Portal:Minerals
Portal maintenance status: (May 2019)
|
The Minerals Portal

In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.
The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks.
The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases.
Some natural solid substances without a definite crystalline structure, such as opal or obsidian, are more properly called mineraloids. If a chemical compound occurs naturally with different crystal structures, each structure is considered a different mineral species. Thus, for example, quartz and stishovite are two different minerals consisting of the same compound, silicon dioxide. (Full article...)
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...)
Selected articles
-
Image 1Beachy Head is a part of the extensive Southern England Chalk Formation.
Chalk is a soft, white, porous, sedimentary carbonate rock. It is a form of limestone composed of the mineral calcite and originally formed deep under the sea by the compression of microscopic plankton that had settled to the sea floor. Chalk is common throughout Western Europe, where deposits underlie parts of France, and steep cliffs are often seen where they meet the sea in places such as the Dover cliffs on the Kent coast of the English Channel.
Chalk is mined for use in industry, such as for quicklime, bricks and builder's putty, and in agriculture, for raising pH in soils with high acidity. It is also used for "blackboard chalk" for writing and drawing on various types of surfaces, although these can also be manufactured from other carbonate-based minerals, or gypsum. (Full article...) -
Image 2

Talc, or talcum, is a clay mineral composed of hydrated magnesium silicate, with the chemical formula Mg3Si4O10(OH)2. Talc in powdered form, often combined with corn starch, is used as baby powder. This mineral is used as a thickening agent and lubricant. It is an ingredient in ceramics, paints, and roofing material. It is a main ingredient in many cosmetics. It occurs as foliated to fibrous masses, and in an exceptionally rare crystal form. It has a perfect basal cleavage and an uneven flat fracture, and it is foliated with a two-dimensional platy form.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 1 as the hardness of talc, the softest mineral. When scraped on a streak plate, talc produces a white streak, though this indicator is of little importance, because most silicate minerals produce a white streak. Talc is translucent to opaque, with colors ranging from whitish grey to green with a vitreous and pearly luster. Talc is not soluble in water, and is slightly soluble in dilute mineral acids.
Soapstone is a metamorphic rock composed predominantly of talc. (Full article...) -
Image 3

Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite.
Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum.
Rutile derives its name from the Latin rutilus ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner using specimens obtained in Horcajuelo de la Sierra, Madrid (Spain), which is consequently the type locality. (Full article...) -
Image 4Deep green isolated fluorite crystal resembling a truncated octahedron, set upon a micaceous matrix, from Erongo Mountain, Erongo Region, Namibia (overall size: 50 mm × 27 mm, crystal size: 19 mm wide, 30 g)
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite.
Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glases, so fluorite is useful in making apochromatic lenses, making it valuable, particularly in photographic optics. Fluorite optics are also usable in the far-ultraviolet and mid-infrared ranges, where conventional glasses are too opaque for use. Fluorite also has low dispersion, and a high refractive index for its density, which can make it useful for some specialized purposes in optics. (Full article...) -
Image 5A sample of andesite (dark groundmass) with amygdaloidal vesicles filled with zeolite. Diameter of view is 8 cm.
Andesite (/ˈændəzaɪt/) is a volcanic rock of intermediate composition. In a general sense, it is the intermediate type between silica-poor basalt and silica-rich rhyolite. It is fine-grained (aphanitic) to porphyritic in texture, and is composed predominantly of sodium-rich plagioclase plus pyroxene or hornblende.
Andesite is the extrusive equivalent of plutonic diorite. Characteristic of subduction zones, andesite represents the dominant rock type in island arcs. The average composition of the continental crust is andesitic. Along with basalts, andesites are a component of the Martian crust.
The name andesite is derived from the Andes mountain range, where this rock type is found in abundance. It was first applied by Christian Leopold von Buch in 1826. (Full article...) -
Image 6A ruby crystal from Dodoma Region, Tanzania
Ruby is a pinkish red to blood-red colored gemstone, a variety of the mineral corundum (aluminium oxide). Ruby is one of the most popular traditional jewelry gems and is very durable. Other varieties of gem-quality corundum are called sapphires. Ruby is one of the traditional cardinal gems, alongside amethyst, sapphire, emerald, and diamond. The word ruby comes from ruber, Latin for red. The color of a ruby is due to the element chromium.
Some gemstones that are popularly or historically called rubies, such as the Black Prince's Ruby in the British Imperial State Crown, are actually spinels. These were once known as "Balas rubies".
The quality of a ruby is determined by its color, cut, and clarity, which, along with carat weight, affect its value. The brightest and most valuable shade of red, called blood-red or pigeon blood, commands a large premium over other rubies of similar quality. After color follows clarity: similar to diamonds, a clear stone will command a premium, but a ruby without any needle-like rutile inclusions may indicate that the stone has been treated. Ruby is the traditional birthstone for July and is usually pinker than garnet, although some rhodolite garnets have a similar pinkish hue to most rubies. The world's most valuable ruby to be sold at auction is the Sunrise Ruby. (Full article...) -
Image 7
Beryl (/ˈbɛrəl/ BERR-əl) is a mineral composed of beryllium aluminium silicate with the chemical formula Be3Al2Si6O18. Well-known varieties of beryl include emerald and aquamarine. Naturally occurring hexagonal crystals of beryl can be up to several meters in size, but terminated crystals are relatively rare. Pure beryl is colorless, but it is frequently tinted by impurities; possible colors are green, blue, yellow, pink, and red (the rarest). It is an ore source of beryllium. (Full article...) -
Image 8

