Cyanine
Cyanines, also referred to as tetramethylindo(di)-carbocyanines[1] are a synthetic dye family belonging to the polymethine group. Although the name derives etymologically from terms for shades of blue, the cyanine family covers the electromagnetic spectrum from near IR to UV.
Chemically, cyanines are a conjugated system between two nitrogen atoms; in each resonance structure, exactly one nitrogen atom is oxidized to an iminium. Typically, they form part of a nitrogenous heterocyclic system.[2]
The main application for cyanine dyes is in biological labeling. Nevertheless, there is a wide literature on both their synthesis and uses, and cyanines are common in some CD and DVD media.
Structure[edit]

I = Streptocyanines,
II = Hemicyanines,
III = Closed cyanines
Cyanines have been classified in many ways:[3]
- Streptocyanines or open chain cyanines:
- R2N+=CH[CH=CH]n-NR2 (I)
- Hemicyanines:
- Aryl=N+=CH[CH=CH]n-NR2 (II)
- Closed chain cyanines:
- Aryl=N+=CH[CH=CH]n-N=Aryl (III)
Additionally, these classes are recognized:[4]
- Neutrocyanines:
- R2N+=CH[CH=CH]n-CN and R2N+=CH[CH=CH]n-CHO
- Merocyanines including spiropyrans and quinophthalones.
- Apocyanines
where two quaternary nitrogens are joined by a polymethine chain.[5] Both nitrogens may each be independently part of a heteroaromatic moiety, such as pyrrole, imidazole, thiazole, pyridine, quinoline, indole, benzothiazole, etc.
History and use in industry[edit]
Cyanines were first synthesized over a century ago. They were originally used, and still are, to increase the sensitivity range of photographic emulsions, i.e., to increase the range of wavelengths which will form an image on the film, making the film panchromatic.[4] Cyanines are also used in CD-R and DVD-R media. The ones used are mostly green or light blue colour, and are chemically unstable. For that reason, unstabilized cyanine discs are unsuitable for archival CD and DVD use. Recent cyanine discs contain stabilizers, typically a metal atom bonded to the cyanine molecule,[6] that slow the deterioration significantly. These discs are often rated with an archival life of 75 years or more. The other dyes used in CD-Rs are phthalocyanine and azo.
Use in biotechnology[edit]
For applications to biotechnology, special cyanine dyes are synthesized from 2, 3, 5 or 7-methine structures with reactive groups on either one or both of the nitrogen ends so that they can be chemically linked to either nucleic acids or protein molecules. Labeling is done for visualization and quantification purposes. Biological applications include comparative genomic hybridization and gene chips, which are used in transcriptomics, and various studies in proteomics such as RNA localization,[7] molecular interaction studies by fluorescence resonance energy transfer (FRET) and fluorescent immunoassays.
Cyanine dyes are available with different modifications such as methyl, ethyl or butyl substituents, carboxyl, acetylmethoxy, and sulfo groups which alter their hydrophilicity.[8]
| Probe | Ex (nm) | Em (nm) | MW | Quantum yield |
|---|---|---|---|---|
| Cy2 | 489 | 506 | 714 | QY 0.12 |
| Cy3 | (512);550 | 570;(615) | 767 | QY 0.15 [9]* |
| Cy3B | 558 | 572;(620) | 658 | QY 0.67 |
| Cy3.5 | 581 | 594;(640) | 1102 | QY 0.15 |
| Cy5 | (625);650 | 670 | 792 | QY 0.27[9] |
| Cy5.5 | 675 | 694 | 1128 | QY 0.28[10] |
| Cy7 | 743 | 767 | 818 | QY 0.28 |
Ex (nm): Excitation wavelength in nanometers
Em (nm): Emission wavelength in nanometers
MW: Molecular weight
QY: Quantum yield
* Depends strongly on viscosity, temperature, and biomolecular interactions.[11]
Common cyanine dyes and their uses[edit]
Because they yield brighter and more stable fluorescence, cyanines can advantageously replace conventional dyes such as fluorescein and rhodamines.
- Cy3 and Cy5 are the most popular, used typically combined for 2 colors detection.
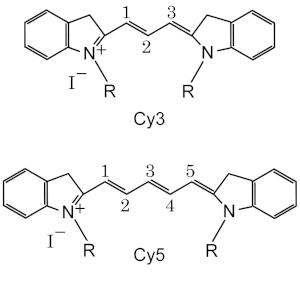
Cy3 fluoresces greenish yellow (~550 nm excitation, ~570 nm emission), while Cy5 is fluorescent in the far-red region (~650 excitation, 670 nm emission).[12] Cy3 can be detected by various fluorometers, imagers, and microscopes with standard filters for Tetramethylrhodamine (TRITC). Due to its high molar extinction coefficient, this dye is also easily detected by naked eye on electrophoresis gels, and in solution. Cy5 became a popular replacement for far red fluorescent dyes because of its high extinction coefficient (as small as 1 nanomol can be detected in gel electrophoresis by naked eye) and its fluorophore emission maximum in the red region, where many CCD detectors have maximum sensitivity and biological objects give low background interference.
