Thermogenesis
This article needs additional citations for verification. (February 2007) |
Thermogenesis is the process of heat production in organisms. It occurs in all warm-blooded animals, and also in a few species of thermogenic plants such as the Eastern skunk cabbage, the Voodoo lily (Sauromatum venosum), and the giant water lilies of the genus Victoria. The lodgepole pine dwarf mistletoe, Arceuthobium americanum, disperses its seeds explosively through thermogenesis.[1]
Types
Depending on whether or not they are initiated through locomotion and intentional movement of the muscles, thermogenic processes can be classified as one of the following:
- Exercise-associated thermogenesis (EAT)
- Non-exercise activity thermogenesis (NEAT), energy expended for everything that is not sleeping, eating or sports-like exercise.[2]
- Diet-induced thermogenesis (DIT)
Shivering
One method to raise temperature is through shivering. It produces heat because the conversion of the chemical energy of ATP into kinetic energy causes almost all of the energy to show up as heat. Shivering is the process by which the body temperature of hibernating mammals (such as some bats and ground squirrels) is raised as these animals emerge from hibernation.
Non-shivering
This section may be too technical for most readers to understand. (December 2018) |
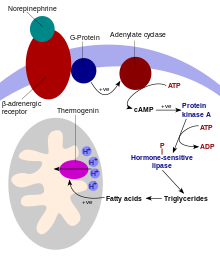
Non-shivering thermogenesis occurs in brown adipose tissue (brown fat)[3] that is present in almost all eutherians (swine being the only exception currently known[4][5]).[6] Brown adipose tissue has a unique uncoupling protein (thermogenin, also known as uncoupling protein 1) that allows the uncoupling of protons (H+) moving down their mitochondrial gradient from the synthesis of ATP, thus allowing the energy to be dissipated as heat.[7] The atomic structure of human uncoupling protein 1 UCP1 has been solved by cryogenic-electron microscopy. The structure has the typical fold of a member of the SLC25 family.[8][9] UCP1 is locked in a cytoplasmic-open state by guanosine triphosphate in a pH-dependent manner, preventing proton leak.[10]
In this process, substances such as free fatty acids (derived from triacylglycerols) remove purine (ADP, GDP and others) inhibition of thermogenin, which causes an influx of H+ into the matrix of the mitochondrion and bypasses the ATP synthase channel. This uncouples oxidative phosphorylation, and the energy from the proton motive force is dissipated as heat rather than producing ATP from ADP, which would store chemical energy for the body's use. Thermogenesis can also be produced by leakage of the sodium-potassium pump and the Ca2+ pump.[11] Thermogenesis is contributed to by futile cycles, such as the simultaneous occurrence of lipogenesis and lipolysis[12] or glycolysis and gluconeogenesis. In a broader context, futile cycles can be influenced by activity/rest cycles such as the Summermatter cycle.[13]
Acetylcholine stimulates muscle to raise metabolic rate.[14]
The low demands of thermogenesis mean that free fatty acids draw, for the most part, on lipolysis as the method of energy production.
A comprehensive list of human and mouse genes regulating cold-induced thermogenesis (CIT) in living animals (in vivo) or tissue samples (ex vivo) has been assembled[15] and is available in CITGeneDB.[16]
Evolutionary history
In avians and eutherians
The biological processes which allow for thermogenesis in animals did not evolve from a singular, common ancestor.[17] Rather, avian (birds) and eutherian (placental mammalian) lineages developed the ability to perform thermogenesis independently through separate evolutionary processes.[17] The fact that the same evolutionary character evolved independently in two different lineages after their last known common ancestor means that thermogenic processes are classified as an example of convergent evolution. However, while both clades are capable of performing thermogenesis, the biological processes involved are different. The reason that both avians and eutherians both developed the capacity to perform thermogenesis is a subject of ongoing study by evolutionary biologists, and two competing explanations have been proposed to explain why this character appears in both lineages.[17]
One explanation for the convergence is the “aerobic capacity” model. This theory suggests that natural selection favored individuals with higher resting metabolic rates, and that as the metabolic capacity of birds and eutherians increased, they developed the capacity for endothermic thermogenesis.[18] Researchers have linked high levels of oxygen consumption with high resting metabolic rates, suggesting that the two are directly correlated. Rather than animals developing the capacity to maintain high and stable body temperatures only to be able to thermoregulate without the aid of the environment, this theory suggests that thermogenesis is actually a by-product of natural selection for higher aerobic and metabolic capacities.[19] These higher metabolic capacities may initially have evolved for the simple reason that animals capable of metabolizing more oxygen for longer periods of time would have been better suited to, for example, run from predators or gather food.[19] This model explaining the development of thermogenesis is older and more widely accepted among evolutionary biologists who study thermogenesis.