Chalcopyrite (/ˌkælkəˈpaɪˌraɪt, -koʊ-/ KAL-kə-PY-ryte, -koh-) is a copper iron sulfide mineral and the most abundant copper ore mineral. It has the chemical formula CuFeS2 and crystallizes in the tetragonal system. It has a brassy to golden yellow color and a hardness of 3.5 to 4 on the Mohs scale. Its streak is diagnostic as green-tinged black.
On exposure to air, chalcopyrite tarnishes to a variety of oxides, hydroxides, and sulfates. Associated copper minerals include the sulfides bornite (Cu5FeS4), chalcocite (Cu2S), covellite (CuS), digenite (Cu9S5); carbonates such as malachite and azurite, and rarely oxides such as cuprite (Cu2O). It is rarely found in association with native copper. Chalcopyrite is a conductor of electricity.
Copper can be extracted from chalcopyrite ore using various methods. The two predominant methods are pyrometallurgy and hydrometallurgy, the former being the most commercially viable. (Full article...) -
Image 9

Green fluorite with prominent cleavage
Cleavage, in mineralogy and materials science, is the tendency of crystalline materials to split along definite crystallographic structural planes. These planes of relative weakness are a result of the regular locations of atoms and ions in the crystal, which create smooth repeating surfaces that are visible both in the microscope and to the naked eye. If bonds in certain directions are weaker than others, the crystal will tend to split along the weakly bonded planes. These flat breaks are termed "cleavage". The classic example of cleavage is mica, which cleaves in a single direction along the basal pinacoid, making the layers seem like pages in a book. In fact, mineralogists often refer to "books of mica".
Diamond and graphite provide examples of cleavage. Each is composed solely of a single element, carbon. In diamond, each carbon atom is bonded to four others in a tetrahedral pattern with short covalent bonds. The planes of weakness (cleavage planes) in a diamond are in four directions, following the faces of the octahedron. In graphite, carbon atoms are contained in layers in a hexagonal pattern where the covalent bonds are shorter (and thus even stronger) than those of diamond. However, each layer is connected to the other with a longer and much weaker van der Waals bond. This gives graphite a single direction of cleavage, parallel to the basal pinacoid. So weak is this bond that it is broken with little force, giving graphite a slippery feel as layers shear apart. As a result, graphite makes an excellent dry lubricant.
While all single crystals will show some tendency to split along atomic planes in their crystal structure, if the differences between one direction or another are not large enough, the mineral will not display cleavage. Corundum, for example, displays no cleavage. (Full article...) -
Image 10

Corundum is a crystalline form of aluminium oxide (Al2O3) typically containing traces of iron, titanium, vanadium, and chromium. It is a rock-forming mineral. It is a naturally transparent material, but can have different colors depending on the presence of transition metal impurities in its crystalline structure. Corundum has two primary gem varieties: ruby and sapphire. Rubies are red due to the presence of chromium, and sapphires exhibit a range of colors depending on what transition metal is present. A rare type of sapphire, padparadscha sapphire, is pink-orange.
The name "corundum" is derived from the Tamil-Dravidian word kurundam (ruby-sapphire) (appearing in Sanskrit as kuruvinda).
Because of corundum's hardness (pure corundum is defined to have 9.0 on the Mohs scale), it can scratch almost all other minerals. It is commonly used as an abrasive on sandpaper and on large tools used in machining metals, plastics, and wood. Emery, a variety of corundum with no value as a gemstone, is commonly used as an abrasive. It is a black granular form of corundum, in which the mineral is intimately mixed with magnetite, hematite, or hercynite.
In addition to its hardness, corundum has a density of 4.02 g/cm3 (251 lb/cu ft), which is unusually high for a transparent mineral composed of the low-atomic mass elements aluminium and oxygen. (Full article...) -
Image 11

