Curium compounds
Curium compounds are compounds containing the element curium (Cm). Curium usually forms compounds in the +3 oxidation state, although compounds with curium in the +4, +5 and +6 oxidation states are also known.
Oxides
[edit]Curium readily reacts with oxygen forming mostly Cm2O3 and CmO2 oxides,[1] but the divalent oxide CmO is also known.[2] Black CmO2 can be obtained by burning curium oxalate (Cm
2(C
2O
4)
3), nitrate (Cm(NO
3)
3), or hydroxide in pure oxygen.[3][4] Upon heating to 600–650 °C in vacuum (about 0.01 Pa), it transforms into the whitish Cm2O3:[3][5]
- .
Or, Cm2O3 can be obtained by reducing CmO2 with molecular hydrogen:[6]
Also, a number of ternary oxides of the type M(II)CmO3 are known, where M stands for a divalent metal, such as barium.[7]
Thermal oxidation of trace quantities of curium hydride (CmH2–3) has been reported to give a volatile form of CmO2 and the volatile trioxide CmO3, one of two known examples of the very rare +6 state for curium.[8] Another observed species was reported to behave similar to a supposed plutonium tetroxide and was tentatively characterized as CmO4, with curium in the extremely rare +8 state;[9] but new experiments seem to indicate that CmO4 does not exist, and have cast doubt on the existence of PuO4 as well.[10]
Halides
[edit]The colorless curium(III) fluoride (CmF3) can be made by adding fluoride ions into curium(III)-containing solutions. The brown tetravalent curium(IV) fluoride (CmF4) on the other hand is only obtained by reacting curium(III) fluoride with molecular fluorine:[11]
A series of ternary fluorides are known of the form A7Cm6F31 (A = alkali metal).[12]
The colorless curium(III) chloride (CmCl3) is made by reacting curium hydroxide (Cm(OH)3) with anhydrous hydrogen chloride gas. It can be further turned into other halides such as curium(III) bromide (colorless to light green) and curium(III) iodide (colorless), by reacting it with the ammonia salt of the corresponding halide at temperatures of ~400–450°C:[13]
Or, one can heat curium oxide to ~600°C with the corresponding acid (such as hydrobromic for curium bromide).[14][15] Vapor phase hydrolysis of curium(III) chloride gives curium oxychloride:[16]
Chalcogenides and pnictides
[edit]Sulfides, selenides and tellurides of curium have been obtained by treating curium with gaseous sulfur, selenium or tellurium in vacuum at elevated temperature.[17][18] Curium pnictides of the type CmX are known for nitrogen, phosphorus, arsenic and antimony.[11] They can be prepared by reacting either curium(III) hydride (CmH3) or metallic curium with these elements at elevated temperature.[19]
Organocurium compounds and biological aspects
[edit]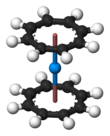
Organometallic complexes analogous to uranocene are known also for other actinides, such as thorium, protactinium, neptunium, plutonium and americium. Molecular orbital theory predicts a stable "curocene" complex (η8-C8H8)2Cm, but it has not been reported experimentally yet.[20][21]
Formation of the complexes of the type Cm(n-C
3H
7-BTP)
3 (BTP = 2,6-di(1,2,4-triazin-3-yl)pyridine), in solutions containing n-C3H7-BTP and Cm3+ ions has been confirmed by EXAFS. Some of these BTP-type complexes selectively interact with curium and thus are useful for separating it from lanthanides and another actinides.[22][23] Dissolved Cm3+ ions bind with many organic compounds, such as hydroxamic acid,[24] urea,[25] fluorescein[26] and adenosine triphosphate.[27] Many of these compounds are related to biological activity of various microorganisms. The resulting complexes show strong yellow-orange emission under UV light excitation, which is convenient not only for their detection, but also for studying interactions between the Cm3+ ion and the ligands via changes in the half-life (of the order ~0.1 ms) and spectrum of the fluorescence.[28][24][25][26][27]
Curium has no biological significance.[29] There are a few reports on biosorption of Cm3+ by bacteria and archaea, but no evidence for incorporation of curium into them.[30][31]
See also
[edit]References
[edit]- ^ Curium (in German)
- ^ Holleman, p. 1972
- ^ a b Asprey, L. B.; Ellinger, F. H.; Fried, S.; Zachariasen, W. H. (1955). "Evidence for Quadrivalent Curium: X-Ray Data on Curium Oxides1". Journal of the American Chemical Society. 77 (6): 1707. doi:10.1021/ja01611a108.
