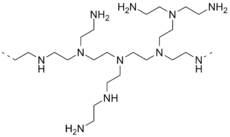
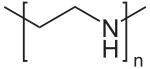
Polyethylenimine
| |
| Names | |
|---|---|
| IUPAC name
Poly(iminoethylene)
| |
| Other names
Polyaziridine, Poly[imino(1,2-ethanediyl)]
| |
| Identifiers | |
| ChemSpider |
|
| ECHA InfoCard | 100.123.818 |
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C2H5N)n, linear form | |
| Molar mass | 43.04 (repeat unit), mass of polymer variable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Polyethylenimine (PEI) or polyaziridine is a polymer with repeating units composed of the amine group and two carbon aliphatic CH2CH2 spacers. Linear polyethyleneimines contain all secondary amines, in contrast to branched PEIs which contain primary, secondary and tertiary amino groups. Totally branched, dendrimeric forms were also reported.[1] PEI is produced on an industrial scale and finds many applications usually derived from its polycationic character.[2]
Properties
[edit]The linear PEI is a semi-crystalline solid at room temperature while branched PEI is a fully amorphous polymer existing as a liquid at all molecular weights. Linear polyethyleneimine is soluble in hot water, at low pH, in methanol, ethanol, or chloroform. It is insoluble in cold water, benzene, ethyl ether, and acetone. Linear polyethyleneimine has a melting point of around 67 °C.[3] Both linear and branched polyethyleneimine can be stored at room temperature. Linear polyethyleneimine is able to form cryogels upon freezing and subsequent thawing of its aqueous solutions.[3]
Synthesis
[edit]Branched PEI can be synthesized by the ring opening polymerization of aziridine.[4] Depending on the reaction conditions different degree of branching can be achieved. Linear PEI is available by post-modification of other polymers like poly(2-oxazolines) [5] or N-substituted polyaziridines.[6] Linear PEI was synthesised by the hydrolysis of poly(2-ethyl-2-oxazoline)[7] and sold as jetPEI.[8] The current generation in-vivo-jetPEI uses bespoke poly(2-ethyl-2-oxazoline) polymers as precursors.[9]
Applications
[edit]Polyethyleneimine finds many applications in products like: detergents, adhesives, water treatment agents and cosmetics.[10] Owing to its ability to modify the surface of cellulose fibres, PEI is employed as a wet-strength agent in the paper-making process.[11] It is also used as flocculating agent with silica sols and as a chelating agent with the ability to complex metal ions such as zinc and zirconium.[12] There are also other highly specialized PEI applications:
Biology
[edit]PEI has a number of uses in laboratory biology, especially tissue culture, but is also toxic to cells if used in excess.[13][14] Toxicity is by two different mechanisms,[15] the disruption of the cell membrane leading to necrotic cell death (immediate) and disruption of the mitochondrial membrane after internalisation leading to apoptosis (delayed).
Attachment promoter
[edit]Polyethyleneimines are used in the cell culture of weakly anchoring cells to increase attachment. PEI is a cationic polymer; the negatively charged outer surfaces of cells are attracted to dishes coated in PEI, facilitating stronger attachments between the cells and the plate.
Transfection reagent
[edit]Poly(ethylenimine) was the second polymeric transfection agent discovered,[16] after poly-L-lysine. PEI condenses DNA into positively charged particles, which bind to anionic cell surface residues and are brought into the cell via endocytosis. Once inside the cell, protonation of the amines results in an influx of counter-ions and a lowering of the osmotic potential. Osmotic swelling results and bursts the vesicle releasing the polymer-DNA complex (polyplex) into the cytoplasm. If the polyplex unpacks then the DNA is free to diffuse to the nucleus.[17][18]
Permeabilization of gram negative bacteria
[edit]Poly(ethylenimine) is also an effective permeabilizer of the outer membrane of Gram-negative bacteria.[19]
CO2 capture
[edit]Both linear and branched polyethylenimine have been used for CO2 capture, frequently impregnated over porous materials. First use of PEI polymer in CO2 capture was devoted to improve the CO2 removal in space craft applications, impregnated over a polymeric matrix.[20] After that, the support was changed to MCM-41, an hexagonal mesostructured silica, and large amounts of PEI were retained in the so-called "molecular basket".[21] MCM-41-PEI adsorbent materials led to higher CO2 adsorption capacities than bulk PEI or MCM-41 material individually considered. The authors claim that, in this case, a synergic effect takes place due to the high PEI dispersion inside the pore structure of the material. As a result of this improvement, further works were developed to study more in depth the behaviour of these materials. Exhaustive works have been focused on the CO2 adsorption capacity as well as the CO2/O2 and CO2/N2 adsorption selectivity of several MCM-41-PEI materials with PEI polymers.[22][23] Also, PEI impregnation has been tested over different supports such as a glass fiber matrix [24] and monoliths.[25] However, for an appropriate performance under real conditions in post-combustion capture (mild temperatures between 45-75 °C and the presence of moisture) it is necessary to use thermally and hydrothermally stable silica materials, such as SBA-15,[26] which also presents an hexagonal mesostructure. Moisture and real world conditions have also been tested when using PEI-impregnated materials to adsorb CO2 from the air.[27]