A crystalline solid: atomic resolution image of strontium titanate. Brighter spots are columns of strontium atoms and darker ones are titanium-oxygen columns.
Crystallography is the branch of science devoted to the study of molecular and crystalline structure and properties. The word crystallography is derived from the Ancient Greek word κρύσταλλος (krústallos; "clear ice, rock-crystal"), and γράφειν (gráphein; "to write"). In July 2012, the United Nations recognised the importance of the science of crystallography by proclaiming 2014 the International Year of Crystallography.
Crystallography is a broad topic, and there are many subareas such as X-ray crystallography which are themselves large scientific topics. It spans science ranging from the fundamentals of crystal structure to the mathematics of crystal geometry including those which are not periodic or quasicrystals. At the atomic scale it can involve the use of X-ray diffraction to produce experimental data which the tools of X-ray crystallography can convert into detailed positions of atoms and sometimes electron density. At the larger scale it includes experimental tools such as orientational imaging to examine the relative orientations at the grain boundary in materials. Crystallography plays a key role in many areas of established biology, chemistry and physics as well new developments in these fields. (Full article...) -
Image 12Amethyst cluster from Artigas, Uruguay
Amethyst is a violet variety of quartz. The name comes from the Koine Greek αμέθυστος amethystos from α- a-, "not" and μεθύσκω (Ancient Greek) methysko / μεθώ metho (Modern Greek), "intoxicate", a reference to the belief that the stone protected its owner from drunkenness. Ancient Greeks wore amethyst and carved drinking vessels from it in the belief that it would prevent intoxication.
Amethyst, a semiprecious stone, is often used in jewelry. (Full article...) -
Image 13
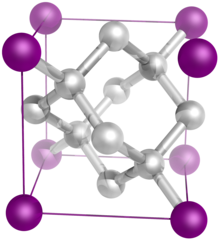
The diamond crystal structure belongs to the face-centered cubic lattice, with a repeated two-atom pattern.
In crystallography, a crystal system is a set of point groups (a group of geometric symmetries with at least one fixed point). A lattice system is a set of Bravais lattices. Space groups are classified into crystal systems according to their point groups, and into lattice systems according to their Bravais lattices. Crystal systems that have space groups assigned to a common lattice system are combined into a crystal family.
The seven crystal systems are triclinic, monoclinic, orthorhombic, tetragonal, trigonal, hexagonal, and cubic. Informally, two crystals are in the same crystal system if they have similar symmetries (though there are many exceptions). (Full article...) -
Image 14
The mineral pyrite (/ˈpaɪraɪt/ PY-ryte), or iron pyrite, also known as fool's gold, is an iron sulfide with the chemical formula FeS2 (iron (II) disulfide). Pyrite is the most abundant sulfide mineral.
Pyrite's metallic luster and pale brass-yellow hue give it a superficial resemblance to gold, hence the well-known nickname of fool's gold. The color has also led to the nicknames brass, brazzle, and brazil, primarily used to refer to pyrite found in coal.
The name pyrite is derived from the Greek πυρίτης λίθος (pyritēs lithos), 'stone or mineral which strikes fire', in turn from πῦρ (pŷr), 'fire'. In ancient Roman times, this name was applied to several types of stone that would create sparks when struck against steel; Pliny the Elder described one of them as being brassy, almost certainly a reference to what is now called pyrite.
By Georgius Agricola's time, c. 1550, the term had become a generic term for all of the sulfide minerals. (Full article...) -
Image 15

Garnets ( /ˈɡɑːrnɪt/) are a group of silicate minerals that have been used since the Bronze Age as gemstones and abrasives.
All species of garnets possess similar physical properties and crystal forms, but differ in chemical composition. The different species are pyrope, almandine, spessartine, grossular (varieties of which are hessonite or cinnamon-stone and tsavorite), uvarovite and andradite. The garnets make up two solid solution series: pyrope-almandine-spessartine (pyralspite), with the composition range [Mg,Fe,Mn]3Al2(SiO4)3; and uvarovite-grossular-andradite (ugrandite), with the composition range Ca3[Cr,Al,Fe]2(SiO4)3. (Full article...) -
Image 16Halite from the Wieliczka salt mine, Małopolskie, Poland
Halite (/ˈhælaɪt, ˈheɪlaɪt/ HAL-yte, HAY-lyte), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride (NaCl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, pink, red, orange, yellow or gray depending on inclusion of other materials, impurities, and structural or isotopic abnormalities in the crystals. It commonly occurs with other evaporite deposit minerals such as several of the sulfates, halides, and borates. The name halite is derived from the Ancient Greek word for "salt", ἅλς (háls). (Full article...) -
Image 17

