Methoxetamine: Difference between revisions
No edit summary |
removed advertising |
||
| Line 23: | Line 23: | ||
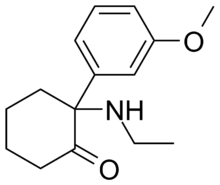
'''Methoxetamine''' or 3-MeO-2-Oxo-PCE is a chemical of the [[arylcyclohexylamine]] class. It is an [[chemical analog|analogue]] of [[ketamine]] that also contains structural features of [[eticyclidine]] and [[3-MeO-PCP]]. Like ketamine, it is thought to behave as a [[NMDA receptor antagonist]] and [[dopamine reuptake inhibitor]], though it has not been formally profiled pharmacologically. Anecdotal reports suggest a longer duration than ketamine.<ref name="Methoxetamine">[http://www.bluelight.ru/vb/showpost.php?p=9110438&postcount=18], knockando. (14 February 2011). "Methoxetamine". Bluelight.ru. Retrieved 2011-02-14.</ref> Methoxetamine differs from many other dissociative anesthetics of the arylcyclohexylamine class in that it was designed for [[grey-market]] distribution. Methoxetamine is a product of rational [[drug design]]: its N-ethyl group was chosen to increase potency, lessing the risk of [[Interstitial cystitis]] that can result from the accumulation of ketamine-like metabolites in the urinary bladder.<ref name="Interview with a ketamine chemist">[http://www.viceland.com/int/v18n2/htdocs/interview-with-ketamine-chemist-704.php?page=1], Morris, H. (11 February 2011). "Interview with a ketamine chemist: or to be more precise, an arylcyclohexylamine chemist". Vice Magazine. Retrieved 2011-02-11.</ref> |
'''Methoxetamine''' or 3-MeO-2-Oxo-PCE is a chemical of the [[arylcyclohexylamine]] class. It is an [[chemical analog|analogue]] of [[ketamine]] that also contains structural features of [[eticyclidine]] and [[3-MeO-PCP]]. Like ketamine, it is thought to behave as a [[NMDA receptor antagonist]] and [[dopamine reuptake inhibitor]], though it has not been formally profiled pharmacologically. Anecdotal reports suggest a longer duration than ketamine.<ref name="Methoxetamine">[http://www.bluelight.ru/vb/showpost.php?p=9110438&postcount=18], knockando. (14 February 2011). "Methoxetamine". Bluelight.ru. Retrieved 2011-02-14.</ref> Methoxetamine differs from many other dissociative anesthetics of the arylcyclohexylamine class in that it was designed for [[grey-market]] distribution. Methoxetamine is a product of rational [[drug design]]: its N-ethyl group was chosen to increase potency, lessing the risk of [[Interstitial cystitis]] that can result from the accumulation of ketamine-like metabolites in the urinary bladder.<ref name="Interview with a ketamine chemist">[http://www.viceland.com/int/v18n2/htdocs/interview-with-ketamine-chemist-704.php?page=1], Morris, H. (11 February 2011). "Interview with a ketamine chemist: or to be more precise, an arylcyclohexylamine chemist". Vice Magazine. Retrieved 2011-02-11.</ref> |
||
However it is worth noting that it only has a structural similarities to Ketamine and Methoxetamine is a totally different substance. This is a research chemical. Drug users should be aware that this compound is geared towards the scientific community. It is also trademarked stating "Methoxetamine is a legal Ketamine alternative for the laboratory research."<ref name="Methoxetamine™">[www.methoxetamine.co.uk/]</ref> |
|||
==See also== |
==See also== |
||
Revision as of 07:03, 8 March 2011
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C15H21NO2 |
| Molar mass | 231.3327 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| (verify) | |
Methoxetamine or 3-MeO-2-Oxo-PCE is a chemical of the arylcyclohexylamine class. It is an analogue of ketamine that also contains structural features of eticyclidine and 3-MeO-PCP. Like ketamine, it is thought to behave as a NMDA receptor antagonist and dopamine reuptake inhibitor, though it has not been formally profiled pharmacologically. Anecdotal reports suggest a longer duration than ketamine.[1] Methoxetamine differs from many other dissociative anesthetics of the arylcyclohexylamine class in that it was designed for grey-market distribution. Methoxetamine is a product of rational drug design: its N-ethyl group was chosen to increase potency, lessing the risk of Interstitial cystitis that can result from the accumulation of ketamine-like metabolites in the urinary bladder.[2]
