miR-324-5p
miR-324-5p is a microRNA that functions in cell growth, apoptosis, cancer,[1] epilepsy,[2][3] neuronal differentiation,[4] psychiatric conditions,[5] cardiac disease pathology,[6][1] and more.[7] As a microRNA, it regulates gene expression through targeting mRNAs. Additionally, miR-324-5p it is both an intracellular miRNA, meaning it is commonly found within the microenvironment of the cell, and one of several circulating miRNAs found throughout the body.[8] Its presence throughout the body both within and external to cells may contribute to miR-324-5p's wide array of functions and role in numerous disease pathologies – especially cancer – in various organ systems.
History
miR-324-5p first appeared in literature in a paper published by John Kim et. al. in early 2004 that identified 32 entirely new miRNAs from cultured rat cortical neurons using miRNA cloning and RNA analysis.[9] The miRNA quickly gained traction in scientific literature, appearing in articles about the evolutionary conservation of microRNAs,[10] HIV,[11] cancer,[12] and other topics within a few years. Today, the functions and roles of miR-324-5p are still not yet fully characterized.[13]
Structure and targets
miR-324-5p is a reverse strand miRNA, meaning it is produced from the 5' end of the associated RNA, and spans from position 7,223,342 to 7,223,364 on chromosome 17.[14] Its sequence is CGCAUCCCCUAGGGCAUUGGUG.[15][16]
miRNA forms following cleavage of pre-miRNA at the hairpin loop by the enzyme dicer within the cytosol. Interestingly, both strands of miR-324's pre-miRNA hairpin loop structure, miR-324-5p and miR-324-3p, become active miRNAs with distinct targets and functions.[17] miR-324-5p has between 166 and 469 predicted targets,[18][19][14] including regulators of cell growth, proliferation, survival, cytoskeletal structure, ATP transport, and ion channels.[18] Though miR-324-5p is found on chromosome 17, its targets span across all chromosomes.[16]
Functions
Cell growth and survival
miR-324-5p likely regulates cell growth and survival through interaction with multiple pathways. Published research demonstrates that this miRNA interacts with the Hedgehog (HH) signaling pathway via interactions with HH transcription factor Gli1[20] and HH protein receptor Smo,[21] often contributing to tumorigenesis. miR-324-5p's activating interaction with the protein NfkB also regulates numerous components of cell survival, including cell cycle control, enzyme synthesis, and cell adhesion.[22] In addition, miR-324-5p regulates components of the MAPK pathway, influencing cell growth, proliferation, and survival. Specifically, miR-324-5p downregulates RAF and ERK and is necessary for normal levels of cell growth. Reduced expression leads to increased cell growth and proliferation, and overexpression limits growth, leading to its role in oncogenesis.[23]
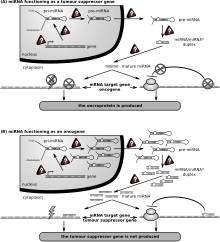
Cancer
Both up and downregulation of miR-324-5p is shown to contribute to various types of cancer.[17]
miR-324-5p plays a role in inflammation and tumorigenesis in colorectal cancer through regulation of CUEDC2, which regulates inflammation via interaction with NF-kB signaling.[12] miR-324-5p can inhibit glioma proliferation,[20] suppress hepatocellular carcinoma and nasopharyngeal carcinoma cell invasion,[24][25] and regulate growth and pathology in multiple myeloma.[26] Additionally, chromosome 17 deletions, which include deletion of miR-324-5p, are present in 10% of multiple myeloma patients and are associated with poorer prognosis.[26]
In contrast, overexpression of miR-324-5p in gastric cancer cells reduces cell death and promotes growth and proliferation.[27] miR-324-5p has also been shown to reduce the viability of gastric cancer cells via downregulation of TSPAN8, and miR-324-5p expression increased apoptosis in these same gastric cancer cells.[28]
Epilepsy
Seizures are characterized by high levels of synchronized neuronal activity. One important regulator of neuronal activity is the hyperpolarizing A-type current mediated by potassium channel KV4.2.[29] miR-324-5p downregulates KV4.2, exacerbating conditions that lead to seizure onset, and downregulation of miR-324-5p in mouse models of epilepsy is seizure-suppressive.[3]
Changes in miRNA expression are seen in epileptogenesis and in other disease pathologies.[30][31] In epilepsy, miR-324-5p expression has been shown to increase[32] and decrease[33] at different timepoints and loci.
