JNJ-26990990
(Redirected from JNJ26990990)
 | |
| Clinical data | |
|---|---|
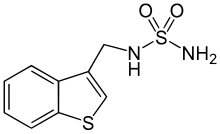
| Other names | N-((Benzo[b]thien-3-yl)methyl)sulfamide |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C9H10N2O2S2 |
| Molar mass | 242.31 g·mol−1 |
| | |
JNJ-26990990 is a broad-spectrum anticonvulsant drug currently under development by Janssen Pharmaceutica as a second-generation follow-up to the marketed drug topiramate.[1][2] It was designed to have the same anticonvulsant effects as topiramate, but without the side effects associated with topiramate's carbonic anhydrase inhibition.[1] It also has potential use in the treatment of inflammatory pain, neuropathic pain, and depression.[3]
JNJ-26990990 entered phase II clinical trials in October 2007.[4]
Metabolites and radioactive isotope-labeled derivatives of JNJ-26990990 have also been prepared to support its development.[5]
See also[edit]
References[edit]
- ^ a b Parker MH, Smith-Swintosky VL, McComsey DF, Huang Y, Brenneman D, Klein B, et al. (December 2009). "Novel, broad-spectrum anticonvulsants containing a sulfamide group: advancement of N-((benzo[b]thien-3-yl)methyl)sulfamide (JNJ-26990990) into human clinical studies". Journal of Medicinal Chemistry. 52 (23): 7528–7536. doi:10.1021/jm801432r. PMID 19388676.
- ^ WO patent 2006023861, Parker MH, Reitz AB, Maryanoff BE, "Preparation of benzo-fused heteroaryl sulfamide derivatives for the treatment of epilepsy and related disorders", issued 2006-03-02, assigned to Janssen Pharmaceutica
- ^ "JNJ 26990990 Johnson & Johnson developing anticonvulsant Johnson & Johnson preclinical data". R & D Focus Drug News. September 22, 2008. Archived from the original on October 12, 2012.
- ^ Fischer J, Ganellin CR (2010). "Chapter 2. Standalone Drugs". Analogue-Based Drug Discovery II. doi:10.1002/9783527630035.ch2.
- ^ Lin R, Weaner LE, Hoerr DC, Salter R, Gong Y (January 2013). "Expeditious syntheses of stable and radioactive isotope-labeled anticonvulsant agent, JNJ-26990990, and its metabolites". Journal of Labelled Compounds & Radiopharmaceuticals. 56 (1): 22–26. doi:10.1002/jlcr.3013. PMID 24285137.
