Bacteriorhodopsin
| Bacteriorhodopsin | |||||||
|---|---|---|---|---|---|---|---|
 Halobacterium salinarum bacteriorhodopsin homotrimer viewed from the cytoplasm. Bound retinal shown in black. PDB: 6RQP | |||||||
| Identifiers | |||||||
| Organism | |||||||
| Symbol | bop | ||||||
| UniProt | P02945 | ||||||
| |||||||
Bacteriorhodopsin (Bop) is a protein used by Archaea, most notably by haloarchaea, a class of the Euryarchaeota.[1] It acts as a proton pump; that is, it captures light energy and uses it to move protons across the membrane out of the cell.[2] The resulting proton gradient is subsequently converted into chemical energy.[3]
Function
[edit]Bacteriorhodopsin is a light-driven H+ ion transporter found in some haloarchaea, most notably Halobacterium salinarum (formerly known as syn. H. halobium). The proton-motive force generated by the protein is used by ATP synthase to generate adenosine triphosphate (ATP). By expressing bacteriorhodopsin, the archaea cells are able to synthesise ATP in the absence of a carbon source.[4][5]
Structure
[edit]
Bacteriorhodopsin is a 27 kDa integral membrane protein usually found in two-dimensional crystalline patches known as "purple membrane", which can occupy almost 50% of the surface area of the archaeal cell. The repeating element of the hexagonal lattice is composed of three identical protein chains, each rotated by 120 degrees relative to the others.[6] Each monomer has seven transmembrane alpha helices and an extracellular-facing, two-stranded beta sheet.[7][8]
Bacteriorhodopsin is synthesized as a protein precursor, known as bacterio-opsin, which is extensively modified after translation.[9][10] The modifications are:
- Covalent conjugation of a retinal molecule to residue Lys216, via a Schiff base, to create the retinylidene chromophore.[11]
- Cleavage of the signal peptide, the first 13 amino acids at the N-terminus, and the conversion of residue Gln14 to pyroglutamate[12]
- Removal of residue Asp262 at the C-terminus[12]
Spectral properties
[edit]Bacteriorhodopsin molecule is purple and is most efficient at absorbing green light (in the wavelength range 500-650 nm). In the native membrane, the protein has a maximum absorbance at 553 nm, however addition of detergent disrupts the trimeric form, leading a loss of exciton coupling between the chromophores, and the monomeric form consequently has an absorption maximum of 568 nm.[13][14]
Bacteriorhodopsin has a broad excitation spectrum. For a detection wavelength between 700 and 800 nm, it has an appreciable detected emission for excitation wavelengths between 470 nm and 650 nm (with a peak at 570 nm).[15] When pumped at 633 nm, the emission spectrum has appreciable intensity between 650 nm and 850 nm.[16]
Mechanism
[edit]Photocycle overview
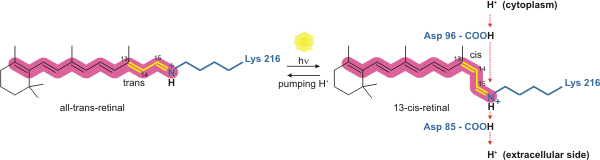
[edit]Bacteriorhodopsin is a light-driven proton pump. It is the retinal molecule that changes its isomerization state from all-trans to 13-cis when it absorbs a photon. The surrounding protein responds to the change in the chromophore shape, by undergoing an ordered sequence of conformational changes (collectively known as the photocycle).[17] The conformational changes alter the pKa values of conserved amino acids in the core of the protein, including Asp85, Asp96 and the Schiff base N atom (Lys216). These sequential changes in acid dissociation constant, result in the transfer of one proton from the intracellular side to the extracellular side of the membrane for each photon absorbed by the chromophore.
The bacteriorhodopsin photocycle consists of nine distinct stages, starting from the ground or resting state, which is denoted 'bR'. The intermediates are identified by single letters and may be distinguished by their absorption spectra.[18] The nine stages are:
- bR + photon → K ⇌ L ⇌ M1 ⇌ M2 ⇌ M2' ⇌ N ⇌ N' ⇌ O ⇌ bR[18]
Ground state + photon → K state → L state
[edit]
Bacteriorhodopsin in the ground state absorbs a photon and the retinal changes isomerization from all-trans 15-anti to the strained 13-cis 15-anti in the K state. The isomerisation reaction is fast and occurs in less than 1 ps. The retinal adopts a less strained conformation to form the L intermediate.
