High-resolution melting analysis: Difference between revisions
No edit summary |
m →Cite journal with Wikipedia template filling, links |
||
| Line 3: | Line 3: | ||
==High Resolution Melting (HRM) analysis== |
==High Resolution Melting (HRM) analysis== |
||
In molecular biology '''High Resolution Melt''' or HRM analysis as it will be referred to herein is a powerful technique for the detection of |
In [[molecular biology]] '''High Resolution Melt''' or HRM analysis as it will be referred to herein is a powerful technique for the detection of [[mutation]]s, polymorphisms and [[epigenetic]] differences in double stranded DNA samples. It was discovered and developed by Idaho Technology and the University of Utah<ref>for academic treatment of the history of HRM see http://www.dna.utah.edu/Hi-Res/TOP_Hi-Res%20Melting.html</ref>. It has advantages over other [[genotyping]] technologies. Namely: |
||
* It is cost effective vs. other genotyping technologies such as sequencing and Taqman SNP typing. This makes it ideal for large scale genotyping projects. |
* It is cost effective vs. other genotyping technologies such as sequencing and Taqman SNP typing. This makes it ideal for large scale genotyping projects. |
||
| Line 25: | Line 25: | ||
[[Image:DNA melting scematic curve 2.jpg|500px]] |
[[Image:DNA melting scematic curve 2.jpg|500px]] |
||
'''Wild type, heterozygote or homozygote?''' |
'''Wild type, [[heterozygote]] or [[homozygote]]?''' |
||
Things become slightly more complicated than this because organisms contain two copies of each gene, known as the two |
Things become slightly more complicated than this because organisms contain two copies of each gene, known as the two [[allele]]s. So, if a sample is taken from a patient and amplified using PCR both copies of the region of DNA (alleles) of interest are amplified. So if we are looking for mutation there are now three possibilities: |
||
# Neither allele contains a mutation |
# Neither allele contains a mutation |
||
| Line 39: | Line 39: | ||
: |
: |
||
'''SNP typing/Point mutation detection''' <br /> |
'''SNP typing/Point mutation detection''' <br /> |
||
Conventional [[Single-nucleotide polymorphism|SNP]] typing methods are typically time consuming and expensive, requiring several probe bases assays to be multiplexed together or the use of DNA microarrays. HRM is more cost effective and reduces the need to design multiple pairs of primers and the need to purchase expensive probes. The HRM method has been successfully used to detect a single G to A substitution in the gene Vssc (Voltage Sensitive Sodium Channel) which confers resistance to the acaricide [[permethrin]] in Scabies mite. This mutation results in a coding change in the protein (G1535D). The analysis of scabies mites collected from suspected permethrin susceptible and tolerant populations by HRM showed distinct melting profiles. The [[amplicons]] from the sensitive mites were observed to have a higher melting temperature relative to the tolerant mites, as expected from the higher thermostability of the GC [[base pair]] <ref> |
Conventional [[Single-nucleotide polymorphism|SNP]] typing methods are typically time consuming and expensive, requiring several probe bases assays to be multiplexed together or the use of DNA microarrays. HRM is more cost effective and reduces the need to design multiple pairs of primers and the need to purchase expensive probes. The HRM method has been successfully used to detect a single G to A substitution in the gene Vssc (Voltage Sensitive Sodium Channel) which confers resistance to the acaricide [[permethrin]] in Scabies mite. This mutation results in a coding change in the protein (G1535D). The analysis of scabies mites collected from suspected permethrin susceptible and tolerant populations by HRM showed distinct melting profiles. The [[amplicons]] from the sensitive mites were observed to have a higher melting temperature relative to the tolerant mites, as expected from the higher thermostability of the GC [[base pair]] <ref>{{cite journal |author=Pasay C, Arlian L, Morgan M, ''et al.'' |title=High-resolution melt analysis for the detection of a mutation associated with permethrin resistance in a population of scabies mites |journal=Med. Vet. Entomol. |volume=22 |issue=1 |pages=82–8 |year=2008 |month=March |pmid=18380658 |doi=10.1111/j.1365-2915.2008.00716.x |url=http://www3.interscience.wiley.com/resolve/openurl?genre=article&sid=nlm:pubmed&issn=0269-283X&date=2008&volume=22&issue=1&spage=82}}</ref><br/> |
||
