Methoxymethanol: Difference between revisions
Citation bot (talk | contribs) Alter: url. Add: pmid, s2cid. | You can use this bot yourself. Report bugs here. | Suggested by Graeme Bartlett | via #UCB_toolbar |
|||
| Line 30: | Line 30: | ||
}} |
}} |
||
}} |
}} |
||
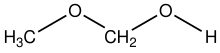
'''Methoxymethanol''' is a chemical compound which is both an [[ether]] and an [[alcohol]], a [[hemiacetal|hemiformal]].<ref name=mai>{{cite journal |last1=Maiwald |first1=Michael |last2=Fischer |first2=Holger H. |last3=Ott |first3=Michael |last4=Peschla |first4=Roger |last5=Kuhnert |first5=Christian |last6=Kreiter |first6=Cornelius G. |last7=Maurer |first7=Gerd |last8=Hasse |first8=Hans |title=Quantitative NMR Spectroscopy of Complex Liquid Mixtures: Methods and Results for Chemical Equilibria in Formaldehyde−Water−Methanol at Temperatures up to 383 K |journal=Industrial & Engineering Chemistry Research |date=January 2003 |volume=42 |issue=2 |pages=259–266 |doi=10.1021/ie0203072}}</ref> The structural formula can be written as CH<sub>3</sub>OCH<sub>2</sub>OH. It has been discovered in space.<ref>{{cite journal |last1=McGuire |first1=Brett A. |last2=Shingledecker |first2=Christopher N. |last3=Willis |first3=Eric R. |last4=Burkhardt |first4=Andrew M. |last5=El-Abd |first5=Samer |last6=Motiyenko |first6=Roman A. |last7=Brogan |first7=Crystal L. |last8=Hunter |first8=Todd R. |last9=Margulès |first9=Laurent |last10=Guillemin |first10=Jean-Claude |last11=Garrod |first11=Robin T. |last12=Herbst |first12=Eric |last13=Remijan |first13=Anthony J. |title=ALMA Detection of Interstellar Methoxymethanol (CH3OCH2OH) |journal=The Astrophysical Journal |date=2017 |volume=851 |issue=2 |pages=L46 |doi=10.3847/2041-8213/aaa0c3 |language=en|bibcode=2017ApJ...851L..46M |arxiv=1712.03256 }}</ref> |
'''Methoxymethanol''' is a chemical compound which is both an [[ether]] and an [[alcohol]], a [[hemiacetal|hemiformal]].<ref name=mai>{{cite journal |last1=Maiwald |first1=Michael |last2=Fischer |first2=Holger H. |last3=Ott |first3=Michael |last4=Peschla |first4=Roger |last5=Kuhnert |first5=Christian |last6=Kreiter |first6=Cornelius G. |last7=Maurer |first7=Gerd |last8=Hasse |first8=Hans |title=Quantitative NMR Spectroscopy of Complex Liquid Mixtures: Methods and Results for Chemical Equilibria in Formaldehyde−Water−Methanol at Temperatures up to 383 K |journal=Industrial & Engineering Chemistry Research |date=January 2003 |volume=42 |issue=2 |pages=259–266 |doi=10.1021/ie0203072}}</ref> The structural formula can be written as CH<sub>3</sub>OCH<sub>2</sub>OH. It has been discovered in space.<ref>{{cite journal |last1=McGuire |first1=Brett A. |last2=Shingledecker |first2=Christopher N. |last3=Willis |first3=Eric R. |last4=Burkhardt |first4=Andrew M. |last5=El-Abd |first5=Samer |last6=Motiyenko |first6=Roman A. |last7=Brogan |first7=Crystal L. |last8=Hunter |first8=Todd R. |last9=Margulès |first9=Laurent |last10=Guillemin |first10=Jean-Claude |last11=Garrod |first11=Robin T. |last12=Herbst |first12=Eric |last13=Remijan |first13=Anthony J. |title=ALMA Detection of Interstellar Methoxymethanol (CH3OCH2OH) |journal=The Astrophysical Journal |date=2017 |volume=851 |issue=2 |pages=L46 |doi=10.3847/2041-8213/aaa0c3 |language=en|bibcode=2017ApJ...851L..46M |arxiv=1712.03256 |s2cid=119211919 }}</ref> |
