Dendralene
A dendralene is a discrete acyclic cross-conjugated polyene.[1] [2] [3] The simplest dendralene is buta-1,3-diene (1) or [2]dendralene followed by [3]dendralene (2), [4]dendralene (3) and [5]dendralene (4) and so forth. [2]dendralene (butadiene) is the only one not cross-conjugated.
The name dendralene is pulled together from the words dendrimer, linear and alkene. The higher dendralenes are of scientific interest because they open up a large array of new organic compounds from a relatively simple precursor especially by Diels-Alder chemistry. Their cyclic counterparts are aptly called radialenes.
Synthesis
Vinylbutadiene ([3]dendralene) was first prepared in 1955 by pyrolysis of a triacetate:[4] [5]
This compound reacts with two equivalents of maleic anhydride in a tandem DA reaction:[6]
With benzoquinone the reaction product was a linear polymer.
Several syntheses of substituted [3]dendralenes have been reported, one via an allene,[7] one via a Horner–Wadsworth–Emmons reaction,[8] one via a cross-coupling reaction [9] and one from an allylic carbonate.[10]
One synthetic route to [4]dendralene starts from chloroprene.[11] This compound is converted to a Grignard reagent by action of magnesium metal which is then reacted with copper(I) chloride to an organocopper intermediate which is in turn dimerized using copper(II) chloride in an oxidative coupling reaction to give the butadiene dimer called [4]dendralene.
The gas-phase molecular structure of [4]dendralene has been reported [12]
The [8]-dendralene compound was reported in 2009:[13]
in a successive Kumada–Tamao–Corriu coupling and Negishi coupling.
A series of [9] to [12]-dendralenes has been reported in 2016 [14]
Properties
Even-membered dendralenes (e.g. [6]dendralene, [8]dendralene) tend to behave as chains of decoupled and isolated diene units. The ultraviolet absorption maxima equal that of butadiene itself. The dendralenes with an odd number of alkene units are more reactive due to the presence of favorable s-cis diene conformations and Diels-Alder reactions take place more easily with a preference for the termini.
Reactions
With simple dienophiles, dendrales can give quick access to complex molecules in Diels-Alder reactions. Several reaction schemes have been reported [11] [15] [16] [17] [18] [19] [20] [21] [22]
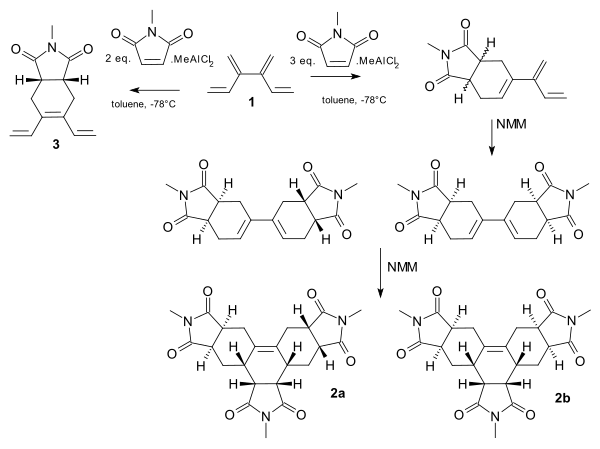
[4]dendralene shows a tandem Diels-Alder reaction with the dienophile N-methyl-maleimide (NMM).[11] Complete site selectivity is possible with the addition of the Lewis acid methyldichloroaluminium. With one set of premixing and 2 equivalents of NMM, the central diene group is targeted to the monoadduct 3. With another set and a larger amount of dienophile, the terminal groups react and the reaction proceeds from the monoadduct to the trisadducts 2 and 2b.
One reaction variation is cyclopropanation to a compound class called ivyanes with a reported synthesis in a Simmons–Smith reaction (diethyl zinc / trifluoroacetic acid) of the first 6 members.[23] These 1,1-oligocyclopropanes are stable (except when exposed to acids) and have a large heat of combustion with [6]ivyane exceeding that of cubane. The oligocyclopropane chains adopt a helical conformation. For [3]dendralene a photochemical cyclisation reaction has been reported [24]
Derviatives
A bicyclic [4]dendralene compound has been reported. [25]
References
- ^ Henning Hopf, Classics in Hydrocarbon Chemistry, Wiley VCH, 2000.