L state → M1 state
[edit]Asp85 accepts a proton from the Schiff base N atom. In the M1 intermediate, neither the Schiff base nor Asp85 are charged.
M1 state → M2 state
[edit]The Schiff base rotates away from the extracellular side of the protein towards the cytoplasmic side, in preparation to accept a new proton.
M2 state → M2' state
[edit]A proton is released from Glu204 and Glu194 to the extracellular medium.
M2' state → N state
[edit]The retinal Schiff base accepts a proton from Asp96. In the N state, both Asp96 and the Schiff base are charged.
N state → N' state
[edit]Asp96 accepts a proton from the cytoplasmic side of the membrane and becomes uncharged.
N' state → O state
[edit]Retinal reisomerizes to the all-trans state.
O state → ground state
[edit]Asp85 transfers a proton to Glu194 and Glu204[19][20] on the extracellular face of the protein.
Homologs and other similar proteins
[edit]Bacteriorhodopsin belongs to the microbial rhodopsin family. Its homologs include the archaerhodopsins,[21] the light-driven chloride pump halorhodopsin (for which the crystal structure is also known), and some directly light-activated channels such as channelrhodopsin.
Bacteriorhodopsin is similar to vertebrate rhodopsins, the pigments that sense light in the retina. Rhodopsins also contain retinal; however, the functions of rhodopsin and bacteriorhodopsin are different, and there is limited similarity in their amino acid sequences. Both rhodopsin and bacteriorhodopsin belong to the 7TM receptor family of proteins, but rhodopsin is a G protein-coupled receptor and bacteriorhodopsin is not. In the first use of electron crystallography to obtain an atomic-level protein structure, the structure of bacteriorhodopsin was resolved in 1990.[22] It was then used as a template to build models of G protein-coupled receptors before crystallographic structures were also available for these proteins. It has been excessively studied on both mica[23][24] and glass substrates using Atomic force microscopy and Femtosecond crystallography.[25]
All other phototrophic systems in bacteria, algae, and plants use chlorophylls or bacteriochlorophylls rather than bacteriorhodopsin. These also produce a proton gradient, but in a quite different and more indirect way involving an electron transfer chain consisting of several other proteins. Furthermore, chlorophylls are aided in capturing light energy by other pigments known as "antennas"; these are not present in bacteriorhodopsin-based systems. It is possible that phototrophy independently evolved at least twice, once in bacteria and once in archaea.
Gallery
[edit]-
Bacteriorhodopsin single monomer with retinal molecule between 7 vertical alpha helixes (PDB ID: 1X0S [26][27][28]). One more small helix is light blue, beta sheet yellow.
See also
[edit]Literature
[edit]- ^ Schoch CL, Ciufo S, Domrachev M, Hotton CL, Kannan S, Khovanskaya R, Leipe D, McVeigh R, O'Neill K, Robbertse B, Sharma S, Soussov V, Sullivan JP, Sun L, Turner S, Karsch-Mizrachi (2020). "Halobacteria". NCBI Taxonomy: a comprehensive update on curation, resources and tools. National Center for Biotechnology Information. Retrieved 31 March 2021.
- ^ Voet, Judith G.; Voet, Donald (2004). Biochemistry. New York: J. Wiley & Sons. ISBN 978-0-471-19350-0.
- ^ "Bacteriorhodopsin: Pumping Ions".
- ^ Nicholls DG; Ferguson SJ (1992). Bioenergetics 2 (2nd ed.). San Diego: Academic Press. ISBN 9780125181242.
- ^ Stryer, Lubert (1995). Biochemistry (fourth ed.). New York - Basingstoke: WH Freeman and Company. ISBN 978-0716720096.
- ^ Essen LO, Siegert R, Lehman WD, Oesterhelt D (1998). "Lipid patches in membrane protein oligomers: Crystal structure of the bacteriorhodopsin-lipid complex". Proceedings of the National Academy of Sciences of the United States of America. 95 (20): 11673–11678. Bibcode:1998PNAS...9511673E. doi:10.1073/pnas.95.20.11673. PMC 21699. PMID 9751724.
- ^ Pebay-Peroua E, Rummel G, Rosenbusch JP, Landau EM (1997). "X-ray structure of bacteriorhodopsin at 2.5 Å from microcrystals grown in lipidic cubic phases". Science. 277 (5332): 1676–1681. doi:10.1126/science.277.5332.1676. PMID 9287223.