Importantly, miR-324-5p has increased association with the RISC complex following seizure in mice, indicating more suppressive activity.[3][34]
Overall, this suggests that miR-324-5p plays a role in epileptogenesis via targeting of potassium channel KV4.2.
Cardiac disease
miR-324-5p contributes to cardiac disease pathophysiology and cardiomyocite death through translational inhibition of Mtfr1, leading to reduced mitochondrial fission, apoptosis, and myocardial infarction.[6]
Psychiatric conditions
MiRNA expression profiles are altered in psychiatric conditions, including depression,[5] anxiety,[35] and PTSD.[36] It has been demonstrated that miR-324-5p expression is altered in the brains of suicide victims with depression[5] and in the amygdala, the fear center of the brain, in PTSD.[36] MiRNAs are an underexplored potential biomarker and target for treatment for psychiatric disease.[37]
Future research and potential in medicine
miRNA-324-5p is a relatively new and understudied microRNA. It is an important regulator in several diseases, and its effects span across the body from neuronal dysregulation in seizure to hepatocellular carcinoma and cardiac disease. Because microRNAs have numerous targets, they are capable of regulating multiple pathways and circuits, an ability that may be useful in the treatment of complex disorders like epilepsy in which many subsystems are dysregulated. However, the wide-ranging functions of miRNAs may be limiting as well. microRNA expression modulation could lead to unanticipated physiological effects and not provide adequate specificity.[38]
References
- ^ a b Katoh M (2014). "Cardio-miRNAs and onco-miRNAs: circulating miRNA-based diagnostics for non-cancerous and cancerous diseases". Frontiers in Cell and Developmental Biology. 2: 61. doi:10.3389/fcell.2014.00061. PMC 4207049. PMID 25364765.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Yao X (2012). Regulation of A-type potassium channel Kv4.2 expression by FMRP and miR-324-5p (Ph.D. thesis). Emory University.
- ^ a b c Gross C, Yao X, Engel T, Tiwari D, Xing L, Rowley S, et al. (September 2016). "MicroRNA-Mediated Downregulation of the Potassium Channel Kv4.2 Contributes to Seizure Onset". Cell Reports. 17 (1): 37–45. doi:10.1016/j.celrep.2016.08.074. PMC 5061042. PMID 27681419.
- ^ Stappert L, Borghese L, Roese-Koerner B, Weinhold S, Koch P, Terstegge S, et al. (2013). "MicroRNA-based promotion of human neuronal differentiation and subtype specification". PLOS One. 8 (3): e59011. doi:10.1371/journal.pone.0059011. PMC 3601127. PMID 23527072.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c Smalheiser NR, Lugli G, Rizavi HS, Torvik VI, Turecki G, Dwivedi Y (2012). "MicroRNA expression is down-regulated and reorganized in prefrontal cortex of depressed suicide subjects". PLOS One. 7 (3): e33201. doi:10.1371/journal.pone.0033201. PMC 3302855. PMID 22427989.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b Wang K, Zhang DL, Long B, An T, Zhang J, Zhou LY, et al. (December 2015). "NFAT4-dependent miR-324-5p regulates mitochondrial morphology and cardiomyocyte cell death by targeting Mtfr1". Cell Death & Disease. 6 (12): e2007. doi:10.1038/cddis.2015.348. PMC 4720883. PMID 26633713.
- ^ Hervé M, Ibrahim EC (August 2016). "MicroRNA screening identifies a link between NOVA1 expression and a low level of IKAP in familial dysautonomia". Disease Models & Mechanisms. 9 (8): 899–909. doi:10.1242/dmm.025841. PMC 5007982. PMID 27483351.
- ^ Ellis KL, Cameron VA, Troughton RW, Frampton CM, Ellmers LJ, Richards AM (October 2013). "Circulating microRNAs as candidate markers to distinguish heart failure in breathless patients". European Journal of Heart Failure. 15 (10): 1138–47. doi:10.1093/eurjhf/hft078. PMID 23696613.
- ^ Kim J, Krichevsky A, Grad Y, Hayes GD, Kosik KS, Church GM, et al. (January 2004). "Identification of many microRNAs that copurify with polyribosomes in mammalian neurons". Proceedings of the National Academy of Sciences of the United States of America. 101 (1): 360–5. doi:10.1073/pnas.2333854100. PMC 314190. PMID 14691248.
