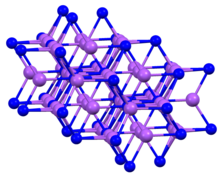
Sodium arsenide
| |
| Identifiers | |
|---|---|
| ECHA InfoCard | 100.031.762 |
CompTox Dashboard (EPA)
|
|
| Properties | |
| Na3As | |
| Molar mass | 143,89 g·mol−1 |
| Appearance | violet brown solid |
| Density | 2,36 g·cm−3 |
| hydrolysis | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium arsenide is the inorganic compound of sodium and arsenic with the formula Na3As. It is a dark colored solid that degrades upon contact with water or air. It is prepared by the reaction of the elements at 200–400 °C.[1] The compound is mainly of interest as exhibiting an archetypal structure. The normal pressure "sodium arsenide" phase is adopted by many alkali metal pnictides. At 3.6 gigapascals, Na3As adopts the Li3Bi structure, which is another archetypal structure.[2]
References
- ^ E. Dönges "Alkali Metals" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, New York. p. 986.
- ^ Beister, H.J.; Syassen, K.; Klein, J. "Phase transition of Na3As under pressure" Zeitschrift für Naturforschung B: Chemical Sciences 1990, volume 45, pp. 1388–1392. doi:10.1515/znb-1990-1007