- ^ Luecke H, Schobert B, Richter HT, Cartailler JP, Lanyi JK (1999). "Structure of bacteriorhodopsin at 1.55 Å resolution". Journal of Molecular Biology. 291 (4): 899–911. doi:10.1006/jmbi.1999.3027. PMID 10452895.
- ^ Oesterhelt, Dieter; Stoeckenius, Walther (1971). "Rhodopsin-like Protein from the Purple Membrane of Halobacterium halobium". Nature New Biology. 233 (39): 149–152. doi:10.1038/newbio233149a0. PMID 4940442.
- ^ Oesterhelt, Dieter (1982). "[3] Reconstitution of the retinal proteins bacteriorhodopsin and halorhodopsin". Reconstitution of the retinal proteins bacteriorhodopsin and halorhodopsin. Methods in Enzymology. Vol. 88. pp. 10–17. doi:10.1016/0076-6879(82)88006-3. ISBN 9780121819880.
- ^ Bayley H, Huang KS, Radhakrishnan R, Ross AH, Takagaki Y, Khorana HG (1981). "Site of attachment of retinal in bacteriorhodopsin". Proceedings of the National Academy of Sciences of the United States of America. 78 (4): 2225–2229. Bibcode:1981PNAS...78.2225B. doi:10.1073/pnas.78.4.2225. PMC 319317. PMID 6941281.
- ^ a b Hoi KK, Bada Juarez JF, Judge PJ, Yen HY, Wu D, Vinals J, Taylor GF, Watts A, Robinson CV (2021). "Detergent-free Lipodisq Nanoparticles Facilitate High-Resolution Mass Spectrometry of Folded Integral Membrane Proteins". Nano Letters. 21 (7): 2824–2831. Bibcode:2021NanoL..21.2824H. doi:10.1021/acs.nanolett.0c04911. PMC 8050825. PMID 33787280.
- ^ Wang J, Link S, Heyes CD, El-Sayed MA (2002). "Comparison of the Dynamics of the Primary Events of Bacteriorhodopsin in Its Trimeric and Monomeric States". Biophysical Journal. 83 (3): 1557–1566. Bibcode:2002BpJ....83.1557W. doi:10.1016/S0006-3495(02)73925-8. PMC 1302253. PMID 12202380.
- ^ Pescitelli G, Woody RW (2012). "The Exciton Origin of the Visible Circular Dichroism Spectrum of Bacteriorhodopsin". Journal of Physical Chemistry B. 116 (23): 6751–6763. doi:10.1021/jp212166k. PMID 22329810.
- ^ Schenkl, Selma; Zgrablic, Goran; Portuondo-Campa, Erwin; Haacke, Stefan; Chergui, Majed (2007). "On the excitation wavelength dependence of the fluorescence of bacteriorhodopsin". Chemical Physics Letters. 441 (4–6): 322–326. Bibcode:2007CPL...441..322S. doi:10.1016/j.cplett.2007.04.086.
- ^ Ohtani, H.; Tsukamoto, Y.; Sakoda, Y.; Hamaguchi, H. (1995). "Fluorescence spectra of bacteriorhodopsin and the intermediates O and Q at room temperature". FEBS Lett. 359 (1): 65–68. doi:10.1016/0014-5793(94)01440-c. PMID 7851532.
- ^ Hayashi S, Tajkhorshid E, Schulten K (September 2003). "Molecular dynamics simulation of bacteriorhodopsin's photoisomerization using ab initio forces for the excited chromophore". Biophysical Journal. 85 (3): 1440–9. Bibcode:2003BpJ....85.1440H. doi:10.1016/S0006-3495(03)74576-7. PMC 1303320. PMID 12944261.
- ^ a b Ernst OP, Lodowski DT, Elstner M, Hegeman P, Brown LS, Kandori H (2014). "Microbial and animal rhodopsins: Structures, functions and molecular mechanisms". Chemical Reviews. 114 (1): 126–163. doi:10.1021/cr4003769. PMC 3979449. PMID 24364740.
- ^ Dioumaev, A. K.; Richter, H. T.; Brown, L. S.; Tanio, M.; Tuzi, S.; Saito, H.; Kimura, Y.; Needleman, R.; Lanyi, J. K. (1998). "Existence of a proton transfer chain in bacteriorhodopsin: Participation of Glu-194 in the release of protons to the extracellular surface". Biochemistry. 37 (8): 2496–2906. doi:10.1021/bi971842m. PMID 9485398.