The scanners actually use diverse laser emission wavelengths (typically 532 nm and 635 nm) and filter wavelengths (550-600 nm and 655-695 nm) to avoid background contamination. They are thus able to easily distinguish colors from Cy3 and from Cy5, and also able to quantify the amount of Cy3 and Cy5 labeling in one sample (multiparametric detection).
- Other cyanine dyes are useful:
Cy3.5 can replace sulfoRhodamine 101.
Cy5.5 is a near-infrared (IR) fluorescence-emitting dye (excitation/emission maximum 678/694 nm).
Cy7 is a near-IR fluor that is invisible to the naked eye (excitation/emission maximum 750/776 nm). It is used in in vivo imaging applications, as well as the Cy7.5 dye.
Sulfo–Cyanine dyes bear one or two sulfo groups, rendering the Cy dye water-soluble, but tri- and quadri-sulfonated forms are available for even higher water solubility.[8] PEGylation is another modification that confers hydrophilicity, not only to the dye but also to the labeled conjugate.
Nomenclature and structure[edit]
The Cy3 and Cy5 nomenclature was first proposed by Ernst, et al.[5] in 1989, and is non-standard since it gives no hint of their chemical structures. In the original paper the number designated the count of the methines (as shown), and the side chains were unspecified. Due to this ambiguity various structures are designated Cy3 and Cy5 in the literature. The R groups do not have to be identical. In the dyes as used they are short aliphatic chains one or both of which ends in a highly reactive moiety such as N-hydroxysuccinimide or maleimide.
Alternatives[edit]
Many analogs of standard Cy 2 / 3 / 3.5 / 5 / 5.5 / 7 / 7.5 dyes were developed, using diverse modification: Alexa Fluor dyes, Dylight, FluoProbes dyes, Sulfo Cy dyes,[13] Seta dyes,[14] IRIS dyes from Cyanine Technologies [15] and others can be used interchangeably with Cy dyes in most biochemical applications, with claimed improvements in solubility, fluorescence, or photostability.[16][17]
While patent protection for the standard Cy series of dyes has lapsed, the trademarked Cy naming remains in place. Consequently, dyes that are identical to Cy dyes, but called different names, are now sold.
Applications[edit]

Cyanine dyes are used to label proteins, antibodies, peptides, nucleic acid probes, and any kind of other biomolecules to be used in a variety of fluorescence detection techniques: Flow cytometry, Microscopy (mainly Visible range, but also UV, IR), Microplate assays, Microarrays, as well as "light-up Probes," and in vivo imaging.[18]
Nucleic acid labeling[edit]
In microarray experiments DNA or RNA is labeled with either Cy3 or Cy5 that has been synthesized to carry an N-hydroxysuccinimidyl ester (NHS-ester) reactive group. Since NHS-esters react readily only with aliphatic amine groups, which nucleic acids lack, nucleotides have to be modified with aminoallyl groups. This is done through incorporating aminoallyl-modified nucleotides during synthesis reactions. A good ratio is a label every 60 bases such that the labels are not too close to each other, which would result in quenching effects.
Protein labeling[edit]
For protein labeling, Cy3 and Cy5 dyes sometimes bear a succinimidyl group to react with amines, or a maleimide group to react with a sulfhydryl group of cysteine residues.
Cy5 is sensitive to its electronic environment. Changes in the conformation of the protein it is attached to will produce either enhancement or quenching of the emission. The rate of this change can be measured to determine enzyme kinetic parameters. The dyes can be used for similar purposes in FRET experiments.
Cy3 and Cy5 are used in proteomics experiments so that samples from two sources can be mixed and run together through the separation process.[19][20] This eliminates variations due to differing experimental conditions that are inevitable if the samples were run separately. These variations make it extremely difficult, if not impossible, to use computers to automate the acquisition of the data after the separation is complete. Using these dyes makes the automation trivial.
Etymology[edit]
The word cyanin is from the English word "cyan", which conventionally means a shade of blue-green (close to "aqua") and is derived from the Greek κυάνεος/κυανοῦς kyaneos/kyanous which means a somewhat different color: "dark blue".