- ^ Hopf, H.; Sherburn, M. S. (2012). "Dendralenes Branch Out: Cross-Conjugated Oligoenes Allow the Rapid Generation of Molecular Complexity". Angewandte Chemie International Edition in English. 51: 2298–2338. doi:10.1002/anie.201102987.
- ^ Preparation and Synthetic Value of π-Bond-Rich Branched Hydrocarbons Michael S. Sherburn Accounts of Chemical Research 2015 48 (7), 1961-1970 doi:10.1021/acs.accounts.5b00242
- ^ Bailey, William J.; Economy, James (1955). "Pyrolysis of Esters. III. Synthesis of 2-Vinylbutadiene". J. Am. Chem. Soc. 77 (5): 1133–1136. doi:10.1021/ja01610a014.
- ^ Blomquist, A. T.; Verdol, Joseph A. (1955). "2-Vinyl-1,3-butadiene". J. Am. Chem. Soc. 77 (1): 81–83. doi:10.1021/ja01606a025.
- ^ Bailey, William J.; Economy, James; Hermes, Mathew E. (1962). "Polymers. IV. Polymeric Diels-Alder Reactions". J. Org. Chem. 27 (9): 3295–3299. doi:10.1021/jo01056a074.
- ^ Mieko Arisawa, Takumichi Sugihara and Masahiko Yamaguchi Synthesis of cross-conjugated trienes by dimerization of allenes with palladium-phenol catalyst Chem. Commun. 1998; 2615-2616 doi:10.1039/A807527A
- ^ Rekha Singh and Sunil K. Ghosh Synthesis of substituted [3]dendralenes and their unique cycloaddition reactions Chem. Commun. 2011; Advance Article doi:10.1039/C1CC14211A
- ^ Bradford, Tanya A.; Payne, Alan D.; Willis, Anthony C.; Paddon-Row, Michael N.; Sherburn, Michael S. (2007). "Cross-Coupling for Cross-Conjugation:? Practical Synthesis and Diels?Alder Reactions of [3]Dendralenes". Organic Letters. 9 (23): 4861–4864. doi:10.1021/ol7021998.
- ^ Kassem Beydoun, Hui-Jun Zhang, Basker Sundararaju, Bernard Demerseman, Mathieu Achard, Zhenfeng Xi and Christian Bruneau Efficient ruthenium-catalyzed synthesis of [3]dendralenes from 1,3-dienic allylic carbonates Chem. Commun. 2009; 6580-6582 doi:10.1039/B913595B
- ^ a b c Payne, Alan D.; Willis, Anthony C.; Sherburn, Michael S. (2005). "Practical Synthesis and Diels−Alder Chemistry of [4]Dendralene". Journal of the American Chemical Society. 127 (35): 12188–12189. doi:10.1021/ja053772+. ISSN 0002-7863.
- ^ Molecular Structure of 3,4-Dimethylenehexa-1,5-diene ([4]Dendralene), C8H10, in the Gas Phase As Determined by Electron Diffraction and ab Initio Calculations Paul T. Brain,Bruce A. Smart,Heather E. Robertson,Martin J. Davis,†, David W. H. Rankin,*, William J. Henry, and, and Ian Gosney The Journal of Organic Chemistry 1997 62 (9), 2767-2773 doi:10.1021/jo962091h
- ^ Practical Synthesis of the Dendralene Family Reveals Alternation in Behavior Alan D. Payne, Gomotsang Bojase, Michael N. Paddon-Row, and Michael S. Sherburn Angew. Chem. Int. Ed. 2009, 48, doi:10.1002/anie.200901733
- ^ Discovery and Computational Rationalization of Diminishing Alternation in [n]Dendralenes Mehmet F. Saglam, Thomas Fallon, Michael N. Paddon-Row, and Michael S. Sherburn Journal of the American Chemical Society 2016 138 (3), 1022-1032 doi:10.1021/jacs.5b11889
- ^ Brummond, Kay M.; You, Lingfeng (2005). "Consecutive Rh(I)-catalyzed Alder-ene/Diels–Alder/Diels–Alder reaction sequence affording rapid entry to polycyclic compounds". Tetrahedron. 61 (26): 6180–6185. doi:10.1016/j.tet.2005.03.141.
