Anemonin: Difference between revisions
Appearance
Content deleted Content added
SMILESmaster (talk | contribs) removed incorrect SMILES |
Added new info with source, added paragraph |
||
| Line 40: | Line 40: | ||
}} |
}} |
||
'''Anemonin''' is a compound found in plants of the [[buttercup family]] (Ranunculaceae). It is the [[Dimer (chemistry)|dimerization]] product of the [[toxin]] [[protoanemonin]]<ref name="Hager" /> and is easily [[hydrolyse]]d to a [[dicarboxylic acid]].<ref name="TUGraz" /> |
'''Anemonin''' is a compound found in plants of the [[buttercup family]] (Ranunculaceae). It is the [[Dimer (chemistry)|dimerization]] product of the [[toxin]] [[protoanemonin]]<ref name="Hager" /> and is easily [[hydrolyse]]d to a [[dicarboxylic acid]].<ref name="TUGraz" /> |
||
[[File:Anemoninsäure.svg|thumb|left|The hydrolysis product of anemonin]]{{clear-left}} |
|||
The name of the substance comes from the plant genus ''[[Anemone]]'', from which it was first identified.<ref name="ChemOrgVerb" /> [[Antispasmodic]] and [[analgetic]] properties have been described.<ref name="w-online" /> |
The name of the substance comes from the plant genus ''[[Anemone]]'', from which it was first identified.<ref name="ChemOrgVerb" /> [[File:Anemoninsäure.svg|thumb|The hydrolysis product of anemonin|alt=]] |
||
== Potential Uses == |
|||
[[Antispasmodic]] and [[analgetic]] properties have been described.<ref name="w-online" /> |
|||
The compound appears to inhibit pigmentation synthesis, and has therefore been suggested as a potential candidate for cosmetic use.<ref>{{Cite web|url=https://www.sciencedirect.com/science/article/pii/S0923181107002575|title=ScienceDirect|website=www.sciencedirect.com|access-date=2019-04-03}}</ref> |
|||
== References == |
== References == |
||
Revision as of 18:17, 3 April 2019
| |
| |
| Names | |
|---|---|
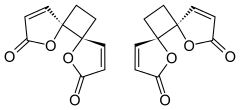
| IUPAC name
trans-4,7-Dioxadispiro[4.0.46.25]dodeca-1,9-diene-3,8-dione
| |
| Identifiers | |
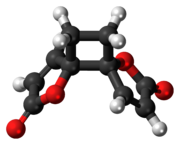
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H8O4 | |
| Molar mass | 192.170 g·mol−1 |
| Appearance | Colourless, odourless solid |
| Density | 1.45g/cm3 |
| Melting point | 158 °C (316 °F; 431 K) |
| Boiling point | 535.7 °C (996.3 °F; 808.9 K) @ 760mmHg |
| low | |
| Solubility in chloroform | soluble |
| Hazards | |
| Flash point | 300.7 °C (573.3 °F; 573.8 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
150 mg·kg−1 (mouse, i. p.) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Anemonin is a compound found in plants of the buttercup family (Ranunculaceae). It is the dimerization product of the toxin protoanemonin[1] and is easily hydrolysed to a dicarboxylic acid.[2]
The name of the substance comes from the plant genus Anemone, from which it was first identified.[3]

Potential Uses
Antispasmodic and analgetic properties have been described.[4]
The compound appears to inhibit pigmentation synthesis, and has therefore been suggested as a potential candidate for cosmetic use.[5]
References
- ^ List, PH; Hörhammer, L, eds. (1979). Hagers Handbuch der pharmazeutischen Praxis (in German) (4 ed.). Springer Verlag. ISBN 3-540-07738-3.
- ^ "Aktuelles aus der Natur" (PDF) (in German). TU Graz. 2 April 2009. p. 4. Retrieved 27 November 2010.[permanent dead link]
- ^ Chemie der organischen Verbindungen, Carl Löwig Template:De icon
- ^ Anemonin, Wissenschaft online Template:De icon
- ^ "ScienceDirect". www.sciencedirect.com. Retrieved 2019-04-03.
