Skraup reaction
| Skraup reaction | |
|---|---|
| Named after | Zdenko Hans Skraup |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000062 |
The Skraup synthesis is a chemical reaction used to synthesize quinolines. It is named after the Czech chemist Zdenko Hans Skraup (1850-1910). In the archetypal Skraup reaction, aniline is heated with sulfuric acid, glycerol, and an oxidizing agent such as nitrobenzene to yield quinoline.[1][2][3][4]

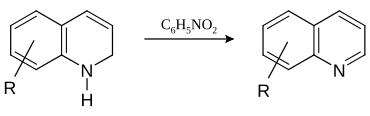
In this example, nitrobenzene serves as both the solvent and the oxidizing agent. The reaction, which otherwise has a reputation for being violent, is typically conducted in the presence of ferrous sulfate.[5] Arsenic acid may be used instead of nitrobenzene and the former is better since the reaction is less violent.[6]



See also
References
- ^ Skraup, Z. H. (1880). "Eine Synthese des Chinolins". Berichte. 13: 2086.
- ^ Manske, R. H. F. (1942). "The Chemistry of Quinolines". Chem. Rev. 30: 113. doi:10.1021/cr60095a006.
- ^ Manske, Richard H. F.; Kulka, Marshall (1953). "The Skraup Synthesis of Quinolines". Org. React. 7: 80–99. doi:10.1002/0471264180.or007.02.
- ^ Wahren, M. (1964). "Stabilisotop markierte verbindungen—II , Untersuchung der skraupschen chinolin-synthese mit hilfe von 15N". Tetrahedron. 20 (12): 2773. doi:10.1016/S0040-4020(01)98495-9.
- ^ Clarke, H. T.; Davis, A. W. (1941). "Quinoline". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 1, p. 478. - ^ Finar, Ivor Lionel (1973). Organic Chemistry, Volume 1 (6th ed.). p. 857. ISBN 978-0582442214.