- ^ Greenwood, p. 1268
- ^ Noe, M.; Fuger, J. (1971). "Self-radiation effects on the lattice parameter of 244CmO2". Inorganic and Nuclear Chemistry Letters. 7 (5): 421. doi:10.1016/0020-1650(71)80177-0.
- ^ Haug, H. (1967). "Curium sesquioxide Cm2O3". Journal of Inorganic and Nuclear Chemistry. 29 (11): 2753. doi:10.1016/0022-1902(67)80014-9.
- ^ Fuger, J.; Haire, R.; Peterson, J. (1993). "Molar enthalpies of formation of BaCmO3 and BaCfO3". Journal of Alloys and Compounds. 200 (1–2): 181. doi:10.1016/0925-8388(93)90491-5.
- ^ Domanov, V. P.; Lobanov, Yu. V. (October 2011). "Formation of volatile curium(VI) trioxide CmO3". Radiochemistry. 53 (5). SP MAIK Nauka/Interperiodica: 453–6. doi:10.1134/S1066362211050018. S2CID 98052484.
- ^ Domanov, V. P. (January 2013). "Possibility of generation of octavalent curium in the gas phase in the form of volatile tetraoxide CmO4". Radiochemistry. 55 (1): 46–51. doi:10.1134/S1066362213010098. S2CID 98076989.
- ^ Zaitsevskii, Andréi; Schwarz, W. H. Eugen (April 2014). "Structures and stability of AnO4 isomers, An = Pu, Am, and Cm: a relativistic density functional study". Physical Chemistry Chemical Physics. 2014 (16): 8997–9001. Bibcode:2014PCCP...16.8997Z. doi:10.1039/c4cp00235k. PMID 24695756.
- ^ a b Morss, L. R.; Edelstein, N. M. and Fugere, J. (eds): The Chemistry of the Actinide Elements and transactinides, volume 3, Springer-Verlag, Dordrecht 2006, ISBN 1-4020-3555-1.
- ^ Keenan, T. (1967). "Lattice constants of K7Cm6F31 trends in the 1:1 and 7:6 alkali metal-actinide(IV) series". Inorganic and Nuclear Chemistry Letters. 3 (10): 391. doi:10.1016/0020-1650(67)80092-8.
- ^ Asprey, L. B.; Keenan, T. K.; Kruse, F. H. (1965). "Crystal Structures of the Trifluorides, Trichlorides, Tribromides, and Triiodides of Americium and Curium". Inorganic Chemistry. 4 (7): 985. doi:10.1021/ic50029a013. S2CID 96551460.
- ^ Burns, J.; Peterson, J. R.; Stevenson, J. N. (1975). "Crystallographic studies of some transuranic trihalides: 239PuCl3, 244CmBr3, 249BkBr3 and 249CfBr3". Journal of Inorganic and Nuclear Chemistry. 37 (3): 743. doi:10.1016/0022-1902(75)80532-X.
- ^ Wallmann, J.; Fuger, J.; Peterson, J. R.; Green, J. L. (1967). "Crystal structure and lattice parameters of curium trichloride". Journal of Inorganic and Nuclear Chemistry. 29 (11): 2745. doi:10.1016/0022-1902(67)80013-7. S2CID 97334114.
- ^ Weigel, F.; Wishnevsky, V.; Hauske, H. (1977). "The vapor phase hydrolysis of PuCl3 and CmCl3: heats of formation of PuOC1 and CmOCl". Journal of the Less Common Metals. 56 (1): 113. doi:10.1016/0022-5088(77)90224-7.
- ^ Troc, R. Actinide Monochalcogenides, Volume 27, Springer, 2009 ISBN 3-540-29177-6, p. 4
- ^ Damien, D.; Charvillat, J. P.; Müller, W. (1975). "Preparation and lattice parameters of curium sulfides and selenides". Inorganic and Nuclear Chemistry Letters. 11 (7–8): 451. doi:10.1016/0020-1650(75)80017-1.