A detailed comparison among PEI and other amino-containing molecules showed an excellent performance of PEI-containing samples with cycles. Also, only a slight decrease was registered in their CO2 uptake when increasing the temperature from 25 to 100 °C, demonstrating a high contribution of chemisorption to the adsorption capacity of these solids. For the same reason, the adsorption capacity under diluted CO2 was up to 90% of the value under pure CO2 and also, a high unwanted selectivity towards SO2 was observed.[28] Lately, many efforts have been made in order to improve PEI diffusion within the porous structure of the support used. A better dispersion of PEI and a higher CO2 efficiency (CO2/NH molar ratio) were achieved by impregnating a template-occluded PE-MCM-41 material rather than perfect cylindrical pores of a calcined material,[29] following a previously described route.[30] The combined use of organosilanes such as aminopropyl-trimethoxysilane, AP, and PEI has also been studied. The first approach used a combination of them to impregnate porous supports, achieving faster CO2-adsorption kinetics and higher stability during reutilization cycles, but no higher efficiencies.[31] A novel method is the so-called "double-functionalization". It is based on the impregnation of materials previously functionalized by grafting (covalent bonding of organosilanes). Amino groups incorporated by both paths have shown synergic effects, achieving high CO2 uptakes up to 235 mg CO2/g (5.34 mmol CO2/g).[32] CO2 adsorption kinetics were also studied for these materials, showing similar adsorption rates as impregnated solids.[33] This is an interesting finding, taking into account the smaller pore volume available in double-functionalized materials. Thus, it can be also concluded that their higher CO2 uptake and efficiency compared to impregnated solids can be ascribed to a synergic effect of the amino groups incorporated by two methods (grafting and impregnation) rather than to a faster adsorption kinetics.
Low work function modifier for electronics
[edit]Poly(ethylenimine) and poly(ethylenimine) ethoxylated (PEIE) have been shown as effective low-work function modifiers for organic electronics by Zhou and Kippelen et al.[34] It could universally reduce the work function of metals, metal oxides, conducting polymers and graphene, and so on. It is very important that low-work function solution-processed conducting polymer could be produced by the PEI or PEIE modification. Based on this discovery, the polymers have been widely used for organic solar cells, organic light-emitting diodes, organic field-effect transistors, perovskite solar cells, perovskite light-emitting diodes, quantum-dot solar cells and light-emitting diodes etc.
Use in delivery of HIV-gene therapies
[edit]Polyethylenimine (PEI), a cationic polymer, has been widely studied and shown great promise as an efficient gene delivery vehicle. Likewise, the HIV-1 Tat peptide, a cell-permeable peptide, has been successfully used for intracellular gene delivery.[35]
See also
[edit]- Tetraethylenepentamine
- Ethylenediamine
- Polyetherimide (Also goes by PEI)
- Polyimine
References
[edit]- ^ Yemul, Omprakash; Imae, Toyoko (2008). "Synthesis and characterization of poly(ethyleneimine) dendrimers". Colloid & Polymer Science. 286 (6–7): 747–752. doi:10.1007/s00396-007-1830-6. S2CID 98538201.
- ^ Davidson, Robert L.; Sittig, Marshall (1968). Water-soluble resins. Reinhold Book Corp. ISBN 978-0278916135.
- ^ a b Soradech, Sitthiphong; Williams, Adrian C.; Khutoryanskiy, Vitaliy V. (2022-10-24). "Physically Cross-Linked Cryogels of Linear Polyethyleneimine: Influence of Cooling Temperature and Solvent Composition". Macromolecules. 55 (21): 9537–9546. Bibcode:2022MaMol..55.9537S. doi:10.1021/acs.macromol.2c01308. ISSN 0024-9297. S2CID 253149614.
- ^ Zhuk, D. S., Gembitskii, P. A., and Kargin V. A. Russian Chemical Reviews; Vol 34:7.1965
- ^ Tanaka, Ryuichi; Ueoka, Isao; Takaki, Yasuhiro; Kataoka, Kazuya; Saito, Shogo (1983). "High molecular weight linear polyethylenimine and poly(N-methylethylenimine)". Macromolecules. 16 (6): 849–853. Bibcode:1983MaMol..16..849T. doi:10.1021/ma00240a003.
