Geodesic polyarene

A geodesic polyarene in organic chemistry is a polycyclic aromatic hydrocarbon with curved convex or concave surfaces.[1] Examples include fullerenes, nanotubes, corannulenes, helicenes and sumanene. The molecular orbitals of the carbon atoms in these systems are to some extent pyramidalized resulting a different pi electron density on either side of the molecule with consequences for reactivity.
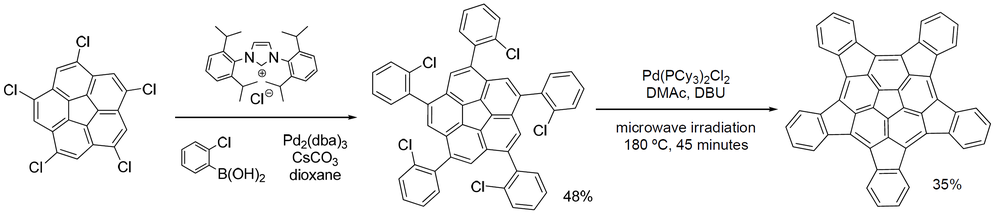
One member of this group of organic compounds, pentaindenocorannulene (depicted below),[2][3][4] can be considered a large fullerene fragment. The experimentally obtained curvature and degree of pyramidalizion (12.6° for the carbons of the pentagon at the center [5]) are both actually larger than that of fullerene but according to its discoverers, the compound is relatively easy to synthesize starting from corannulene and a way is opened to produce larger such fragments by stitching.
The crystal structure of pentaindenocorannulene has been obtained.[4] An illustration of the crystal packing for pentaindenocorannulene is given below.
Another geodesic polyarene that has been synthesized is C50H10.[6][4] C50H10 can be described as a short, rigid, structurally pure [5,5] carbon nanotube. The crystal structure of C50H10 has been obtained. The carbons of the pentagon at the center of C50H10 have a POAV angle of 12.3°, less than that of pentaindenocorannulene. The synthesis is as shown below. FVP stands for flash vacuum pyrolysis.
Some bowl-shaped molecules reported in the literature are in fact partially hydrogenated. A C56H40 hydrocarbon has been synthesized containing 54 of the 60 carbon atoms found in fullerene [7]
See also[edit]
References[edit]
- ^ Geodesic polyarenes with exposed concave surfaces Lawrence T. Scott,† Hindy E. Bronstein, Dorin V. Preda, Ronald B. M. Ansems, Matthew S. Bratcher and Stefan Hagen Pure Appl. Chem., Vol. 71, No. 2, pp. 209–219, 1999 Link
- ^ Pentaindenocorannulene and Tetraindenocorannulene: New Aromatic Hydrocarbon Systems with Curvatures Surpassing That of C60Edward A. Jackson, Brian D. Steinberg, Mihail Bancu, Atsushi Wakamiya, and Lawrence T. Scott J. Am. Chem. Soc.; 2007; 129(3) pp 484 - 485; (Communication) doi:10.1021/ja067487h
- ^ The two steps in this sequence are a Suzuki-Miyaura coupling with an ionic liquid and Tris(dibenzylideneacetone)dipalladium(0) and a Heck reaction with another palladium catalyst, DBU and DMAC
- ^ a b c Jackson, Edward A. (2008). Total Synthesis of a [5,5] Nanotube End-cap (PhD). Boston College. hdl:2345/1357.
- ^ by a so-called p-orbital axis vector (POAV). The value for fullerene is 11.6° and benzene 0°
- ^ Scott, L. T.; Jackson, E.; Zhang, Q.; Steinberg, B.; Bancu, M.; Li, B. (2012). "A Short, Rigid, Structurally Pure Carbon Nanotube by Stepwise Chemical Synthesis". Journal of the American Chemical Society. 134 (1): 107–110. doi:10.1021/ja209461g. PMID 22133011.
- ^ Synthesis of a C56H40 Hydrocarbon Bearing a 54-Carbon Framework of C60 Kung K. Wang, Yu-Hsuan Wang, Hua Yang, Novruz G. Akhmedov, and Jeffrey L. Petersen Org. Lett., 2009 doi:10.1021/ol900749a