- ^ Bompfünewerer AF, Flamm C, Fried C, Fritzsch G, Hofacker IL, Lehmann J, et al. (April 2005). "Evolutionary patterns of non-coding RNAs". Theory in Biosciences = Theorie in Den Biowissenschaften. 123 (4): 301–69. CiteSeerX 10.1.1.144.5935. doi:10.1016/j.thbio.2005.01.002. PMID 18202870.
- ^ Hariharan M, Scaria V, Pillai B, Brahmachari SK (December 2005). "Targets for human encoded microRNAs in HIV genes". Biochemical and Biophysical Research Communications. 337 (4): 1214–8. doi:10.1016/j.bbrc.2005.09.183. PMID 16236258.
- ^ a b Chen Y, Wang SX, Mu R, Luo X, Liu ZS, Liang B, et al. (June 2014). "Dysregulation of the miR-324-5p-CUEDC2 axis leads to macrophage dysfunction and is associated with colon cancer". Cell Reports. 7 (6): 1982–93. doi:10.1016/j.celrep.2014.05.007. PMID 24882011.
- ^ "MIR324 microRNA 324 [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2018-11-18.
- ^ a b "Homo sapiens (human) hsa-miR-324-5p | URS000005481D". RNAcentral. European Molecular Biology Laboratory.
- ^ "MiRNA Entry for MI0000813". miRBase: the microRNA database. The University of Manchester.
- ^ a b "miRNA ID: hsa-miR-324-5p". TargetMiner: Prediction of miRNA Targets. Indian Statistical Institute.
- ^ a b Kuo WT, Yu SY, Li SC, Lam HC, Chang HT, Chen WS, et al. (October 2016). "MicroRNA-324 in Human Cancer: miR-324-5p and miR-324-3p Have Distinct Biological Functions in Human Cancer". Anticancer Research. 36 (10): 5189–5196. doi:10.21873/anticanres.11089. PMID 27798879.
- ^ a b Wang, Xiaowei. "predicted targets for hsa-miR-324-5p in miRDB". miRDB: predicted microRNA targets in animals. St. Louis: Department of Radiation Oncology, Washington University School of Medicine.
{{cite web}}: Unknown parameter|name-list-format=ignored (|name-list-style=suggested) (help) - ^ "Predicted miRNA targets of miR-324-5p". TargetscanHuman 7.1. Whitehead Institute for Biomedical Research.
- ^ a b Xu HS, Zong HL, Shang M, Ming X, Zhao JP, Ma C, et al. (2014-03-30). "MiR-324-5p inhibits proliferation of glioma by target regulation of GLI1". European Review for Medical and Pharmacological Sciences. 18 (6): 828–32. PMID 24706306.
- ^ Ferretti E, De Smaele E, Miele E, Laneve P, Po A, Pelloni M, et al. (October 2008). "Concerted microRNA control of Hedgehog signalling in cerebellar neuronal progenitor and tumour cells". The EMBO Journal. 27 (19): 2616–27. doi:10.1038/emboj.2008.172. PMC 2567402. PMID 18756266.
- ^ Song L, Liu D, Zhao Y, He J, Kang H, Dai Z, et al. (August 2015). "Sinomenine inhibits breast cancer cell invasion and migration by suppressing NF-κB activation mediated by IL-4/miR-324-5p/CUEDC2 axis". Biochemical and Biophysical Research Communications. 464 (3): 705–10. doi:10.1016/j.bbrc.2015.07.004. PMID 26166821.
- ^ Liu SM, Lu J, Lee HC, Chung FH, Ma N (October 2014). "miR-524-5p suppresses the growth of oncogenic BRAF melanoma by targeting BRAF and ERK2". Oncotarget. 5 (19): 9444–59. doi:10.18632/oncotarget.2452. PMC 4253445. PMID 25275294.
- ^ Cao L, Xie B, Yang X, Liang H, Jiang X, Zhang D, et al. (2015). "MiR-324-5p Suppresses Hepatocellular Carcinoma Cell Invasion by Counteracting ECM Degradation through Post-Transcriptionally Downregulating ETS1 and SP1". PLOS One. 10 (7): e0133074. doi:10.1371/journal.pone.0133074. PMC 4503725. PMID 26177288.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Liu C, Li G, Yang N, Su Z, Zhang S, Deng T, Ren S, Lu S, Tian Y, Liu Y, Qiu Y (2017). "miR-324-3p suppresses migration and invasion by targeting WNT2B in nasopharyngeal carcinoma". Cancer Cell International. 17: 2. doi:10.1186/s12935-016-0372-8. PMC 5209830. PMID 28053597.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b Tang B, Xu A, Xu J, Huang H, Chen L, Su Y, et al. (January 2018). "MicroRNA-324-5p regulates stemness, pathogenesis and sensitivity to bortezomib in multiple myeloma cells by targeting hedgehog signaling". International Journal of Cancer. 142 (1): 109–120. doi:10.1002/ijc.31041. PMID 28905994.
