CCK-4
 | |
| Clinical data | |
|---|---|
| Other names | Tetragastrin; Cholecystokinin tetrapeptide |
| Routes of administration | IV |
| Pharmacokinetic data | |
| Bioavailability | 100% |
| Metabolism | plasma protease enzymes |
| Elimination half-life | 13 minutes |
| Excretion | N/A |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C29H35N5O7S |
| Molar mass | 597.69 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
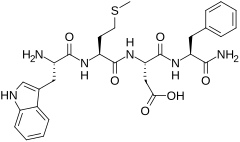
Cholecystokinin tetrapeptide (CCK-4, tetragastrin, Trp-Met-Asp-Phe-NH2) is a peptide fragment derived from the larger peptide hormone cholecystokinin. Unlike cholecystokin which has a variety of roles in the gastrointestinal system as well as central nervous system effects, CCK-4 acts primarily in the brain as an anxiogenic, although it does retain some GI effects, but not as much as CCK-8 or the full length polypeptide CCK-58.
CCK-4 reliably causes severe anxiety symptoms when administered to humans in a dose of as little as 50 μg,[1] and is commonly used in scientific research to induce panic attacks for the purpose of testing new anxiolytic drugs.[2][3][4][5] Since it is a peptide, CCK-4 must be administered by injection, and is rapidly broken down once inside the body so has only a short duration of action,[6] although numerous synthetic analogues with modified properties are known.[7][8][9][10][11][12][13][14][15][16][17]
See also
[edit]References
[edit]- ^ Eser D, di Michele F, Zwanzger P, Pasini A, Baghai TC, Schüle C, et al. (January 2005). "Panic induction with cholecystokinin-tetrapeptide (CCK-4) Increases plasma concentrations of the neuroactive steroid 3alpha, 5alpha tetrahydrodeoxycorticosterone (3alpha, 5alpha-THDOC) in healthy volunteers". Neuropsychopharmacology. 30 (1): 192–5. doi:10.1038/sj.npp.1300572. PMID 15467707.
- ^ Bradwejn J (July 1993). "Neurobiological investigations into the role of cholecystokinin in panic disorder". Journal of Psychiatry & Neuroscience. 18 (4): 178–88. PMC 1188527. PMID 8104032.
- ^ Schunck T, Erb G, Mathis A, Gilles C, Namer IJ, Hode Y, et al. (July 2006). "Functional magnetic resonance imaging characterization of CCK-4-induced panic attack and subsequent anticipatory anxiety". NeuroImage. 31 (3): 1197–208. doi:10.1016/j.neuroimage.2006.01.035. PMID 16600640. S2CID 2430104.
- ^ Eser D, Schüle C, Baghai T, Floesser A, Krebs-Brown A, Enunwa M, et al. (July 2007). "Evaluation of the CCK-4 model as a challenge paradigm in a population of healthy volunteers within a proof-of-concept study". Psychopharmacology. 192 (4): 479–87. doi:10.1007/s00213-007-0738-7. PMID 17318504. S2CID 21457632.
- ^ Eser D, Leicht G, Lutz J, Wenninger S, Kirsch V, Schüle C, et al. (February 2009). "Functional neuroanatomy of CCK-4-induced panic attacks in healthy volunteers". Human Brain Mapping. 30 (2): 511–22. doi:10.1002/hbm.20522. PMC 6870703. PMID 18095276.
- ^ Koulischer D, Moroder L, Deschodt-Lanckman M (August 1982). "Degradation of cholecystokinin octapeptide, related fragments and analogs by human and rat plasma in vitro". Regulatory Peptides. 4 (3): 127–39. doi:10.1016/0167-0115(82)90080-5. PMID 6291099. S2CID 20585962.
- ^ Blommaert AG, Dhôtel H, Ducos B, Durieux C, Goudreau N, Bado A, et al. (February 1997). "Structure-based design of new constrained cyclic agonists of the cholecystokinin CCK-B receptor". Journal of Medicinal Chemistry. 40 (5): 647–58. doi:10.1021/jm9603072. PMID 9057851.
- ^ Bellier B, Million ME, DaNascimento S, Meudal H, Kellou S, Maigret B, Garbay C (October 2000). "Replacement of glycine with dicarbonyl and related moieties in analogues of the C-terminal pentapeptide of cholecystokinin: CCK(2) agonists displaying a novel binding mode". Journal of Medicinal Chemistry. 43 (20): 3614–23. doi:10.1021/jm0000416. PMID 11020275.
- ^ Léna I, Dh tel H, Garbay C, Daugé V (January 2001). "Involvement of D2 dopamine receptors in the opposing effects of two CCK-B agonists in a spatial recognition memory task: role of the anterior nucleus accumbens". Psychopharmacology. 153 (2): 170–9. doi:10.1007/s002130000517. PMID 11205416. S2CID 39490703.
- ^ Bellier B, Garbay C (2003). "How a single inversion of configuration leads to a reversal of the binding mode: proposal of a novel arrangement of CCK2 ligands in their receptor, and contribution to the development of peptidomimetic or non-peptide CCK2 ligands". European Journal of Medicinal Chemistry. 38 (7–8): 671–86. doi:10.1016/S0223-5234(03)00112-0. PMID 12932898.
- ^ Bellier B, Crété D, Million ME, Beslot F, Bado A, Garbay C, Daugé V (November 2004). "New CCK2 agonists confirming the heterogeneity of CCK2 receptors: characterisation of BBL454". Naunyn-Schmiedeberg's Archives of Pharmacology. 370 (5): 404–13. doi:10.1007/s00210-004-0969-7. PMID 15480577. S2CID 20603424.
- ^ Proskuriakova TV, Bespalova Z, Pal'keeva ME, Petrichenko OB, Pankratova NV, Shokhonova VA, Anokhina IP (2005). "[Biological activity of cholecystokinin-(30-33) tetrapeptide analogs]". Bioorganicheskaia Khimiia (in Russian). 31 (2): 130–9. PMID 15889786.
- ^ Anokhina IP, Proskuriakova TV, Bespalova Z, Pal'keeva ME, Shokhonova VA, Petrichenko OB (2006). "[Effect of a cholecystokinin tetrapeptide analogue on opioid reception under acute and chronic morphine administration]". Bioorganicheskaia Khimiia (in Russian). 32 (3): 276–83. doi:10.1134/s106816200603006x. PMID 16808170. S2CID 23993886.
- ^ Agnes RS, Lee YS, Davis P, Ma SW, Badghisi H, Porreca F, et al. (May 2006). "Structure-activity relationships of bifunctional peptides based on overlapping pharmacophores at opioid and cholecystokinin receptors". Journal of Medicinal Chemistry. 49 (10): 2868–75. doi:10.1021/jm050921q. PMC 1484468. PMID 16686530.
- ^ Noble F (2007). "Pharmacology of CCKRs and SAR studies of peptidic analog ligands". Current Topics in Medicinal Chemistry. 7 (12): 1173–9. doi:10.2174/156802607780960447. PMID 17584139.
- ^ García-López MT, González-Muñiz R, Martín-Martínez M, Herranz R (2007). "Strategies for design of non peptide CCK1R agonist/antagonist ligands". Current Topics in Medicinal Chemistry. 7 (12): 1180–94. doi:10.2174/156802607780960537. PMID 17584140.
- ^ Kalindjian SB, McDonald IM (2007). "Strategies for the design of non-peptide CCK2 receptor agonist and antagonist ligand". Current Topics in Medicinal Chemistry. 7 (12): 1195–204. doi:10.2174/156802607780960500. PMID 17584141.
