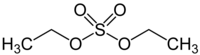
Diethyl sulfate
Appearance
| |

| |
| Names | |
|---|---|
| Other names
Sulfuric acid diethyl ester
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.536 |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10O4S | |
| Molar mass | 154.18 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.2 g/mL |
| Melting point | −25 °C (−13 °F; 248 K) |
| Boiling point | 209 °C (408 °F; 482 K) |
| 0.7 g/100 mL | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 104 °C (219 °F) |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethyl sulfate is a highly toxic and likely carcinogenic[1] chemical compound with formula (Template:Carbon2Template:Hydrogen5)2Template:SulfurTemplate:Oxygen4. It occurs as a colorless liquid with a peppermint odor.
Diethyl sulfate is used as an alkylating agent to prepare ethyl derivatives of phenols, amines, and thiols.
References
Further reading
- Buck, J. R. Park, M.; Wang, Z.; Prudhomme, D. R.; Rizzo, C. J. "9-Ethyl-3,6-Dimethylcarbazole (DMECZ)". Organic Syntheses. 77: 153
{{cite journal}}: CS1 maint: multiple names: authors list (link). - Shammi Theodore and P.S.T. Sai (2001). "Esterification of Ethanol with Sulfuric Acid: A Kinetic Study" (PDF). Canadian Journal of Chemical Engineering. 79: 54. doi:10.1002/cjce.5450790109.

