Cembrene A
(Redirected from Cembrene)
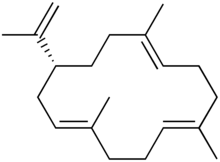
| |
| Names | |
|---|---|
| Preferred IUPAC name
(1E,5E,9E,12R)-1,5,9-Trimethyl-12-(prop-1-en-2-yl)cyclotetradeca-1,5,9-triene | |
| Other names
(R,1E,5E,9E)-1,5,9-Trimethyl-12-(prop-1-en-2-yl)cyclotetradeca-1,5,9-triene
Neocembrene-A | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C20H32 | |
| Molar mass | 272.47 g/mol |
| Boiling point | 150 to 152 °C (302 to 306 °F; 423 to 425 K) at 0.8 mmHg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cembrene A, or sometimes neocembrene, is a natural monocyclic diterpene isolated from corals of the genus Nephthea.[1] It is a colorless oil with a faint wax-like odor.
Cembrene A itself has little importance as a chemical entity, being a trail pheromone for termites;[2] however, the chemical structure of cembrene is central to a very wide variety of other natural products found both in plants and in animals.[3]
Cembrenes are biosynthesized by macrocyclization of geranylgeranyl pyrophosphate.[3]
References[edit]
- ^ Vanderah, David J.; Rutledge, Neal; Schmitz, Francis J.; Ciereszko, Leon S (1978). "Marine natural products: cembrene-A and cembrene-C from a soft coral, Nephthea species". Journal of Organic Chemistry. 43 (8): 1614–1616. doi:10.1021/jo00402a040.
- ^ Birch, A. J.; Brown, W. V.; Corrie, J. E. T.; Moore, B. P (1972). "Neocembrene-A, a termite trail pheromone". Journal of the Chemical Society, Perkin Transactions 1. 21: 2653–2658. doi:10.1039/p19720002653.
- ^ a b Terpenes: Flavors, Fragrances, Pharmaca, Pheromones, Eberhard Breitmaier, page 7. ISBN 978-3-527-31786-8