Cinnabar (/ˈsɪnəˌbɑːr/; from Ancient Greek κιννάβαρι (kinnábari)), or cinnabarite (/ˌsɪnəˈbɑːraɪt/), also known as mercurblende is the bright scarlet to brick-red form of mercury(II) sulfide (HgS). It is the most common source ore for refining elemental mercury and is the historic source for the brilliant red or scarlet pigment termed vermilion and associated red mercury pigments.
Cinnabar generally occurs as a vein-filling mineral associated with volcanic activity and alkaline hot springs. The mineral resembles quartz in symmetry and it exhibits birefringence. Cinnabar has a mean refractive index near 3.2, a hardness between 2.0 and 2.5, and a specific gravity of approximately 8.1. The color and properties derive from a structure that is a hexagonal crystalline lattice belonging to the trigonal crystal system, crystals that sometimes exhibit twinning.
Cinnabar has been used for its color since antiquity in the Near East, including as a rouge-type cosmetic, in the New World since the Olmec culture, and in China since as early as the Yangshao culture, where it was used in coloring stoneware.
Associated modern precautions for the use and handling of cinnabar arise from the toxicity of the mercury component, which was recognized as early as ancient Rome. (Full article...) -
Image 18
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the exception of extremely rare native iron deposits, it is the most magnetic of all the naturally occurring minerals on Earth. Naturally magnetized pieces of magnetite, called lodestone, will attract small pieces of iron, which is how ancient peoples first discovered the property of magnetism.
Magnetite is black or brownish-black with a metallic luster, has a Mohs hardness of 5–6 and leaves a black streak. Small grains of magnetite are very common in igneous and metamorphic rocks.
The chemical IUPAC name is iron(II,III) oxide and the common chemical name is ferrous-ferric oxide. (Full article...) -
Image 19

Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula CaSO4·2H2O. It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, drywall and blackboard or sidewalk chalk. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison.
Fine-grained white or lightly tinted forms of gypsum known as alabaster have been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. (Full article...) -
Image 20

Tourmaline (/ˈtʊərməlɪn, -ˌliːn/ TOOR-mə-lin, -leen) is a crystalline silicate mineral group in which boron is compounded with elements such as aluminium, iron, magnesium, sodium, lithium, or potassium. This gemstone comes in a wide variety of colors.
The name is derived from the Sinhalese tōramalli (ටෝරමල්ලි), which refers to the carnelian gemstones. (Full article...) -
Image 21
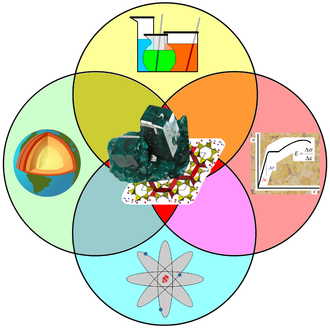
Mineralogy applies principles of chemistry, geology, physics and materials science to the study of minerals
Mineralogy is a subject of geology specializing in the scientific study of the chemistry, crystal structure, and physical (including optical) properties of minerals and mineralized artifacts. Specific studies within mineralogy include the processes of mineral origin and formation, classification of minerals, their geographical distribution, as well as their utilization. (Full article...) -
Image 22
Apatite is a group of phosphate minerals, usually hydroxyapatite, fluorapatite and chlorapatite, with high concentrations of OH−, F− and Cl− ion, respectively, in the crystal. The formula of the admixture of the three most common endmembers is written as Ca10(PO4)6(OH,F,Cl)2, and the crystal unit cell formulae of the individual minerals are written as Ca10(PO4)6(OH)2, Ca10(PO4)6F2 and Ca10(PO4)6Cl2.
The mineral was named apatite by the German geologist Abraham Gottlob Werner in 1786, although the specific mineral he had described was reclassified as fluorapatite in 1860 by the German mineralogist Karl Friedrich August Rammelsberg. Apatite is often mistaken for other minerals. This tendency is reflected in the mineral's name, which is derived from the Greek word ἀπατάω (apatáō), which means to deceive. (Full article...) -
Image 23
Opal is a hydrated amorphous form of silica (SiO2·nH2O); its water content may range from 3% to 21% by weight, but is usually between 6% and 10%. Due to its amorphous property, it is classified as a mineraloid, unlike crystalline forms of silica, which are considered minerals. It is deposited at a relatively low temperature and may occur in the fissures of almost any kind of rock, being most commonly found with limonite, sandstone, rhyolite, marl, and basalt.
The name opal is believed to be derived from the Sanskrit word upala (उपल), which means 'jewel', and later the Greek derivative opállios (ὀπάλλιος).
There are two broad classes of opal: precious and common. Precious opal displays play-of-color (iridescence); common opal does not. Play-of-color is defined as "a pseudo chromatic optical effect resulting in flashes of colored light from certain minerals, as they are turned in white light." The internal structure of precious opal causes it to diffract light, resulting in play-of-color. Depending on the conditions in which it formed, opal may be transparent, translucent, or opaque, and the background color may be white, black, or nearly any color of the visual spectrum. Black opal is considered the rarest, while white, gray, and green opals are the most common. (Full article...) -
Image 24

