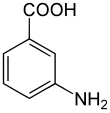
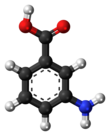
3-Aminobenzoic acid
Appearance
(Redirected from M-Aminobenzoic acid)
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
3-Aminobenzoic acid | |||
| Other names
meta-Aminobenzoic acid
m-Aminobenzoic acid MABA 3-Carboxyaniline m-Carboxyaniline | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.002.477 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C7H7NO2 | |||
| Molar mass | 137.13598 | ||
| Appearance | White solid | ||
| Density | 1.51 g/cm3 | ||
| Melting point | 178 to 180 °C (352 to 356 °F; 451 to 453 K) | ||
| Acidity (pKa) |
| ||
| Hazards | |||
| GHS labelling: | |||

| |||
| Warning | |||
| H302, H315, H319, H335 | |||
| P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
3-Aminobenzoic acid (also known as meta-aminobenzoic acid or MABA) is an organic compound with the molecular formula H2NC6H4CO2H. MABA is a white solid, although commercial samples are often colored. It is only slightly soluble in water. It is soluble in acetone, boiling water, hot alcohol, hot chloroform and ether. It consists of a benzene ring substituted with an amino group and a carboxylic acid.
It is prepared by reduction of 3-nitrobenzoic acid. It is used to prepare some dyes.[4]
See also
[edit]- Aminomethylbenzoic acid
- Anthranilic acid (2-aminobenzoic acid)
- 4-Aminobenzoic acid
- Arene substitution pattern
- Non-proteinogenic amino acids
References
[edit]- ^ m-Carboxyaniline - Compound Summary, PubChem.
- ^ 3-aminobenzoic acid, Medical Subject Headings database.
- ^ Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. p. 5–89. ISBN 978-1498754286.
- ^ Maki, Takao; Takeda, Kazuo (2000). "Benzoic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a03_555. ISBN 978-3-527-30385-4.