of the United States of America. 93 (5): 1988–1992. Bibcode:1996PNAS...93.1988L. doi:10.1073/pnas.93.5.1988. PMC 39896. PMID 8700872.
- Joukov V, Sorsa T, Kumar V, Jeltsch M, Claesson-Welsh L, Cao Y, et al. (July 1997). "Proteolytic processing regulates receptor specificity and activity of VEGF-C". The EMBO Journal. 16 (13): 3898–3911. doi:10.1093/emboj/16.13.3898. PMC 1170014. PMID 9233800.
- Fitz LJ, Morris JC, Towler P, Long A, Burgess P, Greco R, et al. (July 1997). "Characterization of murine Flt4 ligand/VEGF-C". Oncogene. 15 (5): 613–618. doi:10.1038/sj.onc.1201191. PMID 9247316. S2CID 6795440.
- Dunk C, Ahmed A (April 2001). "Expression of VEGF-C and activation of its receptors VEGFR-2 and VEGFR-3 in trophoblast". Histology and Histopathology. 16 (2): 359–375. doi:10.14670/HH-16.359. PMID 11332691.
- Dias S, Choy M, Alitalo K, Rafii S (March 2002). "Vascular endothelial growth factor (VEGF)-C signaling through FLT-4 (VEGFR-3) mediates leukemic cell proliferation, survival, and resistance to chemotherapy". Blood. 99 (6): 2179–2184. doi:10.1182/blood.V99.6.2179. PMID 11877295. S2CID 2838185.
- Ueda M, Terai Y, Yamashita Y, Kumagai K, Ueki K, Yamaguchi H, et al. (March 2002). "Correlation between vascular endothelial growth factor-C expression and invasion phenotype in cervical carcinomas". International Journal of Cancer. 98 (3): 335–343. doi:10.1002/ijc.10193. PMID 11920583. S2CID 10276013.
- Witte D, Thomas A, Ali N, Carlson N, Younes M (2002). "Expression of the vascular endothelial growth factor receptor-3 (VEGFR-3) and its ligand VEGF-C in human colorectal adenocarcinoma". Anticancer Research. 22 (3): 1463–1466. PMID 12168824.
- Schoppmann SF, Birner P, Stöckl J, Kalt R, Ullrich R, Caucig C, et al. (September 2002). "Tumor-associated macrophages express lymphatic endothelial growth factors and are related to peritumoral lymphangiogenesis". The American Journal of Pathology. 161 (3): 947–956. doi:10.1016/S0002-9440(10)64255-1. PMC 1867252. PMID 12213723.
- Shin HY, Smith ML, Toy KJ, Williams PM, Bizios R, Gerritsen ME (December 2002). "VEGF-C mediates cyclic pressure-induced endothelial cell proliferation". Physiological Genomics. 11 (3): 245–251. doi:10.1152/physiolgenomics.00068.2002. PMID 12388793. S2CID 14183060.
- Yu DH, Wen YM, Sun JD, Wei SL, Xie HP, Pang FH (March 2002). "[Relationship among expression of vascular endothelial growth factor-C(VEGF-C), angiogenesis, lymphangiogenesis, and lymphatic metastasis in oral cancer]". AI Zheng = Aizheng = Chinese Journal of Cancer. 21 (3): 319–22. PMID 12452004.
- Nakashima T, Kondoh S, Kitoh H, Ozawa H, Okita S, Harada T, et al. (January 2003). "Vascular endothelial growth factor-C expression in human gallbladder cancer and its relationship to lymph node metastasis". International Journal of Molecular Medicine. 11 (1): 33–39. doi:10.3892/ijmm.11.1.33. PMID 12469214.
- Tsai PW, Shiah SG, Lin MT, Wu CW, Kuo ML (February 2003). "Up-regulation of vascular endothelial growth factor C in breast cancer cells by heregulin-beta 1. A critical role of p38/nuclear factor-kappa B signaling pathway". The Journal of Biological Chemistry. 278 (8): 5750–5759. doi:10.1074/jbc.M204863200. PMID 12471041. S2CID 40780576.
- Masood R, Kundra A, Zhu S, Xia G, Scalia P, Smith DL, Gill PS (May 2003). "Malignant mesothelioma growth inhibition by agents that target the VEGF and VEGF-C autocrine loops". International Journal of Cancer. 104 (5): 603–610. doi:10.1002/ijc.10996. PMID 12594815. S2CID 23085374.
- Ohno M, Nakamura T, Kunimoto Y, Nishimura K, Chung-Kang C, Kuroda Y (2004). "Lymphagenesis correlates with expression of vascular endothelial growth factor-C in colorectal cancer". Oncology Reports. 10 (4): 939–943. doi:10.3892/or.10.4.939. PMID 12792749.