In a field more relevant to clinical diagnostics, HRM has been shown to be suitable in principle for the detection of mutations in the breast cancer susceptibility genes BRCA1 and BRCA2. More than 400 mutations have been identified in these genes.<br />The sequencing of genes is the gold standard for identifying mutations. Sequencing is time consuming and labour intensive and is often preceded by techniques used to identify heteroduplex DNA, which then further amplify these issues. HRM offers a faster and more convenient closed-tube method of assessing the presence of mutations and gives a result which can be further investigated if it is of interest. In a study carried out by Scott et al. in 2006 <ref> |
In a field more relevant to clinical diagnostics, HRM has been shown to be suitable in principle for the detection of mutations in the breast cancer susceptibility genes BRCA1 and BRCA2. More than 400 mutations have been identified in these genes.<br />The sequencing of genes is the gold standard for identifying mutations. Sequencing is time consuming and labour intensive and is often preceded by techniques used to identify heteroduplex DNA, which then further amplify these issues. HRM offers a faster and more convenient closed-tube method of assessing the presence of mutations and gives a result which can be further investigated if it is of interest. In a study carried out by Scott et al. in 2006 <ref>{{cite journal |author=James PA, Doherty R, Harris M, ''et al.'' |title=Optimal selection of individuals for BRCA mutation testing: a comparison of available methods |journal=J. Clin. Oncol. |volume=24 |issue=4 |pages=707–15 |year=2006 |month=February |pmid=16446345 |doi=10.1200/JCO.2005.01.9737 |url=http://jco.ascopubs.org/cgi/content/full/24/4/707}}</ref>, 3 cell lines harbouring different BRCA mutations were used to assess the HRM methodology. It was found that the melting profiles of the resulting PCR products could be used to distinguish the presence or absence of a mutation in the amplicon. Similarly in 2007 Krypuy et al. <ref>{{cite journal |author=Krypuy M, Ahmed AA, Etemadmoghadam D, ''et al.'' |title=High resolution melting for mutation scanning of TP53 exons 5-8 |journal=BMC Cancer |volume=7 |issue= |pages=168 |year=2007 |pmid=17764544 |pmc=2025602 |doi=10.1186/1471-2407-7-168 |url=http://www.biomedcentral.com/1471-2407/7/168}}</ref>. showed that the careful design of HRM assays (with regards to primer placement) could be successfully employed to detect mutations in the TP53 gene, which encodes the tumour suppressor protein p53 in clinical samples of breast and ovarian cancer. Both these studies highlighted that fact that changes in the melting profile can be in the form of a shift in the melting temperature or an obvious difference in the shape of the melt curve. Both of these parameters are a function of the amplicon sequence. |
||
The overall consensus is that HRM is a cost efficient method that can be employed as an initial screen for samples suspected of harbouring polymorphisms or mutations. This would reduce the number of samples which need to be investigated further using more conventional methods. |
The overall consensus is that HRM is a cost efficient method that can be employed as an initial screen for samples suspected of harbouring polymorphisms or mutations. This would reduce the number of samples which need to be investigated further using more conventional methods. |
||
'''Zygosity testing'''<br /> |
'''Zygosity testing'''<br /> |
||
Currently there are many methods used to determine the [[zygosity]] status of a gene at a particular locus. These methods include the use of PCR with specifically designed probes to detect the variants of the genes (SNP typing is the simplest case). In cases where longer stretches of variation is implicated post PCR analysis of the amplicons may be required. Changes in enzyme restriction, electrophoretic and chromatographic profiles can be measured. These methods are usually more time consuming and increase the risk of amplicon contamination in the laboratory, due to the need to work with high concentrations of amplicons in the lab post-PCR. The use of HRM reduces the time required for analysis and the risk of contamination. HRM is a more cost effective solution and the high resolution element not only allows the determination of [[Homozygous#Homozygous|homo]] and [[Homozygous#Heterozygous|heterozygosity]], it also resolves information about the type of homo and heterozygosity, with different gene variants giving rise to differing melt curve shapes. A study by Gundry et al. 2003 <ref |