The second explanation is the “parental care” model. This theory proposes that the convergent evolution of thermogenesis in birds and eutherians is based on shared behavioral traits. Specifically, birds and eutherians both provide high levels of parental care to young offspring. This high level of care is theorized to give new born or hatched animals the opportunity to mature more rapidly because they have to expend less energy to satisfy their food, shelter, and temperature needs.[17] The “parental care” model thus proposes that higher aerobic capacity was selected for in parents as a means of meeting the needs of their offspring.[18] While the “parental care” model does differ from the “aerobic capacity” model, it shares some similarities in that both explanations for the rise of thermogenesis rest on natural selection favoring individuals with higher aerobic capacities for one reason or another. The primary difference between the two theories is that the “parental care” model proposes that a specific biological function (childcare) resulted in selective pressure for higher metabolic rates.
Despite both relying on similar explanations for the process by which organisms gained the capacity to perform non-shivering thermogenesis, neither of these explanations has secured a large enough consensus to be considered completely authoritative on convergent evolution of NST in birds and mammals, and scientists continue to conduct studies which support both positions.[19][17][18]
Non-shivering thermogenesis
Brown Adipose Tissue (BAT) thermogenesis is one of the two known forms of non-shivering thermogenesis (NST). This type of heat-generation occurs only in eutherians, not in birds or other thermogenic organisms. BAT NST occurs when Uncoupling Protein 1 (UCP1) performs oxidative phosphorylation in eutherians’ bodies resulting in the generation of heat (Berg et al., 2006, p. 1178).[20] This process generally only begins in eutherians after they have been subjected to low temperatures for an extended period of time, after which the process allows an organism's body to maintain a high and stable temperature without a reliance on environmental thermoregulation mechanisms (such as sunlight/shade). Because eutherians are the only clade which store brown adipose tissue, scientists previously thought that UCP1 evolved in conjunction with brown adipose tissue. However, recent studies have shown that UCP1 can also be found in non-eutherians like fish, birds, and reptiles.[21] This discovery means that UCP1 probably existed in a common ancestor before the radiation of the eutherian lineage. Since this evolutionary split, though, UCP1 has evolved independently in eutherians, through a process which scientists believe was not driven by natural selection, but rather by neutral processes like genetic drift.[21]
Evolution of Skeletal-Muscle Non-Shivering Thermogenesis
The second form of NST occurs in skeletal muscle. While eutherians use both BAT and skeletal muscle NST for thermogenesis, birds only use the latter form. This process has also been shown to occur in rare instances in fish.[17] In skeletal muscle NST, Calcium ions slip across muscle cells to generate heat.[17] Even though BAT NST was originally thought to be the only process by which animals could maintain endothermy, scientists now suspect that skeletal muscle NST was the original form of the process and that BAT NST developed later.[17] Though scientists once also believed that only birds maintained their body temperatures using skeletal muscle NST, research in the late 2010s showed that mammals and other eutherians also use this process when they do not have adequate stores of brown adipose tissue in their bodies.[22]
Skeletal muscle NST might also be used to maintain body temperature in heterothermic mammals during states of torpor or hibernation.[17] Given that early eutherians and the reptiles which later evolved into avian lineages were either heterothermic or ectothermic, both forms of NST are thought not to have developed fully until after the K-pg extinction roughly 66 million years ago.[23] However, some estimates place the evolution of these characters earlier, at roughly 100 mya.[24] It is most likely that the process of evolving the capacity for thermogenesis as it currently exists was a process which began prior to the K-pg extinction and ended well after. The fact that skeletal muscle NST is common among eutherians during periods of torpor and hibernation further supports the theory that this form of thermogenesis is older than BAT NST. This is because early eutherians would not have had the capacity for non-shivering thermogenesis as it currently exists, so they more frequently used torpor and hibernation as means of thermal regulation, relying on systems which, in theory, predate BAT NST. However, there remains no consensus among evolutionary biologists on the order in which the two processes evolved, nor an exact timeframe for their evolution.