Kaolinite (/ˈkeɪ.ələˌnaɪt, -lɪ-/ KAY-ə-lə-nyte, -lih-; also called kaolin) is a clay mineral, with the chemical composition: Al2Si2O5(OH)4. It is a layered silicate mineral, with one tetrahedral sheet of silica (SiO4) linked through oxygen atoms to one octahedral sheet of alumina (AlO6).
Kaolinite is a soft, earthy, usually white, mineral (dioctahedral phyllosilicate clay), produced by the chemical weathering of aluminium silicate minerals like feldspar. It has a low shrink–swell capacity and a low cation-exchange capacity (1–15 meq/100 g).
Rocks that are rich in kaolinite, and halloysite, are known as kaolin (/ˈkeɪ.əlɪn/) or china clay. In many parts of the world kaolin is colored pink-orange-red by iron oxide, giving it a distinct rust hue. Lower concentrations of iron oxide yield the white, yellow, or light orange colors of kaolin. Alternating lighter and darker layers are sometimes found, as at Providence Canyon State Park in Georgia, United States.
Kaolin is an important raw material in many industries and applications. Commercial grades of kaolin are supplied and transported as powder, lumps, semi-dried noodle or slurry. Global production of kaolin in 2021 was estimated to be 45 million tonnes, with a total market value of $US4.24 billion. (Full article...) -
Image 25Quartz crystal cluster from Brazil
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon–oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is, therefore, classified structurally as a framework silicate mineral and compositionally as an oxide mineral. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar.
Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at 573 °C (846 K; 1,063 °F). Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold.
There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Europe and Asia.
Quartz is the mineral defining the value of 7 on the Mohs scale of hardness, a qualitative scratch method for determining the hardness of a material to abrasion. (Full article...)
Selected mineralogist
-
Image 1

Jean-Claude Delamétherie
Jean-Claude Delamétherie (also de La Métherie, de Lamétherie, 4 September 1743 – 1 July 1817) was a French mineralogist, geologist and paleontologist. (Full article...) -
Image 2

George Washington Carpenter (July 31, 1802 – June 7, 1860) was an American scientist. (Full article...) -
Image 3Oskar Alexander Richard Büttner (28 September 1858 – 1927) was a German botanist and mineralogist who was involved in the exploration of the Congo Basin. (Full article...)
-
Image 4

Félix Pisani (1831–1920)
Félix Pisani (28 April 1831, Constantinople – 7 November 1920, Paris) was a French chemist and mineralogist.
He was born in Istanbul, where his Venetian father worked in the Russian diplomatic service. Beginning in 1854, he studied chemistry in Paris at a private school run by Charles Frédéric Gerhardt (1816–1856). (Full article...) -
Image 5
-
Image 6

Arthur Moritz Schoenflies (German: [ˈʃøːnfliːs]; 17 April 1853 – 27 May 1928), sometimes written as Schönflies, was a German mathematician, known for his contributions to the application of group theory to crystallography, and for work in topology.
Schoenflies was born in Landsberg an der Warthe (modern Gorzów, Poland). Arthur Schoenflies married Emma Levin (1868–1939) in 1896. He studied under Ernst Kummer and Karl Weierstrass, and was influenced by Felix Klein. (Full article...) -
Image 7Adolarius Jacob Forster (1739–1806) was a Prussian mineralogist and dealer in display specimen minerals. The Forster family left Yorkshire in 1649 and settled in Prussia. Adolarius Jacob Forster began dealing in mineral specimens around 1766, at the age of 27. He continued in that profession for 40 years and travelled widely. He had premises in London, Paris and St. Petersburg. The Covent Garden, London shop and one in Soho was run by his wife. His brother, Ingham Henry Forster (1725–1782) ran the business in Paris. Auction catalogues for sales in Paris were written by Rome de l'Isle.
He was related to Johann Georg Adam Forster and Johann Reinhold Forster and his sister married the London dealer naturalist George Humphrey at St-Martin-in-the-Fields, London on August 16, 1768. In 1802 Forster sold a collection to the museum of the St Petersburg Mining Institute, under the auspices of the Emperor of All Russia Alexander I. He spent the last ten years of his life in Russia, and died in St. Petersburg in 1806. The dealership was taken over by his nephew John Henry Heuland. (Full article...) -
Image 8

