Grotthuss mechanism
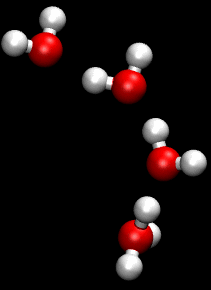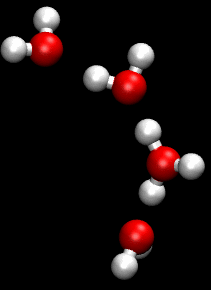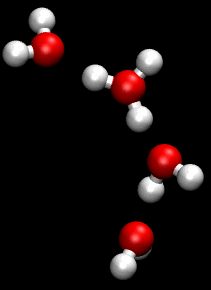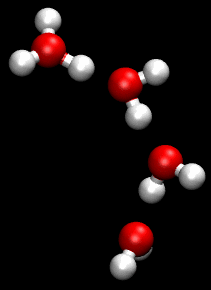
The Grotthuss mechanism is the mechanism by which an 'excess' proton or protonic defect diffuses through the hydrogen bond network of water molecules or other hydrogen-bonded liquids through the formation or cleavage of covalent bonds.
In his 1806 publication “Theory of decomposition of liquids by electrical currents”, Theodor Grotthuss proposed a theory of water conductivity.[1] Grotthuss envisioned the electrolytic reaction as a sort of ‘bucket line’ where each oxygen atom simultaneously passes and receives a single hydrogen atom. It was an astonishing theory to propose at the time,[according to whom?] since the water molecule was thought to be OH not H2O and the existence of ions was not fully understood. On its 200th anniversary, his article was reviewed by Cukierman.[2]
Although Grotthuss was using an incorrect empirical formula of water, his description of the passing of protons through the cooperation of neighboring water molecules proved to be remarkably prescient.[according to whom?]
Proton transport mechanism and proton-hopping mechanism
The Grotthuss mechanism is now a general name for the proton-hopping mechanism. In liquid water the solvation of the excess proton is idealized by two forms: the H9O4+ (Eigen cation) or H5O2+ (Zundel cation). While the transport mechanism is believed to involve the inter-conversion between these two solvation structures, the details of the hopping and transport mechanism is still debated. Currently there are two plausible mechanisms:
- Eigen to Zundel to Eigen (E–Z–E), on the basis of experimental NMR data,[3]
- Zundel to Zundel (Z–Z), on the basis of molecular dynamics simulation.
The calculated energetics of the hydronium solvation shells were reported in 2007 and it was suggested that the activation energies of the two proposed mechanisms do not agree with their calculated hydrogen bond strengths, but mechanism 1 might be the better candidate of the two.[4]
Addition: By use of conditional and time-dependent radial distribution functions (RDF), it was shown that the hydronium RDF can be decomposed into contributions from two distinct structures, Eigen and Zundel. The first peak in g(r)[clarification needed] of the Eigen structure is similar to the equilibrium, standard RDF, only slightly more ordered, while the first peak of the Zundel structure is actually split into two peaks. The actual proton transfer event was then traced (after synchronizing all PT events so that t=0 is the actual event time), revealing that the hydronium indeed starts from an Eigen state, and quickly transforms into the Zundel state as the proton is being transferred, with the first peak of g(r) splitting into two.[5]
The anomalous diffusion of protons
The Grotthuss mechanism, along with the relative lightness and small size of the proton, explains the unusually high diffusion rate of the proton relative to that of other common cations (Table 1), which is due simply to random thermal motion, i.e. Brownian motion. Quantum tunnelling becomes more probable the smaller the mass of the cation is, and the proton is the lightest possible stable cation. Thus there is a minor effect from quantum tunnelling also, although it dominates at low temperatures only.
| Cation | Mobility / cm2 V−1 s−1 |
| NH4+ | 0.763×10−3 |
| Na+ | 0.519×10−3 |
| K+ | 0.762×10−3 |
| H+ | 3.62×10−3 |
References
- ^ de Grotthuss, C.J.T. (1806). "Sur la décomposition de l'eau et des corps qu'elle tient en dissolution à l'aide de l'électricité galvanique". Ann. Chim. 58: 54–73.
- ^ Cukierman, Samuel (2006). "Et tu Grotthuss!". Biochimica et Biophysica Acta. 1757 (8): 876–8. doi:10.1016/j.bbabio.2005.12.001. PMID 16414007.
- ^ Agmon, Noam (1995). "The Grotthuss mechanism". Chem. Phys. Lett. 244 (5–6): 456–462. Bibcode:1995CPL...244..456A. doi:10.1016/0009-2614(95)00905-J.
- ^ Markovitch, Omer; Agmon, Noam (2007). "Structure and energetics of the hydronium hydration shells". J. Phys. Chem. A. 111 (12): 2253–6. doi:10.1021/jp068960g. PMID 17388314.
- ^ Markovitch, Omer; et al. (2008). "Special Pair Dance and Partner Selection: Elementary Steps in Proton Transport in Liquid Water". J. Phys. Chem. B. 112 (31): 9456–9466. doi:10.1021/jp804018y. PMID 18630857.
{{cite journal}}: Explicit use of et al. in:|author2=(help)