Regulation
Non-shivering thermogenesis is regulated mainly by thyroid hormone and the sympathetic nervous system. Some hormones, such as norepinephrine and leptin, may stimulate thermogenesis by activating the sympathetic nervous system. Rising insulin levels after eating may be responsible for diet-induced thermogenesis (thermic effect of food). Progesterone also increases body temperature.
See also
References
- ^ deBruyn RA, Paetkau M, Ross KA, Godfrey DV, Friedman CR (February 2015). "Thermogenesis-triggered seed dispersal in dwarf mistletoe". Nature Communications. 6 (1): 6262. Bibcode:2015NatCo...6.6262D. doi:10.1038/ncomms7262. PMC 4347025. PMID 25662062.
- ^ Levine JA (December 2002). "Non-exercise activity thermogenesis (NEAT)". Best Practice & Research. Clinical Endocrinology & Metabolism. 16 (4): 679–702. doi:10.1053/beem.2002.0227. PMID 12468415.
- ^ Fox SI (2011). Human Physiology (Twelfth ed.). McGraw Hill. p. 667.
- ^ Berg F, Gustafson U, Andersson L (August 2006). "The uncoupling protein 1 gene (UCP1) is disrupted in the pig lineage: a genetic explanation for poor thermoregulation in piglets". PLOS Genetics. 2 (8): e129. doi:10.1371/journal.pgen.0020129. PMC 1550502. PMID 16933999.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Hou L, Hu CY, Wang C (April 2017). "Pig Has No Brown Adipose Tissue". The FASEB Journal. 31 (S1). doi:10.1096/fasebj.31.1_supplement.lb695.
- ^ Hayward JS, Lisson PA (1992). "Evolution of brown fat: its absence in marsupials and monotremes". Canadian Journal of Zoology. 70 (1): 171–179. doi:10.1139/z92-025.
- ^ Cannon B, Nedergaard J (January 2004). "Brown adipose tissue: function and physiological significance". Physiological Reviews. 84 (1): 277–359. doi:10.1152/physrev.00015.2003. PMID 14715917. S2CID 14289041.
- ^ Ruprecht JJ, Kunji ER (March 2020). "The SLC25 Mitochondrial Carrier Family: Structure and Mechanism". Trends in Biochemical Sciences. 45 (3): 244–258. doi:10.1016/j.tibs.2019.11.001. PMC 7611774. PMID 31787485.
- ^ Kunji ER, King MS, Ruprecht JJ, Thangaratnarajah C (September 2020). "The SLC25 Carrier Family: Important Transport Proteins in Mitochondrial Physiology and Pathology". Physiology. 35 (5): 302–327. doi:10.1152/physiol.00009.2020. PMC 7611780. PMID 32783608.
- ^ Jones SA, Gogoi P, Ruprecht JJ, King MS, Lee Y, Zögg T, et al. (June 2023). "Structural basis of purine nucleotide inhibition of human uncoupling protein 1". Science Advances. 9 (22): eadh4251. doi:10.1126/sciadv.adh4251. PMC 10413660. PMID 37256948. S2CID 259002752.
- ^ Morrissette JM, Franck JP, Block BA (March 2003). "Characterization of ryanodine receptor and Ca2+-ATPase isoforms in the thermogenic heater organ of blue marlin (Makaira nigricans)". The Journal of Experimental Biology. 206 (Pt 5): 805–812. doi:10.1242/jeb.00158. PMID 12547935.
