(S)-Ipsdienol: Difference between revisions
Content deleted Content added
←Created page with '{{Chembox | ImageFile = S-Ipsdienol.svg | ImageSize = | ImageAlt = | IUPACName = (4S)-2-methyl-6-methylideneocta-2,7-dien-4-ol | OtherNames = (S)-(+)-Ipsdien...' |
(No difference)
|
Revision as of 02:41, 15 February 2013
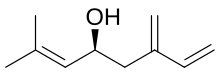
| |
| Names | |
|---|---|
| IUPAC name
(4S)-2-methyl-6-methylideneocta-2,7-dien-4-ol
| |
| Other names
(S)-(+)-Ipsdienol, 2-Methyl-6-methylene-2,7-octadiene-4-ol, Ipsdienol
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.128.974 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C10H16O | |
| Molar mass | 152.23344 |
| Appearance | Colorless |
| Density | 0.87 g/mL (Predicted) |
| Melting point | -25.02 °C (Predicted) |
| Boiling point | 233.59 °C at 760 mmHg (Predicted) |
| Hazards | |
| Flash point | 87 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
S-Ipsdienol is a terpene alcohol, it is one of the major aggregation pheromones of the bark beetle. It was first identified from Ips Confusus, in which it is believed to be a principle sex attractant[1]
It is suggested that the compound plays a role in interspecies communication between Ips Latidens and Ips Ini, falicitiating reductions in competition for breeding material and/or mating interference.[2]
Synthesis
The compound has been synthesized from D-mannitol[3]
References
- ^ Silverstein, Robert M (1966). "Sex Attractants in Frass Produced by Male Ips confusus in Ponderosa Pine". Science. 154 (3748): 509–510. doi:10.1126/science.154.3748.509.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Miller, Daniel R. (1 August 1991). "Ipsenol: an aggregation pheromone forIps latidens (Leconte) (Coleoptera: Scolytidae)". Journal of Chemical Ecology. 17 (8): 1517–1527. doi:10.1007/BF00984685.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Hanessian, Stephen (1983). Total Synthesis of Natural Products: The 'Chiron' Approach. Pergamon press. p. 63. ISBN 0-08-0292427-X.
{{cite book}}: Check|isbn=value: length (help)