||
==Formation== |
==Formation== |
||
Methoxymethanol forms spontaneously when a water solution of [[formaldehyde]] and [[methanol]] are mixed.<ref name=hays>{{cite journal |last1=Hays |first1=Brian M. |last2=Widicus Weaver |first2=Susanna L. |title=Theoretical Examination of O(<sup>1</sup>D) Insertion Reactions to Form Methanediol, Methoxymethanol, and Aminomethanol |journal=The Journal of Physical Chemistry A |date=6 May 2013 |volume=117 |issue=32 |pages=7142–7148 |doi=10.1021/jp400753r|bibcode=2013JPCA..117.7142H }}</ref><ref name=mai/> or when formaldehyde is bubbled through methanol.<ref>{{cite journal |last1=Celik |first1=Fuat E. |last2=Lawrence |first2=Henry |last3=Bell |first3=Alexis T. |title=Synthesis of precursors to ethylene glycol from formaldehyde and methyl formate catalyzed by heteropoly acids |journal=Journal of Molecular Catalysis A: Chemical |date=June 2008 |volume=288 |issue=1–2 |pages=87–96 |doi=10.1016/j.molcata.2008.03.029}}</ref> |
Methoxymethanol forms spontaneously when a water solution of [[formaldehyde]] and [[methanol]] are mixed.<ref name=hays>{{cite journal |last1=Hays |first1=Brian M. |last2=Widicus Weaver |first2=Susanna L. |title=Theoretical Examination of O(<sup>1</sup>D) Insertion Reactions to Form Methanediol, Methoxymethanol, and Aminomethanol |journal=The Journal of Physical Chemistry A |date=6 May 2013 |volume=117 |issue=32 |pages=7142–7148 |doi=10.1021/jp400753r|pmid=23646865 |bibcode=2013JPCA..117.7142H }}</ref><ref name=mai/> or when formaldehyde is bubbled through methanol.<ref>{{cite journal |last1=Celik |first1=Fuat E. |last2=Lawrence |first2=Henry |last3=Bell |first3=Alexis T. |title=Synthesis of precursors to ethylene glycol from formaldehyde and methyl formate catalyzed by heteropoly acids |journal=Journal of Molecular Catalysis A: Chemical |date=June 2008 |volume=288 |issue=1–2 |pages=87–96 |doi=10.1016/j.molcata.2008.03.029}}</ref> |
||
In space methoxymethanol can form when methanol radicals (CH<sub>2</sub>OH or CH<sub>3</sub>O) react. These are radiolysis products derived when ultraviolet light or cosmic rays hit frozen methanol.<ref name=hays/> |
In space methoxymethanol can form when methanol radicals (CH<sub>2</sub>OH or CH<sub>3</sub>O) react. These are radiolysis products derived when ultraviolet light or cosmic rays hit frozen methanol.<ref name=hays/> |
||
Methanol can react with [[carbon dioxide]] and hydrogen at 80°C and some pressure with a ruthenium or cobalt catalyst, to yield some methoxymethanol.<ref>{{cite book |last1=Dixneuf |first1=Pierre H. |last2=Soulé |first2=Jean-François |title=Organometallics for Green Catalysis |date=2019 |publisher=Springer |isbn=9783030109554 |pages=69–70 |url=https://books.google.com |
Methanol can react with [[carbon dioxide]] and hydrogen at 80°C and some pressure with a ruthenium or cobalt catalyst, to yield some methoxymethanol.<ref>{{cite book |last1=Dixneuf |first1=Pierre H. |last2=Soulé |first2=Jean-François |title=Organometallics for Green Catalysis |date=2019 |publisher=Springer |isbn=9783030109554 |pages=69–70 |url=https://books.google.com/books?id=IB2KDwAAQBAJ&pg=PA70 |language=en}}</ref> |
||
==Properties== |
==Properties== |
||
Revision as of 12:21, 26 August 2020