- ^ Weyts, Katrien F.; Goethals, Eric J. (1988). "New synthesis of linear polyethyleneimine". Polymer Bulletin. 19 (1): 13–19. doi:10.1007/bf00255018. S2CID 97101501.
- ^ Brissault, B.; et al. (2003). "Synthesis of Linear Polyethylenimine Derivatives for DNA Transfection". Bioconjugate Chemistry. 14 (3): 581–587. doi:10.1021/bc0200529. PMID 12757382.
- ^ "High Throughput Screening « Polyplus Transfection". Archived from the original on 2010-03-02. Retrieved 2010-04-02.
- ^ "Method for Manufacturing Linear Polyethylenimine (pei) for Transfection Purpose and Linear Pei Obtained with Such Method". Archived from the original on 2012-08-05.
- ^ Steuerle, Ulrich; Feuerhake, Robert (2006). "Aziridines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_239.pub2. ISBN 3527306730.
- ^ Wågberg, Lars (2000). "Polyelectrolyte adsorption onto cellulose fibres – a review". Nordic Pulp & Paper Research Journal. 15 (5): 586–597. doi:10.3183/NPPRJ-2000-15-05-p586-597. S2CID 4942367.
- ^ Madkour, Tarek M. (1999). Polymer Data Handbook. Oxford University Press, Inc. p. 490. ISBN 978-0195107890.
- ^ Vancha AR; et al. (2004). "Use of polyethyleneimine polymer in cell culture as attachment factor and lipofection enhancer". BMC Biotechnology. 4: 23. doi:10.1186/1472-6750-4-23. PMC 526208. PMID 15485583.
- ^ Hunter, A. C. (2006). "Molecular hurdles in polyfectin design and mechanistic background to polycation induced cytotoxicity". Advanced Drug Delivery Reviews. 58 (14): 1523–1531. doi:10.1016/j.addr.2006.09.008. PMID 17079050.
- ^ Moghimi, S. M.; et al. (2005). "A two-stage poly(ethylenimine)-mediated cytotoxicity: implications for gene transfer/therapy". Molecular Therapy. 11 (6): 990–995. doi:10.1016/j.ymthe.2005.02.010. PMID 15922971.
- ^ Boussif, O.; et al. (1995). "A Versatile Vector for Gene and Oligonucleotide Transfer into Cells in Culture and in vivo: Polyethylenimine". Proceedings of the National Academy of Sciences. 92 (16): 7297–7301. Bibcode:1995PNAS...92.7297B. doi:10.1073/pnas.92.16.7297. PMC 41326. PMID 7638184.
- ^ Rudolph, C; Lausier, J; Naundorf, S; Müller, RH; Rosenecker, J (2000). "In vivo gene delivery to the lung using polyethylenimine and fractured polyamidoamine dendrimers". Journal of Gene Medicine. 2 (4): 269–78. doi:10.1002/1521-2254(200007/08)2:4<269::AID-JGM112>3.0.CO;2-F. PMID 10953918. S2CID 31273799.
- ^ Akinc, A; Thomas, M; Klibanov, AM; Langer, R (2004). "Exploring polyethylenimine-mediated DNA transfection and the proton sponge hypothesis". Journal of Gene Medicine. 7 (5): 657–663. doi:10.1002/jgm.696. PMID 15543529. S2CID 25740208.
- ^ Helander, I. M.; Alakomi, H.-L.; Latva-Kala, K.; Koski, P. (1997-10-01). "Polyethyleneimine is an effective permeabilizer of Gram-negative bacteria". Microbiology. 143 (10). Microbiology Society: 3193–3199. doi:10.1099/00221287-143-10-3193. ISSN 1350-0872. PMID 9353921.
- ^ Satyapal, S.; Filburn, T.; Trela, J.; Strange, J. (2001). "Performance and Properties of a Solid Amine Sorbent for Carbon Dioxide Removal in Space Life Support Applications". Energy & Fuels. 15 (2): 250–255. doi:10.1021/ef0002391.
- ^ Xu, X.; Song, C.; Andrésen, J. M.; Miller, B. G.; Scaroni, A. W. (2002). "Novel Polyethylenimine-Modified Mesoporous Molecular Sieve of MCM-41 Type as High-Capacity Adsorbent for CO2Capture". Energy & Fuels. 16 (6): 1463–1469. doi:10.1021/ef020058u.