Currently there are many methods used to determine the [[zygosity]] status of a gene at a particular locus. These methods include the use of PCR with specifically designed probes to detect the variants of the genes (SNP typing is the simplest case). In cases where longer stretches of variation is implicated post PCR analysis of the amplicons may be required. Changes in enzyme restriction, electrophoretic and chromatographic profiles can be measured. These methods are usually more time consuming and increase the risk of amplicon contamination in the laboratory, due to the need to work with high concentrations of amplicons in the lab post-PCR. The use of HRM reduces the time required for analysis and the risk of contamination. HRM is a more cost effective solution and the high resolution element not only allows the determination of [[Homozygous#Homozygous|homo]] and [[Homozygous#Heterozygous|heterozygosity]], it also resolves information about the type of homo and heterozygosity, with different gene variants giving rise to differing melt curve shapes. A study by Gundry et al. 2003 <ref name=Gundry03/>, showed that [[fluorescent labelling]] of one primer (in the pair) has been shown to be favourable over using an intercalating dye such as [[SYBR Green|SYBR green I]]. However, progress has been made in the development and use of improved intercalating dyes <ref>{{cite journal |author=Wittwer CT, Reed GH, Gundry CN, Vandersteen JG, Pryor RJ |title=High-resolution genotyping by amplicon melting analysis using LCGreen |journal=Clin. Chem. |volume=49 |issue=6 Pt 1 |pages=853–60 |year=2003 |month=June |pmid=12765979 |url=http://www.clinchem.org/cgi/content/full/49/6/853}}</ref> which reduce the issue of PCR inhibition and concerns over non-saturating intercalation of the dye. |
||
'''Epigenetics'''<br /> |
'''Epigenetics'''<br /> |
||
The HRM methodology has also been exploited to provide a reliable analysis of the [[DNA methylation|methylation]] status of DNA. This is of significance since changes to the methylation status of tumour suppressor genes, genes that regulate [[apoptosis]] and DNA repair, are characteristics of cancers and also have implications for responses to chemotherapy. For example, cancer patients can be more sensitive to treatment with [[Alkylating antineoplastic agent|DNA alkylating agents]] if the promoter of the DNA repair gene [[O-6-methylguanine-DNA methyltransferase|MGMT]] of the patient is methylated. In a study which tested the methylation status of the MGMT promoter on 19 colorectal samples, 8 samples were found to be methylated <ref> |
The HRM methodology has also been exploited to provide a reliable analysis of the [[DNA methylation|methylation]] status of DNA. This is of significance since changes to the methylation status of tumour suppressor genes, genes that regulate [[apoptosis]] and DNA repair, are characteristics of cancers and also have implications for responses to chemotherapy. For example, cancer patients can be more sensitive to treatment with [[Alkylating antineoplastic agent|DNA alkylating agents]] if the promoter of the DNA repair gene [[O-6-methylguanine-DNA methyltransferase|MGMT]] of the patient is methylated. In a study which tested the methylation status of the MGMT promoter on 19 colorectal samples, 8 samples were found to be methylated <ref>{{cite journal |author=Wojdacz TK, Dobrovic A |title=Methylation-sensitive high resolution melting (MS-HRM): a new approach for sensitive and high-throughput assessment of methylation |journal=Nucleic Acids Res. |volume=35 |issue=6 |pages=e41 |year=2007 |pmid=17289753 |pmc=1874596 |doi=10.1093/nar/gkm013 |url=http://nar.oxfordjournals.org/cgi/content/full/35/6/e41}}</ref>. |
||
Methylated DNA can be treated by bi-sulphite modification, which converts non-methylated [[Deamination#Cytosine|cytosines to uracil]]. Therefore, PCR products resulting from a template that was originally unmethylated will have a lower melting point than those derived from a methylated template. HRM also offers the possibility of determining the proportion of methylation in a given sample, by comparing it to a standard curve which is generated by mixing different ratios of methylated and non-methylated DNA together. This can offer information regarding the degree of methylation that a tumour may have and thus give an indication of the character of the tumour and how far it deviates from what is “normal”. |
Methylated DNA can be treated by bi-sulphite modification, which converts non-methylated [[Deamination#Cytosine|cytosines to uracil]]. Therefore, PCR products resulting from a template that was originally unmethylated will have a lower melting point than those derived from a methylated template. HRM also offers the possibility of determining the proportion of methylation in a given sample, by comparing it to a standard curve which is generated by mixing different ratios of methylated and non-methylated DNA together. This can offer information regarding the degree of methylation that a tumour may have and thus give an indication of the character of the tumour and how far it deviates from what is “normal”. |
