User:Mr. Ibrahem/Midazolam
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /mɪˈdæzəlæm/ |
| Trade names | Dormicum, Hypnovel, Versed, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a609003 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular, intravenous, buccal, intranasal |
| Drug class | Benzodiazepine |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | By mouth (variable, around 40%)[1][2] intramuscular 90%+ |
| Protein binding | 97% |
| Metabolism | Liver 3A3, 3A4, 3A5 |
| Onset of action | Within 5 min (IV), 15 min (IM), 20 min (oral)[3] |
| Elimination half-life | 1.5–2.5 hours[4] |
| Duration of action | 1 to 6 hrs[3] |
| Excretion | Kidney |
| Identifiers | |
| |
| Chemical and physical data | |
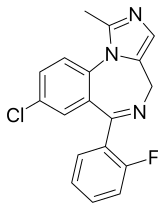
| Formula | C18H13ClFN3 |
| Molar mass | 325.78 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Midazolam, marketed under the trade name Versed, among others, is a benzodiazepine medication used for anesthesia, procedural sedation, trouble sleeping, and severe agitation.[3] It works by inducing sleepiness, decreasing anxiety, and causing a loss of ability to create new memories.[3] It is also useful for the treatment of seizures.[6] Midazolam can be given by mouth, intravenously, or injection into a muscle, by spraying into the nose, or through the cheek.[3][6] When given intravenously, it typically begins working within five minutes; when injected into a muscle, it can take fifteen minutes to begin working.[3] Effects last for between one and six hours.[3]
Side effects can include a decrease in efforts to breathe, low blood pressure, and sleepiness.[3] Tolerance to its effects and withdrawal syndrome may occur following long-term use.[7] Paradoxical effects, such as increased activity, can occur especially in children and older people.[7] There is evidence of risk when used during pregnancy but no evidence of harm with a single dose during breastfeeding.[8][9] It belongs to the benzodiazepine class of drugs and works by increasing the activity of the GABA neurotransmitter in the brain.[3]
Midazolam was patented in 1974 and came into medical use in 1982.[10] It is on the World Health Organization's List of Essential Medicines as an alternative to lorazepam.[11] It is available as a generic medication and is not very expensive.[8] The wholesale cost in the developing world of a vial is about US$0.35.[12] In many countries, it is a controlled substance.[3]
References[edit]
- ^ Heizmann P, Eckert M, Ziegler WH (2012). "Pharmacokinetics and bioavailability of midazolam in man". British Journal of Clinical Pharmacology. 16 Suppl 1: 43S–49S. doi:10.1111/j.1365-2125.1983.tb02270.x. PMC 1428091. PMID 6138080.
- ^ Johnson TN, Rostami-Hodjegan A, Goddard JM, Tanner MS, Tucker GT (September 2002). "Contribution of midazolam and its 1-hydroxy metabolite to preoperative sedation in children: a pharmacokinetic-pharmacodynamic analysis" (PDF). British Journal of Anaesthesia. 89 (3): 428–37. doi:10.1093/bja/aef213. PMID 12402721.
- ^ a b c d e f g h i j k l "Midazolam Hydrochloride". The American Society of Health-System Pharmacists. Archived from the original on 2015-09-05. Retrieved Aug 1, 2015.
- ^ "Midazolam Injection" (PDF). Medsafe. New Zealand Ministry of Health. 26 October 2012. Archived from the original (PDF) on 22 February 2016. Retrieved 6 April 2016.
- ^ "WHOCC - ATC/DDD Index". www.whocc.no. Archived from the original on 26 October 2020. Retrieved 22 September 2020.
- ^ a b Brigo F, Nardone R, Tezzon F, Trinka E (August 2015). "Nonintravenous midazolam versus intravenous or rectal diazepam for the treatment of early status epilepticus: A systematic review with meta-analysis". Epilepsy & Behavior. 49: 325–36. doi:10.1016/j.yebeh.2015.02.030. PMID 25817929.
- ^ a b Riss J, Cloyd J, Gates J, Collins S (August 2008). "Benzodiazepines in epilepsy: pharmacology and pharmacokinetics". Acta Neurologica Scandinavica. 118 (2): 69–86. doi:10.1111/j.1600-0404.2008.01004.x. PMID 18384456.
- ^ a b Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 21. ISBN 9781284057560.
- ^ "Midazolam use while Breastfeeding". Archived from the original on 5 September 2015. Retrieved 29 August 2015.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 539. ISBN 9783527607495. Archived from the original on 6 March 2019. Retrieved 2 March 2019.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Midazolam". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 29 August 2015.