| |
| Names | |
|---|---|
| Preferred IUPAC name
Methoxymethanol | |
| Other names
Formaldehyde methyl hemiacetal
| |
| Identifiers | |
3D model (JSmol)
|
|
| 1900186 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.022.476 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H6O2 | |
| Molar mass | 62.068 g·mol−1 |
| Density | 0.948 |
| Hazards | |
| GHS labelling: | |
  
| |
| Warning | |
| H226, H302, H371 | |
| P210, P233, P240, P241, P242, P243, P260, P264, P270, P280, P301+P312, P303+P361+P353, P309+P311, P330, P370+P378, P403+P235, P405, P501 | |
| Flash point | 39.9 °C (103.8 °F; 313.0 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Methoxymethanol is a chemical compound which is both an ether and an alcohol, a hemiformal.[1] The structural formula can be written as CH3OCH2OH. It has been discovered in space.[2]
Formation
Methoxymethanol forms spontaneously when a water solution of formaldehyde and methanol are mixed.[3][1] or when formaldehyde is bubbled through methanol.[4]
In space methoxymethanol can form when methanol radicals (CH2OH or CH3O) react. These are radiolysis products derived when ultraviolet light or cosmic rays hit frozen methanol.[3]
Methanol can react with carbon dioxide and hydrogen at 80°C and some pressure with a ruthenium or cobalt catalyst, to yield some methoxymethanol.[5]
Properties
Different conformations of the molecule are Gauche-gauce (Gg), Gauche-gauce' (Gg'), and Trans-gauche (Tg).[6]
References
- ^ a b Maiwald, Michael; Fischer, Holger H.; Ott, Michael; Peschla, Roger; Kuhnert, Christian; Kreiter, Cornelius G.; Maurer, Gerd; Hasse, Hans (January 2003). "Quantitative NMR Spectroscopy of Complex Liquid Mixtures: Methods and Results for Chemical Equilibria in Formaldehyde−Water−Methanol at Temperatures up to 383 K". Industrial & Engineering Chemistry Research. 42 (2): 259–266. doi:10.1021/ie0203072.
- ^ McGuire, Brett A.; Shingledecker, Christopher N.; Willis, Eric R.; Burkhardt, Andrew M.; El-Abd, Samer; Motiyenko, Roman A.; Brogan, Crystal L.; Hunter, Todd R.; Margulès, Laurent; Guillemin, Jean-Claude; Garrod, Robin T.; Herbst, Eric; Remijan, Anthony J. (2017). "ALMA Detection of Interstellar Methoxymethanol (CH3OCH2OH)". The Astrophysical Journal. 851 (2): L46. arXiv:1712.03256. Bibcode:2017ApJ...851L..46M. doi:10.3847/2041-8213/aaa0c3. S2CID 119211919.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b Hays, Brian M.; Widicus Weaver, Susanna L. (6 May 2013). "Theoretical Examination of O(1D) Insertion Reactions to Form Methanediol, Methoxymethanol, and Aminomethanol". The Journal of Physical Chemistry A. 117 (32): 7142–7148. Bibcode:2013JPCA..117.7142H. doi:10.1021/jp400753r. PMID 23646865.
- ^ Celik, Fuat E.; Lawrence, Henry; Bell, Alexis T. (June 2008). "Synthesis of precursors to ethylene glycol from formaldehyde and methyl formate catalyzed by heteropoly acids". Journal of Molecular Catalysis A: Chemical. 288 (1–2): 87–96. doi:10.1016/j.molcata.2008.03.029.
- ^ Dixneuf, Pierre H.; Soulé, Jean-François (2019). Organometallics for Green Catalysis. Springer. pp. 69–70. ISBN 9783030109554.
- ^ Motiyenko, R. A. (21 June 2016). "Millimeter-wave spectroscopy of methoxymethanol" (Document). hdl:2142/91121.
{{cite document}}: Cite document requires|publisher=(help)