||
HRM also is practically advantageous for use in diagnostics, due to its capacity to be adapted to high throughput screening testing, and again it minimises the possibility of amplicon spread and contamination within a laboratory, owing to its closed-tube format. |
HRM also is practically advantageous for use in diagnostics, due to its capacity to be adapted to high throughput screening testing, and again it minimises the possibility of amplicon spread and contamination within a laboratory, owing to its closed-tube format. |
||
'''Intercalating dyes'''<br /> |
'''Intercalating dyes'''<br /> |
||
To follow the transition of dsDNA (double-stranded) to ssDNA (single-stranded), intercalating dyes are employed. These dyes show differential fluorescence emission dependent on their association with double-stranded or single-stranded DNA. [[SYBR Green|SYBR Green I]] is a first generation dye for HRM. It fluoresces when intercalated into dsDNA and not ssDNA. Because it may inhibit PCR at high concentrations, it is used at sub-saturating concentrations. Recently, some researchers have discouraged the use of SYBR Green I for HRM <ref name= |
To follow the transition of dsDNA (double-stranded) to ssDNA (single-stranded), intercalating dyes are employed. These dyes show differential fluorescence emission dependent on their association with double-stranded or single-stranded DNA. [[SYBR Green|SYBR Green I]] is a first generation dye for HRM. It fluoresces when intercalated into dsDNA and not ssDNA. Because it may inhibit PCR at high concentrations, it is used at sub-saturating concentrations. Recently, some researchers have discouraged the use of SYBR Green I for HRM <ref name=Reed07>{{cite journal |author=Reed GH, Kent JO, Wittwer CT |title=High-resolution DNA melting analysis for simple and efficient molecular diagnostics |journal=Pharmacogenomics |volume=8 |issue=6 |pages=597–608 |year=2007 |month=June |pmid=17559349 |doi=10.2217/14622416.8.6.597 |url=http://www.futuremedicine.com/doi/abs/10.2217/14622416.8.6.597?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%3dncbi.nlm.nih.gov}} [http://www.gene-quantification.de/reed-hrm-2007.pdf as PDF]</ref>, claiming that substantial protocol modifications are required. This is because it is suggested that the lack of accuracy may result from “dye jumping”, where dye from a melted duplex may get reincorporated into regions of dsDNA which had not yet melted <ref name=Gundry03>{{cite journal |author=Gundry CN, Vandersteen JG, Reed GH, Pryor RJ, Chen J, Wittwer CT |title=Amplicon melting analysis with labeled primers: a closed-tube method for differentiating homozygotes and heterozygotes |journal=Clin. Chem. |volume=49 |issue=3 |pages=396–406 |year=2003 |month=March |pmid=12600951 |url=http://www.clinchem.org/cgi/pmidlookup?view=long&pmid=12600951}}</ref><ref name=Reed07/>. New saturating dyes such as LC Green and LC Green Plus , ResoLight, EvaGreen and SYTO 9 are available on the market and have been used successfully for HRM. However, some groups have successfully used SYBR Green I for HRM with the Corbett Rotorgene instruments <ref>{{cite journal |author=Pornprasert S, Phusua A, Suanta S, Saetung R, Sanguansermsri T |title=Detection of alpha-thalassemia-1 Southeast Asian type using real-time gap-PCR with SYBR Green1 and high resolution melting analysis |journal=Eur. J. Haematol. |volume=80 |issue=6 |pages=510–4 |year=2008 |month=June |pmid=18284625 |doi=10.1111/j.1600-0609.2008.01055.x |url=http://www3.interscience.wiley.com/resolve/openurl?genre=article&sid=nlm:pubmed&issn=0902-4441&date=2008&volume=80&issue=6&spage=510}}</ref> and advocate the use of SYBR Green I for HRM applications. |
||
==References== |
==References== |
||
{{reflist}} |
|||
<references /> |
|||
==Further information== |
==Further information== |
||
Revision as of 01:17, 5 April 2010
| Template:Wikify is deprecated. Please use a more specific cleanup template as listed in the documentation. |
This article is written like a personal reflection, personal essay, or argumentative essay that states a Wikipedia editor's personal feelings or presents an original argument about a topic. (January 2009) |
High Resolution Melting (HRM) analysis
In molecular biology High Resolution Melt or HRM analysis as it will be referred to herein is a powerful technique for the detection of mutations, polymorphisms and epigenetic differences in double stranded DNA samples. It was discovered and developed by Idaho Technology and the University of Utah[1]. It has advantages over other genotyping technologies. Namely:
- It is cost effective vs. other genotyping technologies such as sequencing and Taqman SNP typing. This makes it ideal for large scale genotyping projects.
