Gentiobiose
| |
| Names | |
|---|---|
| IUPAC name
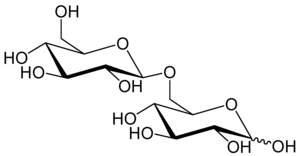
6-O-β-D-glucopyranosyl-D-glucose
| |
| Other names
amygdalose
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H22O11 | |
| Molar mass | 342.30 g/mol |
| Melting point | 190–195 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Gentiobiose is a disaccharide composed of two units of D-glucose joined with a β(1->6) linkage. It is a white crystalline solid that is soluble in water or hot methanol. Gentiobiose is incorporated into the chemical structure of crocin, the chemical compound that gives saffron its color. It is a product of the caramelization of glucose. [2]
References
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (11th ed.), Merck, 1989, p. 4288, ISBN 091191028X
- ^ Sugisawa, Hirqshi; Edo, Hiroshi (1966). "The Thermal Degradation of Sugars I. Thermal Polymerization of Glucose". Journal of Food Science. 31 (4): 561. doi:10.1111/j.1365-2621.1966.tb01905.x.
