4-Nitroaniline
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
4-Nitroaniline | |||
| Systematic IUPAC name
4-Nitrobenzenamine | |||
| Other names
p-Nitroaniline
1-Amino-4-nitrobenzene p-Nitrophenylamine | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.555 | ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C6H6N2O2 | |||
| Molar mass | 138.12 g/mol | ||
| Appearance | yellow or brown powder | ||
| Odor | faint, ammonia-like | ||
| Density | 1.437 g/ml, solid | ||
| Melting point | 146 to 149 °C (295 to 300 °F; 419 to 422 K) (lit.) | ||
| Boiling point | 332 °C (630 °F; 605 K) | ||
| 0.8 mg/ml at 18.5 °C (IPCS) | |||
| Vapor pressure | 0.00002 mmHg (20°C)[1] | ||
| -66.43·10−6 cm3/mol | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Toxic | ||
| NFPA 704 (fire diamond) | |||
| Flash point | 199 °C (390 °F; 472 K) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
3249 mg/kg (rat, oral) 750 mg/kg (rat, oral) 450 mg/kg (guinea pig, oral) 810 mg/kg (mouse, oral)[2] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 6 mg/m3 (1 ppm) [skin][1] | ||
REL (Recommended)
|
TWA 3 mg/m3 [skin][1] | ||
IDLH (Immediate danger)
|
300 mg/m3[1] | ||
| Safety data sheet (SDS) | JT Baker | ||
| Related compounds | |||
Related compounds
|
2-Nitroaniline, 3-Nitroaniline | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
4-Nitroaniline, p-nitroaniline or 1-amino-4-nitrobenzene is an organic compound with the formula C6H6N2O2. It is an organic chemical compound, consisting of a benzene ring in which an amino group is para to a nitro group. This chemical is commonly used as an intermediate in the synthesis of dyes, antioxidants, pharmaceuticals, and gasoline, in gum inhibitors, poultry medicines, and as a corrosion inhibitor.
Synthesis
It is produced industrially via the amination of 4-nitrochlorobenzene:[3]
- ClC6H4NO2 + 2 NH3 → H2NC6H4NO2 + NH4Cl
Below is a laboratory synthesis of 4-nitroaniline from aniline. The key step in this reaction sequence is an electrophilic aromatic substitution to install the nitro group para to the amino group. After this reaction, a separation must be performed to remove 2-nitroaniline, which is also formed in a small amount during the reaction.[4]
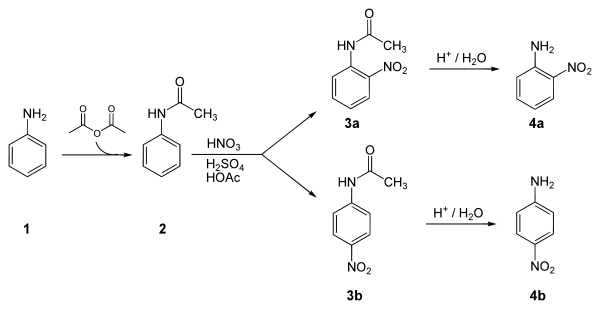
Applications
4-Nitroaniline is mainly consumed industrially as a precursor to p-phenylenediamine, an important dye component. The reduction is effected using iron metal and by catalytic hydrogenation.[3]
It is a starting material for the synthesis of Para Red, the first azo dye:[5]
When heated with sulfuric acid, it polymerizes explosively into a rigid foam.[6]

Laboratory use
Nitroaniline is a solvatochromic dye used for determining Kamlet-Taft solvent parameters. The position of its UV-visual peak changes with the balance of hydrogen bonding acceptors and donors in the solvent.[citation needed]
Toxicity
The compound is toxic by way of inhalation, ingestion, and absorption, and should be handled with care. Its LD50 in rats is 750 mg/kg when administered orally. 4-Nitroaniline is particularly harmful to all aquatic organisms, and can cause long-term damage to the environment if released as a pollutant.[citation needed]
See also
References
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0449". National Institute for Occupational Safety and Health (NIOSH).
- ^ "p-Nitroaniline". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b Gerald Booth (2007). "Nitro Compounds, Aromatic". Weinheim: Wiley-VCH. doi:10.1002/14356007.a17_411.
{{cite journal}}: Cite journal requires|journal=(help); Unknown parameter|encyclopedia=ignored (help) - ^ Mohrig, J.R.; Morrill, T.C.; Hammond, C.N.; Neckers, D.C. "Synthesis 5: Synthesis of the Dye Para Red from Aniline." Experimental Organic Chemistry. Freeman: New York, NY, 1997; pp 456-467.
- ^ Williamson, Kenneth L. (2002). Macroscale and Microscale Organic Experiments, Fourth Edition. Houghton-Mifflin. ISBN 0-618-19702-8.
- ^ "Studies on nitroaniline–sulfuric acid compositions: Aphrogenic pyrostats". doi:10.1002/app.1970.070140813.
{{cite journal}}: Cite journal requires|journal=(help)



