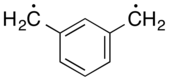
Xylylene
 |
 |
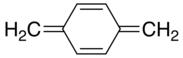 |
In organic chemistry, a xylylene (sometimes quinone-dimethide) is any of the constitutional isomers having the formula C6H4(CH2)2. These compounds are related to the corresponding quinones and quinone methides by replacement of the oxygen atoms by CH2 groups. ortho- and para-xylylene are best known, although neither is stable in solid or liquid form. The meta form is a diradical. Certain substituted derivatives of xylylenes are however highly stable, such as tetracyanoquinodimethane and the xylylene dichlorides.
p-Xylylene[edit]
p-Xylylene forms upon pyrolysis of p-xylene or, more readily, the α-substituted derivatives. p-Xylylene dimerizes with moderate efficiency to give p-cyclophane:[1]

Further heating of the p-cyclophane gives poly(para-xylylene).
o-Xylylenes[edit]
o-Xylylenes (o-quinodimethanes) are often generated in situ,[2] e.g., by the pyrolysis of the corresponding sulfone.[3] Another method involves 1,4-elimination of ortho benzylic silanes.[4] or stannanes,[5][6][7]
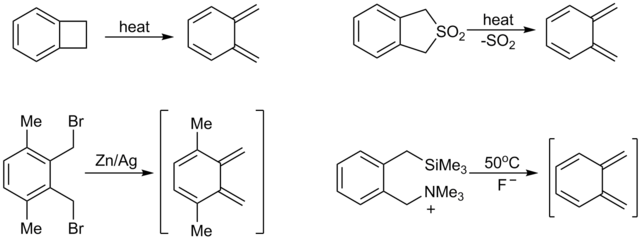
α,α'-ortho Xylene dibromides have been well developed for generating o-xylyenes.[8] For example, reaction of tetrabromo-o-xylene (C6H4(CHBr2)2) with sodium iodide affords α,α'-dibromo-o-xylylene, which can be trapped to give naphthylene derivatives. In the absence of trapping agents, the xylylene relaxes to α,α'-dibromobenzocyclobutane:[9]
- C6H4(CHBr2)2 + 2 NaI → C6H4(=CHBr)2 + 2 NaBr + I2
- C6H4(=CHBr)2 → C6H4(CHBr)2
Cycloadditions of these o-xylylenes provides a pathway to acenes.[10]
The diene unit formed by the two exocyclic alkene units of the ortho isomer can serve as a ligand in coordination complexes. For example, reaction of α,α'-dibromo-o-xylene with iron carbonyls affords low yields of the xylylene complex Fe(CO)3[η4-C6H4(CH2)2]. This product is structurally analogous to Fe(CO)3[η4-1,3-butadiene].[11]
At high temperatures, benzocyclobutenes undergo electrocyclic ring-opening to form o-xylylenes. This and other syntheses of o-xylylenes, and their subsequent dimerization by [4+4] cycloaddition to form cycloctyl structures, were used repeatedly in the synthesis of superphane.[12]
Electronic structure[edit]
Despite the observed chemistry of para-xylylene (i.e. its rapid polymerization to poly-p-xylylene), which suggests the compound exists as a diradical, physical evidence unanimously concludes that the lowest electronic state of p-xylylene is a closed shell singlet. Additionally, several computational methods confirm this assignment.[13] Conversely, meta-xylylene is a non-Kekulé molecule that has a triplet ground-state.[14]
References[edit]
- ^ H. E. Winberg, F. S. Fawcett "[2.2]Paracyclophane" Organic Syntheses, Coll. Vol. 5, p.883 (1973); Vol. 42, p.83 (1962) Link.
- ^ Klundt, I. L. (1970). "Benzocyclobutene and its derivatives". Chemical Reviews. 70 (4): 471–487. doi:10.1021/cr60266a002.
- ^ Nicolaou, K. C.; Snyder, S. A.; Montagnon, T.; Vassilikogiannakis, G. (2002). "The Diels-Alder Reaction in Total Synthesis". Angewandte Chemie International Edition. 41 (10): 1668–1698. doi:10.1002/1521-3773(20020517)41:10<1668::AID-ANIE1668>3.0.CO;2-Z. PMID 19750686.
- ^ Ito, Y.; Nakatsuka, M.; Saegusa, T. (1982). "Syntheses of polycyclic ring systems based on the new generation of o-quinodimethanes". Journal of the American Chemical Society. 104 (26): 7609–7622. doi:10.1021/ja00390a036.
- ^ Sano, H.; Ohtsuka, H.; Migita, T. (1988). "A convenient method for the generation of o-quinodimethanes by proton induced 1,4-elimination of o-(1-hydroxyalkyl)benzyltributylstannanes". Journal of the American Chemical Society. 110 (6): 2014–2015. doi:10.1021/ja00214a083.
- ^ Soon, H. W. (1993). "A novel method for the generation of o-quinodimethane by selenium-induced fragmentation of o-vinyl benzyltributylstannane". Tetrahedron Letters. 34 (47): 7587–7590. doi:10.1016/S0040-4039(00)60407-0.
- ^ Soon, H. W. (1994). "Lewis acid-promoted generation of α-oxy-o-quinodimethanes and cycloaddition reactions". Tetrahedron Letters. 35 (23): 3975–3978. doi:10.1016/S0040-4039(00)76717-7.
- ^ Rubottom, G. M.; Way, J. E. (1984). "An Improved Method for the Preparation of o-Quinodimethanes". Synthetic Communications. 14 (6): 507–514. doi:10.1080/00397918408059572.
- ^ Cava, M. P.; Deana, A. A.; Muth, K. (1959). "Condensed Cyclobutane Aromatic Compounds. VIII. The Mechanism of Formation of 1,2-Dibromobenzocyclobutene; A New Diels-Alder Synthesis". Journal of the American Chemical Society. 81 (24): 6458–6460. doi:10.1021/ja01533a032.
- ^ Paddon-Row, Michael N.; Patney, Harish K. (1986). "An Efficient Synthetic Strategy for Naphthalene Annellation of Norbornenylogous Systems". Synthesis. 1986 (4): 328–330. doi:10.1055/s-1986-31603.
- ^ Kerber, Robert C.; Ribakove, Everett C. (1991). "Formation of Iron Carbonyl Complexes of Reactive Polyenes from Dihalides involving the Free Polyene". Organometallics. 10 (8): 2848–2853. doi:10.1021/om00054a059.
- ^ Sekine, Y.; Brown, M.; Boekelheide, V. (1979). "[2.2.2.2.2.2](1,2,3,4,5,6)Cyclophane: superphane". Journal of the American Chemical Society. 101 (11): 3126–3127. doi:10.1021/ja00505a053.
- ^ Montgomery, L. K., Huffman, J. C., Jurczak, E. A. & Grendze, M. P. The molecular structures of Thiele’s and Chichibabin’s hydrocarbons. J. Am. Chem. Soc. 108, 6004–6011 (1986) doi:10.1021/ja00279a056
- ^ Steglich, Mathias; Custodis, Victoria B. F.; Trevitt, Adam J.; daSilva, Gabriel; Bodi, Andras; Hemberger, Patrick (2017). "Photoelectron Spectrum and Energetics of the meta-Xylylene Diradical". J. Am. Chem. Soc. 139 (41): 14348–14351. doi:10.1021/jacs.7b06714. PMID 28965395.
