Chorismic acid

| |
| Names | |
|---|---|
| IUPAC name
(3R,4R)-3-[(1-carboxyvinyl)oxy]-4-hydroxycyclohexa-1,5-diene-1-carboxylic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.164.204 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H10O6 | |
| Molar mass | 226.18 g/mol |
| Melting point | 140 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Chorismic acid, more commonly known as its anionic form chorismate, is an important biochemical intermediate in plants and microorganisms. It is a precursor for:
- The aromatic amino acids phenylalanine and tyrosine
- Indole, indole derivatives and tryptophan
- 2,3-dihydroxybenzoic acid (DHB) used for enterobactin biosynthesis
- The plant hormone salicylic acid[1]
- Many alkaloids and other aromatic metabolites.
The name chorismic acid derives from a classical greek word, χωρίζω meaning "to separate",[2] because the compound plays a role as a branch-point in aromatic amino acid biosynthesis.[3]
Biosynthesis
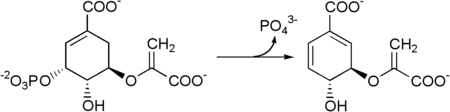
Shikimate → shikimate-3-phosphate → 5-enolpyruvylshikimate-3-phosphate → chorismate.
External links
References
- ^ Wildermuth MC, Dewdney J, Wu G, Ausubel FM (2001). "Isochorismate synthase is required to synthesize salicylic acid for plant defence". Nature. 414 (6863): 562–5. doi:10.1038/35107108. PMID 11734859.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ . ISBN 0-19-864226-1.
{{cite book}}: Cite uses deprecated parameter|authors=(help); Missing or empty|title=(help); Unknown parameter|name=ignored (help) - ^ Attention: This template ({{cite doi}}) is deprecated. To cite the publication identified by doi:10.1016/S0968-0004(98)01330-9, please use {{cite journal}} (if it was published in a bona fide academic journal, otherwise {{cite report}} with
|doi=10.1016/S0968-0004(98)01330-9instead.