Karl Franz Anton von Schreibers
Carl Franz Anton Ritter von Schreibers (15 August 1775 – 21 May 1852) was an Austrian naturalist who was a native of Pressburg, Hungary, Habsburg Empire (today Bratislava, Slovakia).
In 1847, an uncommon iron-nickel-phosphide ((Fe,Ni)3P) mineral was named in his honor by Wilhelm Karl Ritter von Haidinger (1775–1871). The mineral is found in meteorites, and is known today as schreibersite. As a zoologist, he was the first to perform a comprehensive anatomical study of the olm, a cave-dwelling, aquatic amphibian. The plant genus Schreibersia (synonym Augusta, family Rubiaceae) was named in his honor by Johann Baptist Emanuel Pohl. The common bent-wing bat (Miniopterus schreibersii), also known as the Schreibers's long-fingered bat or Schreibers's bat, is a species of insectivorous bat. They appear to have dispersed from a subtropical origin and distributed throughout the southern Palearctic, Ethiopic, Oriental, and Australian regions. In Europe, it is present in the southern half on the continent from Iberia to the Caucasus, with the largest populations found in the warmer Mediterranean area. The common and scientific names honor Carl Franz Anton Ritter von Schreibers. (Full article...) -
Image 9For the French Napoleonic War General see Marshall Étienne Macdonald
Marshall McDonald (October 18, 1835 – September 1, 1895) was an American engineer, geologist, mineralogist, pisciculturist, and fisheries scientist. McDonald served as the commissioner of the United States Commission of Fish and Fisheries from 1888 until his death in 1895. He is best known for his inventions of a number of fish hatching apparatuses and a fish ladder that enabled salmon and other migrating fish species to ascend the rapids of watercourses resulting in an increased spawning ground. McDonald's administration of the U.S. Commission of Fish and Fisheries was notably free of scandal and furthered the "protection and culture" of fish species throughout the United States. (Full article...) -
Image 10Jean-André Mongez (21 November 1750 – May 1788) was a French priest and mineralogist. He is presumed to have died at Vanikoro, on the La Pérouse expedition. (Full article...)
-
Image 11

Matthias Joseph Anker.
Matthias Joseph Anker (6 May 1771 – 3 April 1843) was an Austrian mineralogist and geologist born in Graz. Some sources place his birthdate as 1 May 1772.
He received his education in Vienna, afterwards working as a surgeon in the town of Stainz. In 1807, he was called to Graz as a district surgeon, from where he intensified his scientific studies in mineralogy. Four years later, he joined the staff of mineralogist Friedrich Mohs at the Johanneum in Graz, where he eventually became a professor of mineralogy as well as curator of the mineral cabinet. In 1839, he resigned from his teaching position, but stayed on as director of collections. (Full article...) -
Image 12Charles-Victor Mauguin (French: [ʃaʁl.vik.tɔʁ mo.gɛ̃]; 19 September 1878 – 25 April 1958), more often Charles Mauguin, was a French mineralogist and crystallographer. He and Carl Hermann invented an international standard notation for crystallographic groups called Hermann–Mauguin notation (also sometimes called international notation). (Full article...)
-
Image 13Portrait by Oscar Pereira da Silva
José Bonifácio de Andrada e Silva (Portuguese pronunciation: [ʒuˈzɛ boniˈfasju dʒi ɐ̃ˈdɾadɐ i ˈsiwvɐ]; 13 June 1763 – 6 April 1838) was a Brazilian statesman, naturalist, mineralist, professor and poet, born in Santos, São Paulo, then part of the Portuguese Empire.
He was one of the most important mentors of Brazilian independence, and his actions were decisive for the success of Emperor Pedro I. He supported public education, was an abolitionist and suggested that a new national capital be created in Brazil's underdeveloped interior (effected over a century later as Brasília). His career as naturalist was marked by the discovery of four new minerals. (Full article...) -
Image 14
Georgius Agricola (/əˈɡrɪkələ/; born Georg Bauer; 24 March 1494 – 21 November 1555) was a German Humanist scholar, mineralogist and metallurgist. Born in the small town of Glauchau, in the Electorate of Saxony of the Holy Roman Empire, he was broadly educated, but took a particular interest in the mining and refining of metals. He was the first to drop the Arabic definite article al-, exclusively writing chymia and chymista in describing activity that we today would characterize as chemical or alchemical, giving chemistry its modern name. For his groundbreaking work De Natura Fossilium published in 1546, he is generally referred to as the Father of Mineralogy and the founder of geology as a scientific discipline.
He is well known for his pioneering work De re metallica libri XII, that was published in 1556, one year after his death. This 12-volume work is a comprehensive and systematic study, classification and methodical guide on all available factual and practical aspects, that are of concern for mining, the mining sciences and metallurgy, investigated and researched in its natural environment by means of direct observation. Unrivalled in its complexity and accuracy, it served as the standard reference work for two centuries. Agricola stated in the preface, that he will exclude "all those things which I have not myself seen, or have not read or heard of". He continued, "That which I have neither seen, nor carefully considered after reading or hearing of, I have not written about." (Full article...) -
Image 15

