Beta decay: Difference between revisions
Undid revision 255626841 by 142.162.37.181 (talk) Kurie is correct, see refs |
No edit summary |
||
| Line 86: | Line 86: | ||
In a famous letter written in 1930 [[Wolfgang Pauli]] suggested that in addition to electrons and protons atoms also contained an extremely light neutral particle which he called the neutron. He suggested that this "neutron" was also emitted during beta decay and had simply not yet been observed. In 1931 [[Enrico Fermi]] renamed Pauli's "neutron" to [[neutrino]], and in 1934 Fermi published a very successful [[Fermi's interaction|model of beta decay]] in which neutrinos were produced. |
In a famous letter written in 1930 [[Wolfgang Pauli]] suggested that in addition to electrons and protons atoms also contained an extremely light neutral particle which he called the neutron. He suggested that this "neutron" was also emitted during beta decay and had simply not yet been observed. In 1931 [[Enrico Fermi]] renamed Pauli's "neutron" to [[neutrino]], and in 1934 Fermi published a very successful [[Fermi's interaction|model of beta decay]] in which neutrinos were produced. |
||
There was also a guy named Sir Fracsico Elmo and he played happy time with all the elements. |
|||
==See also== |
==See also== |
||
Revision as of 22:14, 4 December 2008
| Nuclear physics |
|---|
 |
In nuclear physics, beta decay is a type of radioactive decay in which a beta particle (an electron or a positron) is emitted. In the case of electron emission, it is referred to as "beta minus" (
β−
), while in the case of a positron emission as "beta plus" (
β+
). Kinetic energy of beta particles has continuous spectrum ranging from 0 to maximal available energy (Q), which depends on parent and daughter nuclear states participating in the decay. Typical Q is of order of 1 MeV, but it can range from a few keV to a few tens of MeV. The most energetic beta particles are ultrarelativistic, with speeds very close to the speed of light.
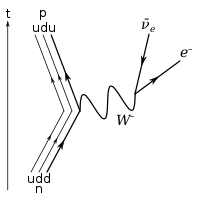
β−
decay

β−
) decay. The intermediate emission of a
W−
boson is omitted.
In
β−
decay, the weak interaction converts a neutron (
n0
) into a proton (
p+
) while emitting an electron (
e−
) and an antineutrino (
ν
e):
At the fundamental level (as depicted in the Feynman diagram below), this is due to the conversion of a down quark to an up quark by emission of a
W−
boson; the
W−
boson subsequently decays into an electron and an antineutrino.
β+
decay
W−
boson
In
β+
decay, energy is used to convert a proton into a neutron, a positron (
e+
) and a neutrino (
ν
e):
So, unlike
β−
,
β+
decay cannot occur in isolation, because it requires energy, the mass of the neutron being greater than the mass of the proton.
β+
decay can only happen inside nuclei when the absolute value of the binding energy of the mother nucleus is lower than that of the daughter nucleus. The difference between these energies goes into the reaction of converting a proton into a neutron, a positron and a neutrino and into the kinetic energy of these particles.
Electron capture (K-capture)
In all the cases where
β+
decay is allowed energetically (and the proton is a part of a nucleus with electron shells), it is accompanied by the electron capture process, when an atomic electron is captured by a nucleus with the emission of a neutrino:
But if the energy difference between initial and final states is low (less than 2mec2), then
β+
decay is not energetically possible, and electron capture is the sole decay mode.
Nuclear transmutation
If the proton and neutron are part of an atomic nucleus, these decay processes transmute one chemical element into another. For example:
137
55Cs
→ 137
56Ba
+
e−
+
ν
e(beta minus decay) 22
11Na
→ 22
10Ne
+
e+
+
ν
e(beta plus decay) 22
11Na
+
e−
→ 22
10Ne
+
ν
e(electron capture)
Beta decay does not change the number of nucleons,A, in the nucleus but changes only its charge, Z. Thus the set of all nuclides with the same A can be introduced; these isobaric nuclides may turn into each other via beta decay. Among them, several nuclides (at least one) are beta stable, because they present local minima of the mass excess: if such a nucleus has (A, Z) numbers, the neighbour nuclei (A, Z−1) and (A, Z+1) have higher mass excess and can beta decay into (A, Z), but not vice versa. A beta-stable nucleus may undergo other kinds of radioactive decay (alpha decay, for example). In nature, most isotopes are beta stable, but a few exceptions exist with half-lives so long that they have not had enough time to decay since the moment of their nucleosynthesis. One example is 40
19K
, which undergoes all three types of beta decay (
β−
,
β+
and electron capture) with a half life of 1.277×109 years.
Double beta decay
Some nuclei can undergo double beta decay (ββ decay) where the charge of the nucleus changes by two units. In most practically interesting cases, single beta decay is energetically forbidden for such nuclei, because when β and ββ decays are both allowed, the probability of β decay is (usually) much higher, preventing investigations of very rare ββ decays. Thus, ββ decay is usually studied only for beta stable nuclei. Like single beta decay, double beta decay does not change A; thus, at least one of the nuclides with some given A has to be stable with regard to both single and double beta decay.
Beta decay can be considered as a perturbation as described in quantum mechanics, and thus follows Fermi's Golden Rule.
Kurie plot
This section needs expansion. You can help by adding to it. (June 2008) |
A Kurie plot (also known as a Fermi-Kurie plot) is a graph used in studying beta decay, in which the square root of the number of beta particles whose momenta (or energy) lie within a certain narrow range, divided by a function worked out by Fermi, is plotted against beta-particle energy; it is a straight line for allowed transitions and some forbidden transitions, in accord with the Fermi beta-decay theory.
History
This section needs expansion. You can help by adding to it. (June 2008) |
Historically, the study of beta decay provided the first physical evidence of the neutrino. In 1911 Lise Meitner and Otto Hahn performed an experiment that showed that the energies of electrons emitted by beta decay had a continuous rather than discrete spectrum. This was in apparent contradiction to the law of conservation of energy, as it appeared that energy was lost in the beta decay process. A second problem was that the spin of the Nitrogen-14 atom was 1, in contradiction to the Rutherford prediction of ½.
In 1920-1927, Charles Drummond Ellis (along with James Chadwick and colleagues) established clearly that the beta decay spectrum is really continuous, ending all controversies.
In a famous letter written in 1930 Wolfgang Pauli suggested that in addition to electrons and protons atoms also contained an extremely light neutral particle which he called the neutron. He suggested that this "neutron" was also emitted during beta decay and had simply not yet been observed. In 1931 Enrico Fermi renamed Pauli's "neutron" to neutrino, and in 1934 Fermi published a very successful model of beta decay in which neutrinos were produced.
There was also a guy named Sir Fracsico Elmo and he played happy time with all the elements.
See also
References
- Franz N. D. Kurie, J. R. Richardson, H. C. Paxton (1936). "The Radiations Emitted from Artificially Produced Radioactive Substances. I. The Upper Limits and Shapes of the β-Ray Spectra from Several Elements". Physical Review. 49 (5): 368–381. doi:10.1103/PhysRev.49.368.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - F. N. D. Kurie (1948). "On the Use of the Kurie Plot". Physical Review. 73 (10): 1207. doi:10.1103/PhysRev.73.1207.
{{cite journal}}: Unknown parameter|month=ignored (help)