- It is fast and powerful thus able to accurately genotype many samples rapidly.
- It is simple. With a good quality HRM assay, powerful genotyping can be performed by non-geneticists in any laboratory with access to an HRM capable real-time PCR machine.
How does high resolution melting analysis work?
HRM analysis is performed on double stranded DNA samples. Typically the user will use polymerase chain reaction (PCR) prior to HRM analysis to amplify the DNA region in which their mutation of interest lies. Essentially the PCR process turns a tiny amount of your region of DNA of interest in to a large amount, so you have quantities large enough for better analysis. In the tube there are now many of copies of your region of DNA of interest. This region that is amplified is known as the amplicon. After the PCR process the HRM analysis begins. The process is simply a precise warming of the amplicon DNA from around 50˚C up to around 95˚C. At some point during this process, the melting temperature of the amplicon is reached and the two strands of DNA separate or “melt” apart.
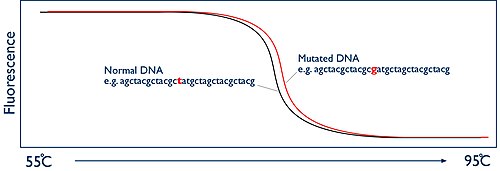
The secret of HRM is to monitor this process happening in real-time. This is achieved by using a fluorescent dye. The dyes that are used for HRM are known as intercalating dyes and have a unique property. They bind specifically to double-stranded DNA and when they are bound they fluoresce brightly. In the absence of double stranded DNA they have nothing to bind to and they only fluoresce at a low level. At the beginning of the HRM analysis there is a high level of fluorescence in the sample because of the billions of copies of the amplicon. But as the sample is heated up and the two strands of the DNA melt apart, presence of double stranded DNA decreases and thus fluorescence is reduced. The HRM machine has a camera that watches this process by measuring the fluorescence. The machine then simply plots this data as a graph known as a melt curve, showing the level of fluorescence vs the temperature:
Spot the difference The melting temperature of the amplicon at which the two DNA strands come apart is entirely predictable. It is dependant on the sequence of the DNA bases. If you are comparing two samples from two different people, they should give exactly the same shaped melt curve. However if one of the people has a mutation in the DNA region you have amplified, then this will alter the temperature at which the DNA strands melt apart. So now the two melt curves appear different. The difference may only be tiny, perhaps a fraction of a degree, but because the HRM machine has the ability to monitor this process in “high resolution”, it is possible to accurately document these changes and therefore identify if a mutation is present or not.
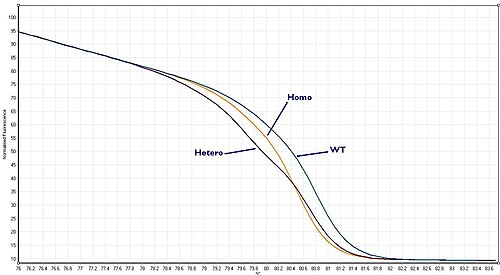
Wild type, heterozygote or homozygote? Things become slightly more complicated than this because organisms contain two copies of each gene, known as the two alleles. So, if a sample is taken from a patient and amplified using PCR both copies of the region of DNA (alleles) of interest are amplified. So if we are looking for mutation there are now three possibilities:
- Neither allele contains a mutation
- One or other allele contains a mutation
- Both alleles contain a mutation.