- ^ Solinas G, Summermatter S, Mainieri D, Gubler M, Pirola L, Wymann MP, et al. (November 2004). "The direct effect of leptin on skeletal muscle thermogenesis is mediated by substrate cycling between de novo lipogenesis and lipid oxidation" (PDF). FEBS Letters. 577 (3): 539–544. doi:10.1016/j.febslet.2004.10.066. PMID 15556643. S2CID 18266296.
- ^ Summermatter S, Handschin C (November 2012). "PGC-1α and exercise in the control of body weight". International Journal of Obesity. 36 (11): 1428–1435. doi:10.1038/ijo.2012.12. PMID 22290535.
- ^ Evans SS, Repasky EA, Fisher DT (June 2015). "Fever and the thermal regulation of immunity: the immune system feels the heat". Nature Reviews. Immunology. 15 (6): 335–349. doi:10.1038/nri3843. PMC 4786079. PMID 25976513.
- ^ Li J, Deng SP, Wei G, Yu P (January 2018). "CITGeneDB: a comprehensive database of human and mouse genes enhancing or suppressing cold-induced thermogenesis validated by perturbation experiments in mice". Database. 2018. doi:10.1093/database/bay012. PMC 5868181. PMID 29688375.
- ^ Li J, Deng SP, Wei G, Yu P (January 2018). "CITGeneDB: a comprehensive database of human and mouse genes enhancing or suppressing cold-induced thermogenesis validated by perturbation experiments in mice". Database. 2018. doi:10.1093/database/bay012. PMC 5868181. PMID 29688375.
- ^ a b c d e f g h i Nowack J, Giroud S, Arnold W, Ruf T (2017-11-09). "Muscle Non-shivering Thermogenesis and Its Role in the Evolution of Endothermy". Frontiers in Physiology. 8: 889. doi:10.3389/fphys.2017.00889. PMC 5684175. PMID 29170642.
- ^ a b c Koteja P (March 2000). "Energy assimilation, parental care and the evolution of endothermy". Proceedings. Biological Sciences. 267 (1442): 479–484. doi:10.1098/rspb.2000.1025. PMC 1690555. PMID 10737405.
- ^ a b c Bennett AF, Ruben JA (November 1979). "Endothermy and activity in vertebrates". Science. 206 (4419): 649–654. Bibcode:1979Sci...206..649B. doi:10.1126/science.493968. PMID 493968.
- ^ Berg F, Gustafson U, Andersson L (August 2006). Barsh GS (ed.). "The uncoupling protein 1 gene (UCP1) is disrupted in the pig lineage: a genetic explanation for poor thermoregulation in piglets". PLOS Genetics. 2 (8): e129. doi:10.1371/journal.pgen.0020129. PMC 1550502. PMID 16933999.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b Hughes DA, Jastroch M, Stoneking M, Klingenspor M (January 2009). "Molecular evolution of UCP1 and the evolutionary history of mammalian non-shivering thermogenesis". BMC Evolutionary Biology. 9 (1): 4. doi:10.1186/1471-2148-9-4. PMC 2627829. PMID 19128480.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Bal NC, Maurya SK, Singh S, Wehrens XH, Periasamy M (August 2016). "Increased Reliance on Muscle-based Thermogenesis upon Acute Minimization of Brown Adipose Tissue Function". The Journal of Biological Chemistry. 291 (33): 17247–17257. doi:10.1074/jbc.M116.728188. PMC 5016124. PMID 27298322.
- ^ Lovegrove BG (2012). "A Single Origin of Heterothermy in Mammals". Living in a Seasonal World. Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 3–11. doi:10.1007/978-3-642-28678-0_1. ISBN 978-3-642-28677-3.
- ^ Labocha MK, Hayes JP (2019). "Endotherm". Encyclopedia of Ecology. Elsevier. pp. 368–374. doi:10.1016/b978-0-12-409548-9.11221-7. ISBN 9780444641304.
External links
- Thermogenesis at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
