Pseudo-oxocarbon anion
In chemistry, the term pseudo-oxocarbon anion is used to refer to a negative ion that is conceptually derived from an oxocarbon anion through replacement of one or more of the basic oxygen atoms by chemically similar elements or functional groups, such as sulfur (S), selenium (Se), or dicyanomethylene (=C(CN)2).
Typical examples are the anions 2-(Dicyanomethylene)croconate, croconate violet, and croconate blue, derived from the croconate anion C
5O2−
5 by replacing one, two, or three oxygen atoms by dicyanomethylene groups:

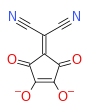
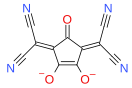

Croconate 2-Dicyanomethylene-
croconateCroconate violet Croconate blue
These anions retain many of the properties of the parent, including the delocalized bond in the ring and the delocalized charge in the atoms attached to the ring.[1][2][3] Similar anions can be obtained from squarate C
4O2−
4.[4]
See also[edit]
References[edit]
- ^ Alexander J. Fatiadi (1978), Synthesis of 1,3-(dicyanomethylene)croconate salts. New bond-delocalized dianion, "Croconate Violet". Journal of the American Chemical Society, volume 100 issue 8, pages 2586–2587. doi:10.1021/ja00476a073
- ^ Alexander J. Fatiadi (1980), Pseudooxocarbons. Synthesis of 1,2,3-tris(dicyanomethylene)croconate salts. A new bond-delocalized dianion, croconate blue. Journal of Organic Chemistry volume 45, pages 1338-1339. doi:10.1021/jo01295a044
- ^ Lawrence M. Doane, Alexander J. Fatiadi (2003) Electrochemical Oxidation of Croconate Salts; Evidence of the Chemical Equivalence of the Carbonyl Oxygen Atom and the Dicyanomethylene Group doi:10.1002/anie.198206351
- ^ Vanessa E. de Oliveira, Gustavo S. de Carvalho, Maria I. Yoshida, Claudio L. Donnici, Nivaldo L. Speziali, Renata Diniz and Luiz Fernando C. de Oliveira (2009), Bis(dicyanomethylene)squarate squaraines in their 1,2- and 1,3-forms: Synthesis, crystal structure and spectroscopic study of compounds containing alkali metals and tetrabutylammonium ions. Journal of Molecular Structure Volume 936, Issues 1-3, Pages 239-249 doi:10.1016/j.molstruc.2009.08.002