These three scenarios are known as “Wild –type”, “Heterozygote” or “homozygote” respectively. Each gives a melt curve that is slightly different. With a high quality HRM assay it is possible to distinguish between all three of these scenarios.
Applications of HRM
SNP typing/Point mutation detection
Conventional SNP typing methods are typically time consuming and expensive, requiring several probe bases assays to be multiplexed together or the use of DNA microarrays. HRM is more cost effective and reduces the need to design multiple pairs of primers and the need to purchase expensive probes. The HRM method has been successfully used to detect a single G to A substitution in the gene Vssc (Voltage Sensitive Sodium Channel) which confers resistance to the acaricide permethrin in Scabies mite. This mutation results in a coding change in the protein (G1535D). The analysis of scabies mites collected from suspected permethrin susceptible and tolerant populations by HRM showed distinct melting profiles. The amplicons from the sensitive mites were observed to have a higher melting temperature relative to the tolerant mites, as expected from the higher thermostability of the GC base pair [2]
In a field more relevant to clinical diagnostics, HRM has been shown to be suitable in principle for the detection of mutations in the breast cancer susceptibility genes BRCA1 and BRCA2. More than 400 mutations have been identified in these genes.
The sequencing of genes is the gold standard for identifying mutations. Sequencing is time consuming and labour intensive and is often preceded by techniques used to identify heteroduplex DNA, which then further amplify these issues. HRM offers a faster and more convenient closed-tube method of assessing the presence of mutations and gives a result which can be further investigated if it is of interest. In a study carried out by Scott et al. in 2006 [3], 3 cell lines harbouring different BRCA mutations were used to assess the HRM methodology. It was found that the melting profiles of the resulting PCR products could be used to distinguish the presence or absence of a mutation in the amplicon. Similarly in 2007 Krypuy et al. [4]. showed that the careful design of HRM assays (with regards to primer placement) could be successfully employed to detect mutations in the TP53 gene, which encodes the tumour suppressor protein p53 in clinical samples of breast and ovarian cancer. Both these studies highlighted that fact that changes in the melting profile can be in the form of a shift in the melting temperature or an obvious difference in the shape of the melt curve. Both of these parameters are a function of the amplicon sequence.
The overall consensus is that HRM is a cost efficient method that can be employed as an initial screen for samples suspected of harbouring polymorphisms or mutations. This would reduce the number of samples which need to be investigated further using more conventional methods.
Zygosity testing
Currently there are many methods used to determine the zygosity status of a gene at a particular locus. These methods include the use of PCR with specifically designed probes to detect the variants of the genes (SNP typing is the simplest case). In cases where longer stretches of variation is implicated post PCR analysis of the amplicons may be required. Changes in enzyme restriction, electrophoretic and chromatographic profiles can be measured. These methods are usually more time consuming and increase the risk of amplicon contamination in the laboratory, due to the need to work with high concentrations of amplicons in the lab post-PCR. The use of HRM reduces the time required for analysis and the risk of contamination. HRM is a more cost effective solution and the high resolution element not only allows the determination of homo and heterozygosity, it also resolves information about the type of homo and heterozygosity, with different gene variants giving rise to differing melt curve shapes. A study by Gundry et al. 2003 [5], showed that fluorescent labelling of one primer (in the pair) has been shown to be favourable over using an intercalating dye such as SYBR green I. However, progress has been made in the development and use of improved intercalating dyes [6] which reduce the issue of PCR inhibition and concerns over non-saturating intercalation of the dye.
Epigenetics
The HRM methodology has also been exploited to provide a reliable analysis of the methylation status of DNA. This is of significance since changes to the methylation status of tumour suppressor genes, genes that regulate apoptosis and DNA repair, are characteristics of cancers and also have implications for responses to chemotherapy. For example, cancer patients can be more sensitive to treatment with DNA alkylating agents if the promoter of the DNA repair gene MGMT of the patient is methylated. In a study which tested the methylation status of the MGMT promoter on 19 colorectal samples, 8 samples were found to be methylated [7].