Hermann Traube (1860–1913)
Hermann Traube (September 24, 1860 – January 29, 1913) was a German mineralogist born in Ratibor, Silesia (presently Racibórz, Poland). He was the son of chemist Moritz Traube (1826–1894).
He studied at the Universities of Leipzig, Heidelberg, Breslau and Greifswald, earning his doctorate in 1884. At Breslau his instructors were Ferdinand Cohn (1828–1898) and Theodor Poleck (1821–1906). (Full article...) -
Image 16Stanley Robert Mitchell (12 February 1881 – 22 March 1963) was an Australian commercial metallurgist as well as an amateur mineralogist and ethnologist. (Full article...)
-
Image 17

René Just Haüy (French pronunciation: [aɥi]) FRS MWS FRSE (28 February 1743 – 1 June 1822) was a French priest and mineralogist, commonly styled the Abbé Haüy after he was made an honorary canon of Notre Dame. Due to his innovative work on crystal structure and his four-volume Traité de Minéralogie (1801), he is often referred to as the "Father of Modern Crystallography". During the French Revolution he also helped to establish the metric system. (Full article...) -
Image 18

Abraham Gottlob Werner (German: [ˈaːbʁaham ˈɡɔtloːp ˈvɛʁnɐ]; 25 September 1749 – 30 June 1817) was a German geologist who set out an early theory about the stratification of the Earth's crust and propounded a history of the Earth that came to be known as Neptunism. While most tenets of Neptunism were eventually set aside, Werner is remembered for his demonstration of chronological succession in rocks; for the zeal with which he infused his pupils; and for the impulse he thereby gave to the study of geology. He has been called the "father of German geology". (Full article...) -
Image 19
Evgraf Stepanovich Fedorov (Russian: Евгра́ф Степа́нович Фёдоров, 22 December [O.S. 10 December] 1853 – 21 May 1919) was a Russian mathematician, crystallographer and mineralogist.
Fedorov was born in the Russian city of Orenburg. His father was a topographical engineer. The family later moved to Saint Petersburg. From the age of fifteen, he was deeply interested in the theory of polytopes, which later became his main research interest. He was a distinguished graduate of the Gorny Institute, which he joined at the age of 26. He was elected the first Director of the Institute in 1905. (Full article...) -
Image 20Portrait by John Opie c1795
Philip Rashleigh FRS FSA (28 December 1729 – 26 June 1811) of Menabilly, Cornwall, was an antiquary and Fellow of the Royal Society and a Cornish squire. He collected and published the Trewhiddle Hoard of Anglo-Saxon treasure, which still gives its name to the "Trewhiddle style" of 9th century decoration. (Full article...) -
Image 21
Sten Anders Hjalmar Sjögren (13 June 1856, Färnebo, Värmland – 23 March 1922, Stockholm) was a Swedish geologist and mineralogist. (Full article...) -
Image 22

Der Mineraloge (The Mineralogist) by Raphael Ritz, 1883. Thought to depict the Bonn mineralogist Gerhard vom Rath (1830-1888), examining mineral specimens from Lengenbach in a nearby house in Binn, Switzerland
Gerhard vom Rath (20 August 1830 – 23 April 1888), was a German mineralogist, born at Duisburg in Prussia. (Full article...) -
Image 23Jacques-Louis, Comte de Bournon FRS, FGS (21 January 1751 – 24 August 1825) was a French soldier and mineralogist who came to England after the French Revolution. He gained prominence in the scientific community, being elected a fellow of the Royal Society and was a founding member of the Geological Society before returning to France after the Bourbon Restoration. (Full article...)
-
Image 24
Franz Ludwig von Cancrin
Franz Ludwig von Cancrin (February 21, 1738 in Breidenbach – 1812) was a German mineralogist, metallurgist, architect and writer.
He was born into a German mining family where he was trained by his father in the science of mining. In 1764, he entered the service of the landgrave of Hesse-Kassel at Hanau, becoming professor of mathematics at the military academy, head of the civil engineering department of the state, director of the theatre and (1774) of the mint. A work on the copper mines of Elesse (1767) earned him a European reputation, and in 1783 he accepted from Catherine II of Russia the directorship of the famous Staraya salt-works, living thenceforth in Russia. (Full article...) -
Image 25

