Tropinone
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
8-Methyl-8-azabicyclo[3.2.1]octan-3-one
| |||
| Other names
3-Tropinone
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ECHA InfoCard | 100.007.756 | ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| Properties | |||
| C8H13NO | |||
| Molar mass | 139.195 g/mol | ||
| Appearance | Brown solid | ||
| Melting point | 42.5 °C (108.5 °F; 315.6 K) | ||
| Boiling point | (decomposes) | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Tropinone is an alkaloid, famously synthesised in 1917 by Robert Robinson as a synthetic precursor to atropine, a scarce commodity during World War I.[1][2] Tropinone and the alkaloids cocaine and atropine all share the same tropane core structure.
Synthesis
The first synthesis of tropinone was by Richard Willstätter in 1901. It started from the seemingly related cycloheptanone, but required many steps and had an overall yield of only 0.75%.[3] Willstätter had previously synthesized cocaine from tropinone, in what was the first synthesis and elucidation of the structure of cocaine.[4]
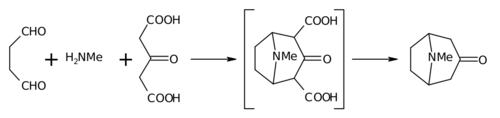
The 1917 synthesis by Robinson is considered a legend in total synthesis[5] due to its simplicity and biomimetic approach. Tropinone is a bicyclic molecule, but the reactants used in its preparation are fairly simple: succinaldehyde, methylamine and acetonedicarboxylic acid (or even acetone). The synthesis is a good example of a biomimetic reaction or biogenetic-type synthesis because biosynthesis makes use of the same building blocks. It also demonstrates a tandem reaction in a one-pot synthesis. Furthermore the yield of the synthesis was 17% and with subsequent improvements exceeded 90%.[3]
Reaction mechanism
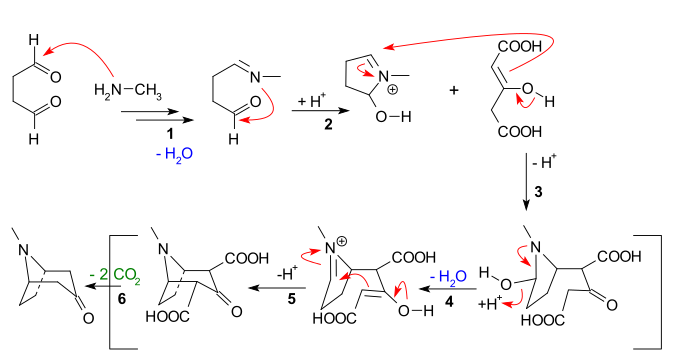
The main features apparent from the reaction sequence below are:
- Nucleophilic addition of secondary amine to aldehyde followed by loss of water to create an imine
- Nucleophilic addition of the imine to the second aldehyde unit and first ring closure
- Intermolecular Mannich reaction of the enolate of acetone dicarboxylate
- New enolate formation and new imine formation with loss of water for
- Second intramolecular mannich reaction and second ring closure
- Loss of 2 carboxylic groups to tropinone
References
- ^ R. Robinson (1917). "A synthesis of tropinone". Journal of the Chemical Society, Transaction. 111: 762–768. doi:10.1039/CT9171100762.
- ^ The Art and Science of Total Synthesis at the Dawn of the Twenty-First Century K. C. Nicolaou, Dionisios Vourloumis, Nicolas Winssinger, and Phil S. Baran Angew. Chem. Int. Ed. 2000, 39, 44 ± 122
- ^ a b Smit, W. et al. (1998) Organic Synthesis, The Science behind the Art. Cambridge: The Royal Society of Chemistry.
- ^ Andrew J. Humphrey and David O'Hagan. Tropane alkaloid biosynthesis. A century old problem unresolved. Natural Products Reports 2001, 18, 494-502.doi:10.1039/b001713m
- ^ Arthur J. Birch. Investigating a Scientific Legend: The Tropinone Synthesis of Sir Robert Robinson, F.R.S. Notes and Records of the Royal Society of London, 1993, 47, 277-296.