- ^ X. Xu, C. Song, R. Wincek J. M. Andrésen, B. G. Miller, A. W. Scaroni, Fuel Chem. Div. Prepr. 2003; 48 162-163
- ^ X. Xu, C. Song, B. G. Miller, A. W. Scaroni, Ind. Eng. Chem. Res. 2005; 44 8113-8119
- ^ Li, P.; Ge, B.; Zhang, S.; Chen, S.; Zhang, Q.; Zhao, Y. (2008). "CO2Capture by Polyethylenimine-Modified Fibrous Adsorbent". Langmuir. 24 (13): 6567–6574. doi:10.1021/la800791s. PMID 18507414.
- ^ C. Chen, S. T. Yang, W. S. Ahn, R. Ryoo, "Title" Chem. Commun. (2009) 3627-3629
- ^ Sanz, R.; Calleja, G.; Arencibia, A.; Sanz-Pérez, E. S. (2010). "CO2 adsorption on branched polyethyleneimine-impregnated mesoporous silica SBA-15". Appl. Surf. Sci. 256 (17): 5323–5328. Bibcode:2010ApSS..256.5323S. doi:10.1016/j.apsusc.2009.12.070.
- ^ Goeppert, A.; Czaun, M.; May, R. B.; Prakash, G. K. Surya; Olah, G. A.; Narayanan, S. R. (2011). "Carbon Dioxide Capture from the Air Using a Polyamine Based Regenerable Solid Adsorbent". Journal of the American Chemical Society. 133 (50): 20164–7. doi:10.1021/ja2100005. PMID 22103291.
- ^ Sanz-Pérez, E.S.; Olivares-Marín, M.; Arencibia, A.; Sanz, R.; Calleja, G.; Maroto-Valer, M.M. (2013). "CO2 adsorption performance of amino-functionalized SBA-15 under post-combustion conditions". Int. J. Greenh. Gas Control. 17: 366. doi:10.1016/j.ijggc.2013.05.011. hdl:10115/11746.
- ^ Heydari-Gorji, A.; Belmabkhout, Y.; Sayari, A. (2011). "Polyethylenimine-Impregnated Mesoporous Silica: Effect of Amine Loading and Surface Alkyl Chains on CO2Adsorption". Langmuir. 27 (20): 12411–6. doi:10.1021/la202972t. PMID 21902260.
- ^ Yue, M.B.; Sun, L.B.; Cao, Y.; Wang, Y.; Wang, Z.J.; Zhu, J.H. (2008). "Efficient CO2 Capturer Derived from As-Synthesized MCM-41 Modified with Amine". Chem. Eur. J. 14 (11): 3442–51. doi:10.1002/chem.200701467. PMID 18283702.
- ^ Choi, S.; Gray, M. L.; Jones, C.W. (2011). "Amine-tethered solid absorbents coupling high adsorption capacity and regenerability for CO2 capture from ambient air". ChemSusChem. 4 (5): 628–35. Bibcode:2011ChSCh...4..628C. doi:10.1002/cssc.201000355. PMID 21548105.
- ^ Sanz, R.; Calleja, G.; Arencibia, A.; Sanz-Pérez, E.S. (2013). "Development of high efficiency adsorbents for CO2 capture based on a double-functionalization method of grafting and impregnation". J. Mater. Chem. A. 1 (6): 1956. doi:10.1039/c2ta01343f.
- ^ Sanz, R.; Calleja, G.; Arencibia, A.; Sanz-Pérez, E.S. (2013). "CO2 Uptake and Adsorption Kinetics of Pore-Expanded SBA-15 Double-Functionalized with Amino Groups". Energy & Fuels. 27 (12): 7637. doi:10.1021/ef4015229.
- ^ Zhou, Y.; Fuentes-Hernandez, C.; Shim, J.; Meyer, J.; Giordano, A. J.; Li, H.; Winget, P.; Papadopoulos, T.; Cheun, H.; Kim, J.; Fenoll, M.; Dindar, A.; Haske, W.; Najafabadi, E.; Khan, T. M.; Sojoudi, H.; Barlow, S.; Graham, S.; Bredas, J.-L.; Marder, S. R.; Kahn, A.; Kippelen, B. (2012). "A Universal Method to Produce Low-Work Function Electrodes for Organic Electronics". Science. 336 (6079): 327–332. Bibcode:2012Sci...336..327Z. doi:10.1126/science.1218829. PMID 22517855. S2CID 9949593.
- ^ Yamano, Seiichi; Dai, Jisen; Hanatani, Shigeru; Haku, Ken; Yamanaka, Takuto; Ishioka, Mika; Takayama, Tadahiro; Yuvienco, Carlo; Khapli, Sachin; Moursi, Amr M.; Montclare, Jin K. (2014-02-01). "Long-term efficient gene delivery using polyethylenimine with modified Tat peptide". Biomaterials. 35 (5): 1705–1715. doi:10.1016/j.biomaterials.2013.11.012. ISSN 0142-9612. PMID 24268201.