Fatty amine

In chemistry, a fatty amine is any amine attached to a hydrocarbon chain of eight or more carbon atoms in length. These compounds are classified as oleochemicals. More commonly fatty amines are derived from C12-C18 hydrocarbons, which in turn are derived from the more abundant fatty acids. They are often mixtures. Commercially important members include coco amine, oleylamine, tallow amine, and soya amine. Some applications of these compounds are in fabric softeners, froth flotation agents (purification of ores), and corrosion inhibitors. They are the basis for a variety of cosmetic formulations.[1]
Production and reactions
Fatty amines are commonly prepared from fatty acids; which are themselves obtained from natural sources, typically seed-oils. The overall reaction is sometimes referred to as the Nitrile Process[2] and begins with a reaction between the fatty acid and ammonia at high temperature (>250 °C) and in the presence of a metal oxide catalyst (e.g., alumina or zinc oxide) to give the fatty nitrile.
- RCOOH + NH3 → RC≡N + 2 H2O
The fatty amine is obtained from this by hydrogenation with any of a number of reagents, including Raney nickel[3] or cobalt, and copper chromite catalysts. When conducted in the presence of excess ammonia the hydrogenation affords the primary amines.
- RCN + 2 H2 → RCH2NH2
In the absence of ammonia, secondary and tertiary amines are produced.[4]
- 2 RCN + 4 H2 → (RCH2)2NH + NH3
- 3 RCN + 6 H2 → (RCH2)3N + 2 NH3
Fatty secondary and tertiary amines
Alternatively, secondary and tertiary fatty amines can be generated by the reaction of fatty alcohols and fatty alkyl bromides with (di)alkylamines. For example 1-bromododecane reacts with dimethyl amine:
- RBr + HNMe2 → RNMe2 + HBr
By reaction with tertiary amines, long-chain alkyl bromides give quaternary ammonium salts, which are used as phase transfer catalysts.[5]
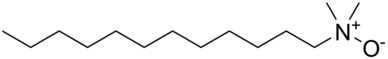
For some applications, the primary and secondary amines are often subjected to the Leuckart reaction. This reaction effects N-methylation using formaldehyde with formic acid as the reductant. These tertiary amines are precursors to quaternary ammonium salts used for a variety of applications.
Applications
The main application of fatty amines is for the production of the corresponding quaternary ammonium salts, which are used as fabric softeners. Fatty amines are also used in froth flotation, for the beneficiation of various ores. The amines bind to the surfaces of certain minerals allowing them to be readily separated from those lacking the bound amine. They are also additives in the production of asphalt.[1]
References
- ^ a b Eller, Karsten; Henkes, Erhard; Rossbacher, Roland; Höke, Hartmut (2000). "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_001. ISBN 978-3527306732.
- ^ Foley, Patrick; Kermanshahi pour, Azadeh; Beach, Evan S.; Zimmerman, Julie B. (2012). "Derivation and synthesis of renewable surfactants". Chem. Soc. Rev. 41 (4): 1499–1518. doi:10.1039/C1CS15217C. PMID 22006024.
- ^ Johansson, editors, Mikael Kjellin, Ingegard (2010). "2. Nitrogen Derivatives of Natural Fats and Oils". Surfactants from renewable resources. Chichester, West Sussex: Wiley. pp. 21-43. ISBN 9780470686607.
{{cite book}}:|first1=has generic name (help)CS1 maint: multiple names: authors list (link) - ^ Barrault, J.; Pouilloux, Y. (August 1997). "Synthesis of fatty amines. Selectivity control in presence of multifunctional catalysts". Catalysis Today. 37 (2): 137–153. doi:10.1016/S0920-5861(97)00006-0.
- ^ Dagani, M. J.; Barda, H. J.; Benya, T. J.; Sanders, D. C. (2012). "Bromine Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a04_405. ISBN 978-3527306732.
