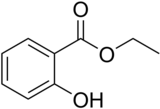
Ethyl salicylate
| |

| |
| Names | |
|---|---|
| IUPAC name
Ethyl 2-hydroxybenzoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.003.878 |
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H10O3 | |
| Molar mass | 166.17 g/mol |
| Density | 1.131 g/cm3 |
| Melting point | 1 °C (34 °F; 274 K) |
| Boiling point | 231 to 234 °C (448 to 453 °F; 504 to 507 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ethyl salicylate is the ester formed by the condensation of salicylic acid and ethanol. It is a clear liquid that is sparingly soluble in water, but soluble in alcohol and ether. It has a pleasant odor resembling wintergreen and is used in perfumery and artificial flavors.
See also
References