- ^ Sun GL, Li Z, Wang WZ, Chen Z, Zhang L, Li Q, et al. (June 2018). "miR-324-3p promotes gastric cancer development by activating Smad4-mediated Wnt/beta-catenin signaling pathway". Journal of Gastroenterology. 53 (6): 725–739. doi:10.1007/s00535-017-1408-0. PMC 5971041. PMID 29103082.
- ^ Lin H, Zhou AJ, Zhang JY, Liu SF, Gu JX (November 2018). "MiR-324-5p reduces viability and induces apoptosis in gastric cancer cells through modulating TSPAN8". The Journal of Pharmacy and Pharmacology. 70 (11): 1513–1520. doi:10.1111/jphp.12995. PMID 30159900.
- ^ Fransén E, Tigerholm J (July 2010). "Role of A-type potassium currents in excitability, network synchronicity, and epilepsy". Hippocampus. 20 (7): 877–87. doi:10.1002/hipo.20694. PMC 3222850. PMID 19777555.
- ^ Brennan GP, Henshall DC (February 2018). "microRNAs in the pathophysiology of epilepsy". Neuroscience Letters. 667: 47–52. doi:10.1016/j.neulet.2017.01.017. PMID 28104433.
- ^ Li Y, Kowdley KV (October 2012). "MicroRNAs in common human diseases". Genomics, Proteomics & Bioinformatics. 10 (5): 246–53. doi:10.1016/j.gpb.2012.07.005. PMC 3611977. PMID 23200134.
- ^ Schouten M, Fratantoni SA, Hubens CJ, Piersma SR, Pham TV, Bielefeld P, Voskuyl RA, Lucassen PJ, Jimenez CR, Fitzsimons CP (July 2015). "MicroRNA-124 and -137 cooperativity controls caspase-3 activity through BCL2L13 in hippocampal neural stem cells". Scientific Reports. 5: 12448. doi:10.1038/srep12448. PMC 4513647. PMID 26207921.
- ^ Bot AM, Dębski KJ, Lukasiuk K (2013). "Alterations in miRNA levels in the dentate gyrus in epileptic rats". PLOS One. 8 (10): e76051. doi:10.1371/journal.pone.0076051. PMC 3795667. PMID 24146813.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Tiwari D, Peariso K, Gross C (January 2018). "MicroRNA-induced silencing in epilepsy: Opportunities and challenges for clinical application". Developmental Dynamics. 247 (1): 94–110. doi:10.1002/dvdy.24582. PMC 5740004. PMID 28850760.
- ^ Meydan C, Shenhar-Tsarfaty S, Soreq H (September 2016). "MicroRNA Regulators of Anxiety and Metabolic Disorders". Trends in Molecular Medicine. 22 (9): 798–812. doi:10.1016/j.molmed.2016.07.001. PMID 27496210.
- ^ a b Balakathiresan NS, Chandran R, Bhomia M, Jia M, Li H, Maheshwari RK (October 2014). "Serum and amygdala microRNA signatures of posttraumatic stress: fear correlation and biomarker potential". Journal of Psychiatric Research. 57: 65–73. doi:10.1016/j.jpsychires.2014.05.020. PMID 24998397.
- ^ Narahari A, Hussain M, Sreeram V (2017). "MicroRNAs as Biomarkers for Psychiatric Conditions: A Review of Current Research". Innovations in Clinical Neuroscience. 14 (1–2): 53–55. PMC 5373795. PMID 28386521.
- ^ Hashimoto Y, Akiyama Y, Yuasa Y (2013-05-08). "Multiple-to-multiple relationships between microRNAs and target genes in gastric cancer". PLOS One. 8 (5): e62589. doi:10.1371/journal.pone.0062589. PMC 3648557. PMID 23667495.
{{cite journal}}: CS1 maint: unflagged free DOI (link)
