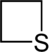
Thietane
Appearance
| |
| Names | |
|---|---|
| IUPAC name
Thietane
| |
| Other names
Thiacyclobutane
Trimethylene sulfide | |
| Identifiers | |
3D model (JSmol)
|
|
| 102383 | |
| ChemSpider | |
| ECHA InfoCard | 100.005.469 |
| EC Number |
|
PubChem CID
|
|
| UN number | 1993 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H6S | |
| Molar mass | 74.14 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Sulfurous |
| Density | 1.028 g cm-3 |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H225, H302 | |
| P210 | |
| NFPA 704 (fire diamond) | |
| Flash point | -11(9) °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thietane is a heterocyclic compound containing a saturated four-membered ring with three carbon atoms and one sulfur atom.[1][2]
References
- ^ Leśniak, S (2008). "Thietanes and Thietes: Monocyclic". Comprehensive Heterocyclic Chemistry III. 2.07: 389–428. doi:10.1016/B978-008044992-0.00207-8.
{{cite journal}}: Cite has empty unknown parameter:|1=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Block, E (1996). "Thietanes and Thietes: Monocyclic". Comprehensive Heterocyclic Chemistry II. 1.24: 773–802. doi:10.1016/B978-008096518-5.00024-1.
{{cite journal}}: Cite has empty unknown parameter:|1=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help)

