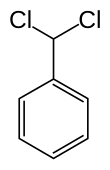
Benzal chloride
Appearance
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Dichloromethylbenzene
| |||
| Other names
Benzal chloride
α,α-Dichlorobenzenea benzylidene chloride benzyl dichloride | |||
| Identifiers | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.463 | ||
PubChem CID
|
|||
| RTECS number |
| ||
CompTox Dashboard (EPA)
|
|||
| Properties | |||
| C7H6Cl2 | |||
| Molar mass | 161.03 g/mol | ||
| Appearance | Colorless liquid | ||
| Density | 1.254 g/cm3, liquid | ||
| Melting point | −17 to −15 °C | ||
| Boiling point | 205 °C (82 °C @10 mm Hg) | ||
| low | |||
| Vapor pressure | 0.6 kPa (45 °C) | ||
| Hazards | |||
| Flash point | 93 °C | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Benzal chloride is an organic compound with the formula C6H5CHCl2.[1] This colourless liquid is a lachrymator and is used as a building block in organic synthesis.
Benzal chloride is produced by the free radical chlorination of toluene, being preceded in the process by benzyl chloride and followed by benzotrichloride
- C6H5CH3 + Cl2 → C6H5CH2Cl + HCl
- C6H5CH2Cl + Cl2 → C6H5CHCl2 + HCl
- C6H5CHCl2 + Cl2 → C6H5CCl3 + HCl
Most benzal chloride is hydrolysed to benzaldehyde:[2]
- C6H5CHCl2 + H2O → C6H5CHO + 2 HCl
Addition of strong base to benzal chloride generates phenylcarbene.
References
- ^ "BENZAL CHLORIDE". International Programme on Chemical Safety. Retrieved 2007-10-30.
- ^ Manfred Rossberg, Wilhelm Lendle, Gerhard Pfleiderer, Adolf Tögel, Eberhard-Ludwig Dreher, Ernst Langer, Heinz Rassaerts, Peter Kleinschmidt, Heinz Strack, Richard Cook, Uwe Beck, Karl-August Lipper, Theodore R. Torkelson, Eckhard Löser, Klaus K. Beutel, “Chlorinated Hydrocarbons” in Ullmann’s Encyclopedia of Chemical Technology, 2007 John Wiley & Sons: New York.