- ^ Lumetta, G. J.; Thompson, M. C.; Penneman, R. A.; Eller, P. G. Curium Archived 2010-07-17 at the Wayback Machine, Chapter Nine in Radioanalytical Chemistry, Springer, 2004, pp. 1420–1421. ISBN 0387341226, ISBN 978-0387 341224
- ^ Elschenbroich, Ch. Organometallic Chemistry, 6th edition, Wiesbaden 2008, ISBN 978-3-8351-0167-8, p. 589
- ^ Kerridge, Andrew; Kaltsoyannis, Nikolas (2009). "Are the Ground States of the Later Actinocenes Multiconfigurational? All-Electron Spin−Orbit Coupled CASPT2 Calculations on An(η8-C8H8)2(An = Th, U, Pu, Cm)". The Journal of Physical Chemistry A. 113 (30): 8737–8745. Bibcode:2009JPCA..113.8737K. doi:10.1021/jp903912q. PMID 19719318.
- ^ Denecke, Melissa A.; Rossberg, André; Panak, Petra J.; Weigl, Michael; Schimmelpfennig, Bernd; Geist, Andreas (2005). "Characterization and Comparison of Cm(III) and Eu(III) Complexed with 2,6-Di(5,6-dipropyl-1,2,4-triazin-3-yl)pyridine Using EXAFS, TRFLS, and Quantum-Chemical Methods". Inorganic Chemistry. 44 (23): 8418–8425. doi:10.1021/ic0511726. PMID 16270980.
- ^ Girnt, Denise; Roesky, Peter W.; Geist, Andreas; Ruff, Christian M.; Panak, Petra J.; Denecke, Melissa A. (2010). "6-(3,5-Dimethyl-1H-pyrazol-1-yl)-2,2′-bipyridine as Ligand for Actinide(III)/Lanthanide(III) Separation". Inorganic Chemistry. 49 (20): 9627–9635. doi:10.1021/ic101309j. PMID 20849125. S2CID 978265.
- ^ a b Glorius, M.; Moll, H.; Bernhard, G. (2008). "Complexation of curium(III) with hydroxamic acids investigated by time-resolved laser-induced fluorescence spectroscopy". Polyhedron. 27 (9–10): 2113. doi:10.1016/j.poly.2008.04.002.
- ^ a b Heller, Anne; Barkleit, Astrid; Bernhard, Gert; Ackermann, Jörg-Uwe (2009). "Complexation study of europium(III) and curium(III) with urea in aqueous solution investigated by time-resolved laser-induced fluorescence spectroscopy". Inorganica Chimica Acta. 362 (4): 1215. doi:10.1016/j.ica.2008.06.016.
- ^ a b Moll, Henry; Johnsson, Anna; Schäfer, Mathias; Pedersen, Karsten; Budzikiewicz, Herbert; Bernhard, Gert (2007). "Curium(III) complexation with pyoverdins secreted by a groundwater strain of Pseudomonas fluorescens". BioMetals. 21 (2): 219–228. doi:10.1007/s10534-007-9111-x. PMID 17653625. S2CID 24565144.
- ^ a b Moll, Henry; Geipel, Gerhard; Bernhard, Gert (2005). "Complexation of curium(III) by adenosine 5′-triphosphate (ATP): A time-resolved laser-induced fluorescence spectroscopy (TRLFS) study". Inorganica Chimica Acta. 358 (7): 2275. doi:10.1016/j.ica.2004.12.055.
- ^ Bünzli, J.-C. G. and Choppin, G. R. Lanthanide probes in life, chemical, and earth sciences: theory and practice, Elsevier, Amsterdam, 1989 ISBN 0-444-88199-9
- ^ "Biochemical Periodic Table – Curium". UMBBD. 2007-06-08. Retrieved 2011-03-25.
- ^ Moll, H.; Stumpf, T.; Merroun, M.; Rossberg, A.; Selenska-Pobell, S.; Bernhard, G. (2004). "Time-resolved laser fluorescence spectroscopy study on the interaction of curium(III) with Desulfovibrio äspöensis DSM 10631T". Environmental Science & Technology. 38 (5): 1455–1459. Bibcode:2004EnST...38.1455M. doi:10.1021/es0301166. PMID 15046347.
- ^ Ozaki, T.; et al. (2002). "Association of Eu(III) and Cm(III) with Bacillus subtilis and Halobacterium salinarium". Journal of Nuclear Science and Technology. Suppl. 3: 950–953. Bibcode:2002JNST...39S.950O. doi:10.1080/00223131.2002.10875626. S2CID 98319565. Archived from the original on 2009-02-25.

![{\displaystyle {\ce {4CmO2 ->[\Delta T] 2Cm2O3 + O2}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d347ad5669ad313e0453be3c15f15e400c2d5ef8)