- ^ Hopf, H.; Yildizhan, Ş. (2011). "Highly Functionalized, Angularly Anellated Aromatic Compounds from Dendralenes". European Journal of Organic Chemistry. 2011: 2029–2034. doi:10.1002/ejoc.201001536.
- ^ A novel and facile stereocontrolled synthetic method for polyhydro-quinolines and pyridopyridazines via a diene-transmissive Diels–Alder reaction involving inverse electron-demand hetero Diels–Alder cycloaddition of cross-conjugated azatrienes Tetrahedron, Volume 64, Issue 41, 6 October 2008, Pages 9705-9716 Satoru Kobayashi, Tomoki Furuya, Takashi Otani and Takao Saito doi:10.1016/j.tet.2008.07.102
- ^ Synthesis of a Potent Antimalarial Amphilectene Sergey V. Pronin and Ryan A. Shenvi Journal of the American Chemical Society 2012 134 (48), 19604-19606 doi:10.1021/ja310129b
- ^ Green, N. J.; Lawrence, A. L.; Bojase, G.; Willis, A. C.; Paddon-Row, M. N.; Sherburn, M. S. (2013). "Domino Cycloaddition Organocascades of Dendralenes". Angew. Chem. Int. Ed. 52: 8333–8336. doi:10.1002/anie.201302185.
- ^ Practical Synthesis and Reactivity of [3]Dendralene Tanya A. Bradford, Alan D. Payne, Anthony C. Willis, Michael N. Paddon-Row, and Michael S. Sherburn The Journal of Organic Chemistry 2010 75 (2), 491-494 doi:10.1021/jo9024557
- ^ Multicomponent Diene-Transmissive Diels–Alder Sequences Featuring Aminodendralenes S. M. Tan, A. C. Willis, M. N. Paddon-Row, M. S. Sherburn, Angew. Chem. Int. Ed. 2016, 55, 3081. doi:10.1002/anie.201510925
- ^ Synthesis and Diels–Alder Reactivity of Substituted [4]Dendralenes Mehmet F. Saglam, Ali R. Alborzi, Alan D. Payne, Anthony C. Willis, Michael N. Paddon-Row, and Michael S. Sherburn The Journal of Organic Chemistry 2016 81 (4), 1461-1475 doi:10.1021/acs.joc.5b02583
- ^ Bojase, Gomotsang; Nguyen, Thanh V.; Payne, Alan D.; Willis, Anthony C.; Sherburn, Michael S. (2011). "Synthesis and properties of the ivyanes: the parent 1,1-oligocyclopropanes". Chem. Sci. 2: 229–232. doi:10.1039/C0SC00500B.
- ^ Tetrahedron Letters Volume 35, Issue 29, 18 July 1994, Pages 5251–5252 A novel photochemical reaction of [3]dendralene derivatives Keiji Okada, Katsuji Maehara, Masaji Oda doi:10.1016/S0040-4039(00)77076-6
- ^ Transformation of azulenes to bicyclic [4]dendralene and heptafulvene derivatives via photochemical cycloaddition of dialkylsilylene Tomoyuki Kosai, Shintaro Ishida ,Takeaki Iwamoto Chem. Commun., 2015,51, 10707-10709 doi:10.1039/C5CC03424H




![[4]dendralene synthetic scheme from chloroprene](http://upload.wikimedia.org/wikipedia/commons/thumb/4/40/4-dendralene.svg/569px-4-dendralene.svg.png)
![[8]-dendralene synthesis](http://upload.wikimedia.org/wikipedia/commons/thumb/9/9d/8-dendralene.svg/411px-8-dendralene.svg.png)