Methylated DNA can be treated by bi-sulphite modification, which converts non-methylated cytosines to uracil. Therefore, PCR products resulting from a template that was originally unmethylated will have a lower melting point than those derived from a methylated template. HRM also offers the possibility of determining the proportion of methylation in a given sample, by comparing it to a standard curve which is generated by mixing different ratios of methylated and non-methylated DNA together. This can offer information regarding the degree of methylation that a tumour may have and thus give an indication of the character of the tumour and how far it deviates from what is “normal”.
HRM also is practically advantageous for use in diagnostics, due to its capacity to be adapted to high throughput screening testing, and again it minimises the possibility of amplicon spread and contamination within a laboratory, owing to its closed-tube format.
Intercalating dyes
To follow the transition of dsDNA (double-stranded) to ssDNA (single-stranded), intercalating dyes are employed. These dyes show differential fluorescence emission dependent on their association with double-stranded or single-stranded DNA. SYBR Green I is a first generation dye for HRM. It fluoresces when intercalated into dsDNA and not ssDNA. Because it may inhibit PCR at high concentrations, it is used at sub-saturating concentrations. Recently, some researchers have discouraged the use of SYBR Green I for HRM [8], claiming that substantial protocol modifications are required. This is because it is suggested that the lack of accuracy may result from “dye jumping”, where dye from a melted duplex may get reincorporated into regions of dsDNA which had not yet melted [5][8]. New saturating dyes such as LC Green and LC Green Plus , ResoLight, EvaGreen and SYTO 9 are available on the market and have been used successfully for HRM. However, some groups have successfully used SYBR Green I for HRM with the Corbett Rotorgene instruments [9] and advocate the use of SYBR Green I for HRM applications.
References
- ^ for academic treatment of the history of HRM see http://www.dna.utah.edu/Hi-Res/TOP_Hi-Res%20Melting.html
- ^ Pasay C, Arlian L, Morgan M; et al. (2008). "High-resolution melt analysis for the detection of a mutation associated with permethrin resistance in a population of scabies mites". Med. Vet. Entomol. 22 (1): 82–8. doi:10.1111/j.1365-2915.2008.00716.x. PMID 18380658.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ James PA, Doherty R, Harris M; et al. (2006). "Optimal selection of individuals for BRCA mutation testing: a comparison of available methods". J. Clin. Oncol. 24 (4): 707–15. doi:10.1200/JCO.2005.01.9737. PMID 16446345.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Krypuy M, Ahmed AA, Etemadmoghadam D; et al. (2007). "High resolution melting for mutation scanning of TP53 exons 5-8". BMC Cancer. 7: 168. doi:10.1186/1471-2407-7-168. PMC 2025602. PMID 17764544.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ a b Gundry CN, Vandersteen JG, Reed GH, Pryor RJ, Chen J, Wittwer CT (2003). "Amplicon melting analysis with labeled primers: a closed-tube method for differentiating homozygotes and heterozygotes". Clin. Chem. 49 (3): 396–406. PMID 12600951.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Wittwer CT, Reed GH, Gundry CN, Vandersteen JG, Pryor RJ (2003). "High-resolution genotyping by amplicon melting analysis using LCGreen". Clin. Chem. 49 (6 Pt 1): 853–60. PMID 12765979.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Wojdacz TK, Dobrovic A (2007). "Methylation-sensitive high resolution melting (MS-HRM): a new approach for sensitive and high-throughput assessment of methylation". Nucleic Acids Res. 35 (6): e41. doi:10.1093/nar/gkm013. PMC 1874596. PMID 17289753.
- ^ a b Reed GH, Kent JO, Wittwer CT (2007). "High-resolution DNA melting analysis for simple and efficient molecular diagnostics". Pharmacogenomics. 8 (6): 597–608. doi:10.2217/14622416.8.6.597. PMID 17559349.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) as PDF - ^ Pornprasert S, Phusua A, Suanta S, Saetung R, Sanguansermsri T (2008). "Detection of alpha-thalassemia-1 Southeast Asian type using real-time gap-PCR with SYBR Green1 and high resolution melting analysis". Eur. J. Haematol. 80 (6): 510–4. doi:10.1111/j.1600-0609.2008.01055.x. PMID 18284625.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link)
Further information
High Resolution Melting analysis information at gene-quantification.info
University of Southampton spinout company specialising in High Resolution Melting analysis