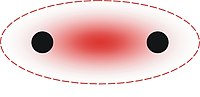
Sigma bond

In chemistry, sigma bonds (σ bonds) are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals. Sigma bonding is most clearly defined for diatomic molecules using the language and tools of symmetry groups. In this formal approach, a σ-bond is symmetrical with respect to rotation about the bond axis. By this definition, common forms of sigma bonds are s+s, pz+pz, s+pz and dz2+dz2 (where z is defined as the axis of the bond). Quantum theory also indicates that molecular orbitals (MO) of identical symmetry actually mix. As a practical consequence of this mixing of diatomic molecules, the wavefunctions s+s and pz+pz molecular orbitals become blended. The extent of this mixing (or blending) depends on the relative energies of the like-symmetry MO's.
For homodiatomics, bonding σ orbitals have no nodal planes between the bonded atoms. The corresponding antibonding, or σ* orbital, is defined by the presence of a nodal plane between these two bonded atoms.
Sigma bonds are the strongest type of covalent bonds, and the electrons in these bonds are sometimes referred to as sigma electrons.
The symbol σ is the Greek letter sigma (s). When viewed down the bond axis, a σ MO resembles an s atomic orbital.

Sigma bonds in polyatomic compounds
They are obtained by head on overlapping of atomic shells. The concept of sigma bonding is extended, albeit loosely, to describe bonding interactions involving overlap of a single lobe of one orbital with a single lobe of another. For example, propane is described as consisting of ten sigma bonds, one each for the two C-C bonds and one each for the eight C-H bonds. The σ bonding in such a polyatomic molecule is highly delocalized, which conflicts with the two-orbital, one-bond concept. Despite this complication, the concept of σ bonding is extremely powerful and therefore pervasive.
Sigma bonds in multiple-bonded species
Compounds that feature multiple bonds, such as ethylene and chromium(II) acetate, have sigma bonds between the multiple bonded atoms. These sigma bonds are supplemented by π-bonds, e.g. in the case of ethylene, and even δ-bonds, e.g. in the case of chromium(II) acetate.
Sigma bonds in organic molecules
Organic molecules are often made up of one cyclic compound or more, such as benzene, and are often made up of many sigma bonds along with pi bonds. According to the sigma bond rule, the number of sigma bonds in a molecule is equivalent to the number of atoms plus the number of rings minus one.
- Nb σ = Nb atoms + Nb rings - 1
This can easily be concluded by realizing that the creation of bonds between atoms that are not connected in a ring requires the same number of atoms minus one (such as in hydrogen gas, H2, where there is only one sigma bond, or ammonia, NH3, where there are only 3 sigma bonds), and that rings do not obey this rule (such as benzene rings, which have 6 sigma bonds within the ring for 6 carbon atoms).