Deferasirox: Difference between revisions
Content deleted Content added
Doctorfluffy (talk | contribs) m unref |
citing from excellent recent review (and open-access, too! |
||
| Line 21: | Line 21: | ||
| routes_of_administration = Oral |
| routes_of_administration = Oral |
||
}} |
}} |
||
'''Deferasirox''' (marketed as '''Exjade |
'''Deferasirox''' (marketed as '''Exjade''') is a [[Drug design#Rational drug design|rationally-designed]]<ref name=Choudhry>{{cite journal |author=Choudhry VP, Naithani R |title=Current status of iron overload and chelation with deferasirox |journal=Indian J Pediatr |volume=74 |issue=8 |pages=759–64 |year=2007 |pmid=17785900 |doi=}} [http://www.ijppediatricsindia.org/text.asp?2007/74/8/759/34579 Free full text]</ref> oral [[iron]] [[Chelation therapy|chelator]]. Its main use is to reduce [[Iron overload disorder|chronic iron overload]] in patients who are receiving long-term [[blood transfusion]]s for conditions such as beta-[[thalassemia]] and other chronic [[anemia]]s.<ref name=Choudhry/> It is the first oral medication approved in the USA for this purpose.<ref name=FDA>{{cite press release | url = http://www.fda.gov/bbs/topics/news/2005/NEW01258.html | title = FDA Approves First Oral Drug for Chronic Iron Overload | date = [[November 9]] [[2005]] | accessdate = 2007-10-31 | publisher = United States [[Food and Drug Administration]]}}</ref> |
||
It was approved by the [[United States]] [[Food and Drug Administration]] (FDA) in November 2005. |
It was approved by the [[United States]] [[Food and Drug Administration]] (FDA) in November 2005.<ref name=Choudhry/><ref name=FDA/> |
||
==References== |
|||
{{Reflist}} |
|||
{{pharma-stub}} |
{{pharma-stub}} |
||
Revision as of 02:58, 31 October 2007
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 70% |
| Protein binding | 99% |
| Metabolism | Hepatic glucuronidation |
| Elimination half-life | 8 to 16 hours |
| Excretion | Fecal (84%) and renal (8%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.211.077 |
| Chemical and physical data | |
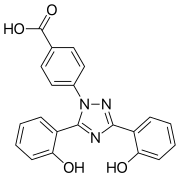
| Formula | C21H15N3O4 |
| Molar mass | 373.362 g/mol g·mol−1 |
Deferasirox (marketed as Exjade) is a rationally-designed[2] oral iron chelator. Its main use is to reduce chronic iron overload in patients who are receiving long-term blood transfusions for conditions such as beta-thalassemia and other chronic anemias.[2] It is the first oral medication approved in the USA for this purpose.[3]
It was approved by the United States Food and Drug Administration (FDA) in November 2005.[2][3]
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ a b c Choudhry VP, Naithani R (2007). "Current status of iron overload and chelation with deferasirox". Indian J Pediatr. 74 (8): 759–64. PMID 17785900. Free full text
- ^ a b "FDA Approves First Oral Drug for Chronic Iron Overload" (Press release). United States Food and Drug Administration. November 9 2005. Retrieved 2007-10-31.
{{cite press release}}: Check date values in:|date=(help)