References[edit]
- ^ Kvach, Maksim V.; Ustinov, Alexey V.; Stepanova, Irina A.; Malakhov, Andrei D.; Skorobogatyi, Mikhail V.; Shmanai, Vadim V.; Korshun, Vladimir A. (2008). "A Convenient Synthesis of Cyanine Dyes: Reagents for the Labeling of Biomolecules". European Journal of Organic Chemistry. 2008 (12): 2107–2117. doi:10.1002/ejoc.200701190. ISSN 1099-0690.
- ^ "Cyanine dyes". The IUPAC Compendium of Chemical Terminology. 2014. doi:10.1351/goldbook.C01487.
- ^ Kim, Eunha; Park, Seung Bum (2010). "Discovery of New Synthetic Dyes: Targeted Synthesis or Combinatorial Approach?". In Demchenko, Alexander P. (ed.). Advanced Fluorescence Reporters in Chemistry and Biology I: Fundamentals and Molecular Design Volume 8 of Springer Series on Fluorescence. Berlin: Springer. p. 172. ISBN 9783642047022.
- ^ a b Berneth, Horst (2008). "Methine Dyes and Pigments". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a16_487.pub2. ISBN 978-3527306732.
- ^ a b Ernst LA, Gupta RK, Mujumdar RB, Waggoner AS (Jan 1989). "Cyanine dye labeling reagents for sulfhydryl groups". Cytometry. 10 (1): 3–10. doi:10.1002/cyto.990100103. PMID 2917472.
- ^ "Archival Lifespan of TDK CD-R". cdrom2go.com. US Digital Media. Retrieved 3 April 2022.
- ^ Blower MD, Feric E, Weis K, Heald R (Dec 2007). "Genome-wide analysis demonstrates conserved localization of messenger RNAs to mitotic microtubules". The Journal of Cell Biology. 179 (7): 1365–73. doi:10.1083/jcb.200705163. PMC 2373496. PMID 18166649.
- ^ a b CYanine dyes
- ^ a b Mujumdar B, Ernst A, Mujumdar SR, Lewis CJ, Waggoner AS (Mar 1993). "Cyanine dye labeling reagents: Sulfoindocyanine succinimidyl esters". Bioconjugate Chemistry. 4 (2): 105–111. doi:10.1021/bc00020a001. PMID 7873641.
- ^ Umezawa K, Matsui A, Nakamura Y, Citterio D, Suzuki K (2009). "Bright, color-tunable fluorescent dyes in the Vis/NIR region: establishment of new "tailor-made" multicolor fluorophores based on borondipyrromethene". Chemistry: A European Journal. 15 (5): 1096–106. doi:10.1002/chem.200801906. PMID 19117043.
- ^ Levitus, Marcia; Ranjit, Suman (2011). "Cyanine dyes in biophysical research: the photophysics of polymethine fluorescent dyes in biomolecular environments". Quarterly Reviews of Biophysics. 44 (1): 123–151. doi:10.1017/S0033583510000247. ISSN 1469-8994. PMID 21108866. S2CID 7345293.
- ^ Jackson ImmunoResearch. "Cyanine Dyes (Cy2, Cy3, and Cy5)". Retrieved 2008-10-31.
- ^ "Cyandye, LLC". Archived from the original on 2018-10-03. Retrieved 2013-11-01.
- ^ SETA BioMedicals
- ^ "Iris Dyes | Cyanine Technologies". Archived from the original on 2015-01-26. Retrieved 2015-01-26.
- ^ FluoProbes488 comparison to FITC, Cyanine2
- ^ FluoProbes547H comparison in Confocal Microscopy
- ^ Armitage, Bruce A. (27 January 2005). "Cyanine Dye–DNA Interactions: Intercalation, Groove Binding, and Aggregation". DNA Binders and Related Subjects. Vol. 253. pp. 55–76. doi:10.1007/b100442. ISBN 978-3-540-22835-6.
{{cite book}}:|journal=ignored (help) - ^ Unlü M, Morgan ME, Minden JS (Oct 1997). "Difference gel electrophoresis: a single gel method for detecting changes in protein extracts". Electrophoresis. 18 (11): 2071–7. doi:10.1002/elps.1150181133. PMID 9420172. S2CID 604007.
- ^ Osterman IA, Ustinov AV, Evdokimov DV, Korshun VA, Sergiev PV, Serebryakova MV, Demina IA, Galyamina MA, Govorun VM, Dontsova OA (Jan 2013). "A nascent proteome study combining click chemistry with 2DE" (PDF). Proteomics. 13 (1): 17–21. doi:10.1002/pmic.201200393. PMID 23161590. S2CID 9002232. Archived from the original (PDF) on 2015-06-30. Retrieved 2015-06-27.