- ^ Balashov, S. P.; Lu, M.; Imasheva, E. S.; Govindjee, R.; Ebrey, T. G.; Othersen b, 3rd; Chen, Y.; Crouch, R. K.; Menick, D. R. (1999). "The proton release group of bacteriorhodopsin controls the rate of the final step of its photocycle at low pH". Biochemistry. 38 (7): 2026–2039. doi:10.1021/bi981926a. PMID 10026285.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ Bada Juarez JF, Judge PJ, Adam S, Axford D, Vinals J, Birch J, Kwan TO, Hoi KK, Yen HY, Vial A, Milhiet PE, Robinson CV, Schapiro I, Moraes I, Watts A (2021). "Structures of the archaerhodopsin 3 transporter reveal that disordering of internal water networks underpins receptor sensitization". Nature Communications. 12 (1): 629. Bibcode:2021NatCo..12..629B. doi:10.1038/s41467-020-20596-0. PMC 7840839. PMID 33504778.
- ^ Henderson R, Baldwin JM, Ceska TA, Zemlin F, Beckmann E, Downing KH (1990). "Model for the structure of bacteriorhodopsin based on high-resolution electron cryo-microscopy". J Mol Biol. 213 (4): 899–929. doi:10.1016/S0022-2836(05)80271-2. PMID 2359127.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Müller, Daniel J.; Dufrêne, Yves F. (2008). "Atomic force microscopy as a multifunctional molecular toolbox in nanobiotechnology". Nature Nanotechnology. 3 (5): 261–269. Bibcode:2008NatNa...3..261M. doi:10.1038/nnano.2008.100. ISSN 1748-3387. PMID 18654521.
- ^ Shibata, Mikihiro; Yamashita, Hayato; Uchihashi, Takayuki; Kandori, Hideki; Ando, Toshio (2010-02-14). "High-speed atomic force microscopy shows dynamic molecular processes in photoactivated bacteriorhodopsin". Nature Nanotechnology. 5 (3): 208–212. Bibcode:2010NatNa...5..208S. doi:10.1038/nnano.2010.7. hdl:2297/23872. ISSN 1748-3387. PMID 20154686.
- ^ Nango, Eriko; Royant, Antoine; Kubo, Minoru; Nakane, Takanori; Wickstrand, Cecilia; Kimura, Tetsunari; Tanaka, Tomoyuki; Tono, Kensuke; Song, Changyong (2016-12-23). "A three-dimensional movie of structural changes in bacteriorhodopsin". Science. 354 (6319): 1552–1557. Bibcode:2016Sci...354.1552N. doi:10.1126/science.aah3497. ISSN 0036-8075. PMID 28008064. S2CID 206651572.
- ^ a b Nishikawa, T.; Murakami, M. (2005-03-28). "Crystal structure of the 13-cis isomer of bacteriorhodopsin". RCSB Protein Data Bank (PDB). doi:10.2210/pdb1x0s/pdb. PDB ID: 1X0S. Retrieved 7 October 2012.
{{cite journal}}: Cite journal requires|journal=(help) - ^ a b Nishikawa, T.; Murakami, M. (2005). "Crystal structure of the 13-cis isomer of bacteriorhodopsin in the dark-adapted state". J. Mol. Biol. 352 (2): 319–328. doi:10.1016/j.jmb.2005.07.021. PMID 16084526. PDB ID: 1X0S.
- ^ a b Image created with RasTop (Molecular Visualization Software).
External links
[edit]- Bacteriorhodopsin: Molecule of the Month, by David Goodsell, RCSB Protein Data Bank
- Protein-Based Artificial Retina Manufacturing: Characterization of the Function and Stability of Bacteriorhodopsin Following Exposure to a Microgravity Environment, by Nicole Wagner and Jordan Greco


![Bacteriorhodopsin single monomer with retinal molecule between 7 vertical alpha helixes (PDB ID: 1X0S [26][27][28]). One more small helix is light blue, beta sheet yellow.](http://upload.wikimedia.org/wikipedia/commons/2/21/Bacteriorhodopsin_subunit_1X0S.png)
![Bacteriorhodopsin trimer with one retinal molecule in each subunit seen from the extracellular side EC (PDB ID: 1X0S [26][27][28])](http://upload.wikimedia.org/wikipedia/commons/c/c4/Bacteriorhodopsin_trimer_1X0S.png)