Christian Ernst Weiss (12 May 1833, in Eilenburg – 4 July 1890, in Schkeuditz) was a German mineralogist, geologist and palaeontologist.
He is not to be confused with the historian Christian Ernst Weiße (1766–1832). (Full article...)
Related portals
Get involved
For editor resources and to collaborate with other editors on improving Wikipedia's Minerals-related articles, see WikiProject Rocks and minerals.
General images
-
Image 1Mohs hardness kit, containing one specimen of each mineral on the ten-point hardness scale (from Mohs scale)
-
Image 2Native gold. Rare specimen of stout crystals growing off of a central stalk, size 3.7 x 1.1 x 0.4 cm, from Venezuela. (from Mineral)
-
Image 3An example of elbaite, a species of tourmaline, with distinctive colour banding. (from Mineral)
-
Image 4Black andradite, an end-member of the orthosilicate garnet group. (from Mineral)
-
Image 6Muscovite, a mineral species in the mica group, within the phyllosilicate subclass (from Mineral)
-
Image 7Mohs Scale versus Absolute Hardness (from Mineral)
-
Image 9Asbestiform tremolite, part of the amphibole group in the inosilicate subclass (from Mineral)
-
Image 10Epidote often has a distinctive pistachio-green colour. (from Mineral)
-
Image 12Sphalerite crystal partially encased in calcite from the Devonian Milwaukee Formation of Wisconsin (from Mineral)
-
Image 14Pink cubic halite (NaCl; halide class) crystals on a nahcolite matrix (NaHCO3; a carbonate, and mineral form of sodium bicarbonate, used as baking soda). (from Mineral)
-
Image 16Schist is a metamorphic rock characterized by an abundance of platy minerals. In this example, the rock has prominent sillimanite porphyroblasts as large as 3 cm (1.2 in). (from Mineral)
-
Image 17When minerals react, the products will sometimes assume the shape of the reagent; the product mineral is termed a pseudomorph of (or after) the reagent. Illustrated here is a pseudomorph of kaolinite after orthoclase. Here, the pseudomorph preserved the Carlsbad twinning common in orthoclase. (from Mineral)
-
Image 19Perfect basal cleavage as seen in biotite (black), and good cleavage seen in the matrix (pink orthoclase). (from Mineral)
-
Image 20Hübnerite, the manganese-rich end-member of the wolframite series, with minor quartz in the background (from Mineral)
-
Image 21Gypsum desert rose (from Mineral)
-
Image 23Diamond is the hardest natural material, and has a Mohs hardness of 10. (from Mineral)
-
Image 24Red cinnabar (HgS), a mercury ore, on dolomite. (from Mineral)
In the news
- 3 May 2024 –
- Panama bans First Quantum Minerals from extracting copper following the closure of its Cobre Panamá mine last year. (Reuters) (The Globe and Mail)
Did you know ...?
- ... that sérandite (pictured) was discovered in Guinea and described in a French journal, and the type material resides in Washington, D.C.?
- ... that the mineral gatehouseite has been found in only one mine in South Australia?
- ... that the mineral motukoreaite is named for Motukorea, the island in New Zealand where it was discovered?
Subcategories
Topics
| Overview | ||
|---|---|---|
| Common minerals | ||
Ore minerals, mineral mixtures and ore deposits | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ores |
| ||||||||
| Deposit types | |||||||||
| Borates | |||||
|---|---|---|---|---|---|
| Carbonates | |||||
| Oxides |
| ||||
| Phosphates | |||||
| Silicates | |||||
| Sulfides | |||||
| Other |
| ||||
| Crystalline | |||||||
|---|---|---|---|---|---|---|---|
| Cryptocrystalline | |||||||
| Amorphous | |||||||
| Miscellaneous | |||||||
| Notable varieties |
| ||||||
| Oxide minerals |
| ||||
|---|---|---|---|---|---|
| Silicate minerals | |||||
| Other | |||||
Gemmological classifications by E. Ya. Kievlenko (1980), updated | |||||||||
| Jewelry stones |
| ||||||||
| Jewelry-Industrial stones |
| ||||||||
| Industrial stones |
| ||||||||
Mineral identification | |
|---|---|
| "Special cases" ("native elements and organic minerals") |
|
|---|---|
| "Sulfides and oxides" |
|
| "Evaporites and similars" |
|
| "Mineral structures with tetrahedral units" (sulfate anion, phosphate anion, silicon, etc.) |
|
Associated Wikimedia
The following Wikimedia Foundation sister projects provide more on this subject:
-
Commons
Free media repository -
Wikibooks
Free textbooks and manuals -
Wikidata
Free knowledge base -
Wikinews
Free-content news -
Wikiquote
Collection of quotations -
Wikisource
Free-content library -
Wikiversity
Free learning tools -
Wiktionary
Dictionary and thesaurus
References
- Pages with German IPA
- Pages with French IPA
- Pages with Portuguese IPA
- Manually maintained portal pages from May 2019
- All manually maintained portal pages
- Portals with triaged subpages from May 2019
- All portals with triaged subpages
- Portals with named maintainer
- Automated article-slideshow portals with 31–40 articles in article list
- Automated article-slideshow portals with 201–500 articles in article list
- Portals needing placement of incoming links