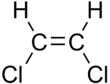
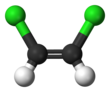
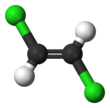
1,2-Dichloroethene
A request that this article title be changed to 1,2-Dichloroethylene is under discussion. Please do not move this article until the discussion is closed. |
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,2-Dichloroethene | |||
| Other names
1,2-Dichloroethylene
1,2-DCE Acetylene dichloride sym-Dichloroethylene | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.007.956 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| UNII |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H2Cl2 | |||
| Molar mass | 96.95 g/mol | ||
| Appearance | colorless liquid | ||
| Odor | sweet[1] | ||
| Density | Z: 1.28 g/cm3 E: 1.26 g/cm3 | ||
| Melting point | Z: −81.47 °C E: −49.44 °C | ||
| Boiling point | Z: 60.2 °C E: 48.5 °C | ||
| |||
| Z: 1.9 D E: 0 D | |||
| Hazards | |||
| Flash point | 2–4 °C; 36–39 °F; 275–277 K | ||
| Explosive limits | 5.6–12.8%[2] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
770 mg/kg (oral, rat) 1275 mg/kg (oral, rat, trans-isomer)[3] | ||
LC50 (median concentration)
|
21,273 ppm (mouse, 6 hr, trans-isomer)[3] | ||
LCLo (lowest published)
|
16,000 ppm (rat, 6 hr, cis-isomer)[3] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 200 ppm (790 mg/m3)[2] | ||
REL (Recommended)
|
TWA 200 ppm (790 mg/m3)[2] | ||
IDLH (Immediate danger)
|
1000 ppm[2] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
1,2-Dichloroethene, commonly called 1,2-dichloroethylene or 1,2-DCE, is the name for a pair of organochlorine compounds with the molecular formula C2H2Cl2. They are both colorless liquids with a sweet odor. It can exist as either of two geometric isomers, cis-1,2-dichloroethene or trans-1,2-dichloroethene, but is often used as a mixture of the two. They have modest solubility in water. These compounds have some applications as a degreasing solvent.[1] In contrast to most cis-trans compounds, the Z isomer (cis) is more stable than the E isomer (trans) by 0.4 kcal/mol.[4]
Production, uses and reactions[edit]
cis-DCE, the Z isomer, is obtainable by the controlled chlorination of acetylene:
- C2H2 + Cl2 → C2H2Cl2
Industrially both isomers arise as byproducts of the production of vinyl chloride, which is produced on a vast scale. Unlike 1,1-dichloroethylene, the 1,2-dichloroethylene isomers do not polymerize.[1]
trans-1,2-DCE has applications including electronics cleaning, precision cleaning, and certain metal cleaning applications.[5]
Both isomers participate in Kumada coupling reactions. trans-1,2-Dichloroethylene participates in cycloaddition reactions.[6]
Safety and environmental concerns[edit]
These compounds have "moderate oral toxicity to rats".[1]
The dichloroethylene isomers occur in some polluted waters and soils, as the decomposition products of trichloroethylene. Significant attention has been paid to their further degradation, e.g. by iron particles.[7][8]
See also[edit]
- 1,1-Dichloroethene
- 1,2-Dichloroethane, which is also often abbreviated as 1,2-DCE
References[edit]
- ^ a b c d E.-L. Dreher; T. R. Torkelson; K. K. Beutel (2011). "Chlorethanes and Chloroethylenes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.o06_o01. ISBN 978-3527306732.
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0195". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c "1,2-Dichloroethylene". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Pitzer, Kenneth S.; Hollenberg, J. L. (1954). "cis- and trans-Dichloroethylenes. The Infrared Spectra from 130–400 Cm.–1 and the Thermodynamic Properties". J. Am. Chem. Soc. 76 (6): 1493–1496. doi:10.1021/ja01635a010.
- ^ "Chlorinated Solvents and Feed Stock - Axiall". Archived from the original on 2016-04-08. Retrieved 2016-03-23.
- ^ Wang, Xiao Min; Hou, Xuelong; Zhou, Zhongyuan; Mak, Thomas C. W.; Wong, Henry N. C. (1993). "Arene synthesis by extrusion reaction. 16. Coplanar and stable derivatives of 13,14-didehydro-tribenzo[a,c,e]cyclooctene: Synthesis of 5,6-didehydro-1,1,14,14-tetramethyl-10,11-methano-1H-benzo[5,6]cycloocta[1,2,3,4-def]fluorene and 5,6-didehydro-10,11-methano-1H-benzo[5,6]cycloocta[1,2,3,4-def]fluorene-1,14-dione and x-ray crystal structures of 1,1,14,14-tetramethyl-10,11-methano-1H-benzo[5,6-cycloocta[1,2,3,4-def]fluorene and 1,12-dihydro-1,1,12,12-tetramethyldicyclopenta[def,JKL]tetraphenylene". The Journal of Organic Chemistry. 58 (26): 7498–7506. doi:10.1021/jo00078a031.
- ^ Mattes, Timothy E.; Alexander, Anne K.; Coleman, Nicholas V. (2010). "Aerobic biodegradation of the chloroethenes: Pathways, enzymes, ecology, and evolution". FEMS Microbiology Reviews. 34 (4): 445–475. doi:10.1111/j.1574-6976.2010.00210.x. PMID 20146755.
- ^ Schrick, Bettina; Blough, Jennifer L.; Jones, A. Daniel; Mallouk, Thomas E. (2002). "Hydrodechlorination of Trichloroethylene to Hydrocarbons Using Bimetallic Nickel−Iron Nanoparticles". Chemistry of Materials. 14 (12): 5140–5147. doi:10.1021/cm020737i.
External links[edit]
- International Chemical Safety Card 0436
- NIOSH Pocket Guide to Chemical Hazards. "#0195". National Institute for Occupational Safety and Health (NIOSH).